Germany Healthcare Contract Development And Manufacturing Organization Market Size & Forecast:
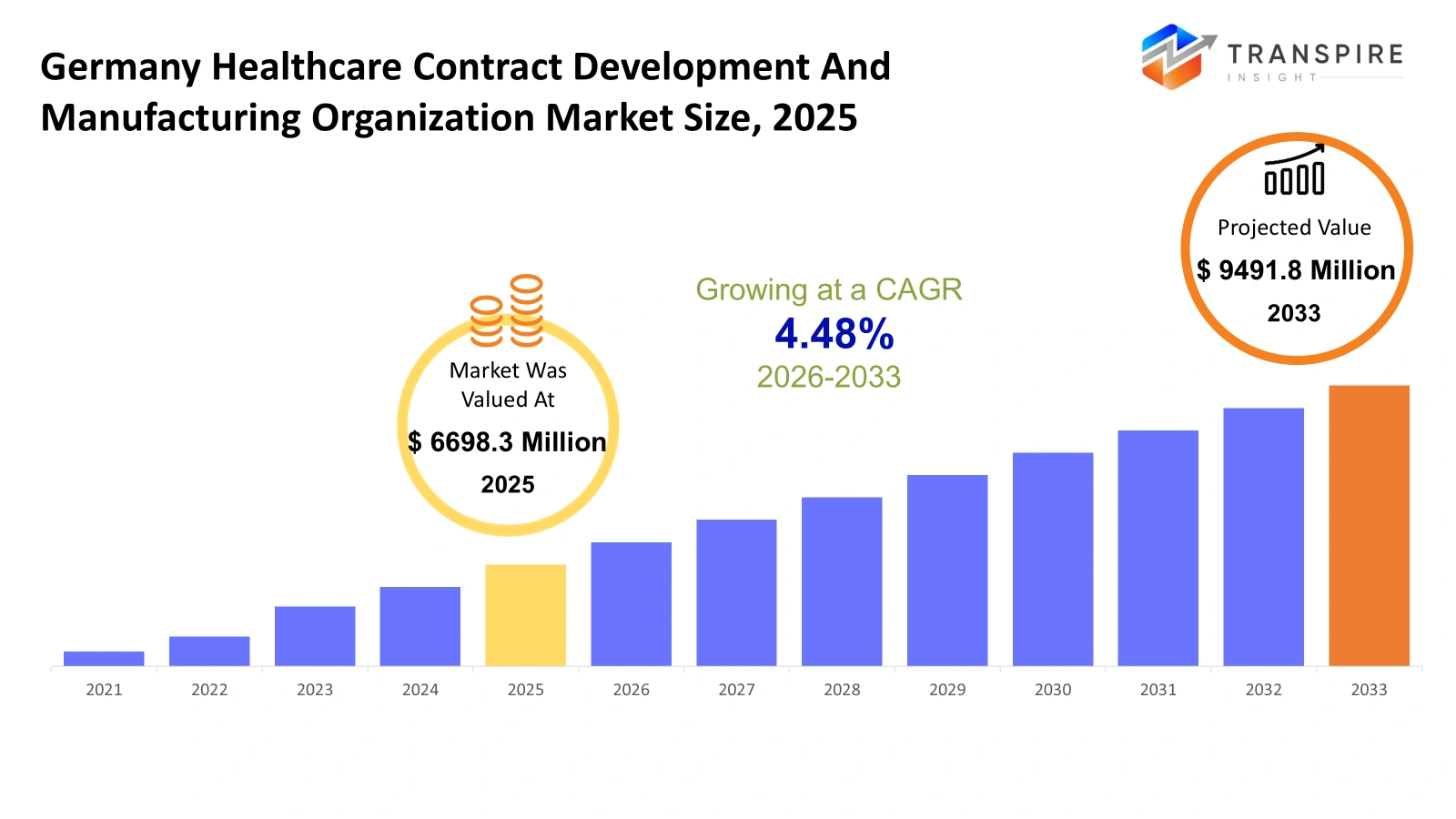
- Germany Healthcare Contract Development And Manufacturing Organization Market Size 2025: USD 6698.3 Million
- Germany Healthcare Contract Development And Manufacturing Organization Market Size 2033: USD 9491.8 Million
- Germany Healthcare Contract Development And Manufacturing Organization Market CAGR: 4.48%
- Germany Healthcare Contract Development And Manufacturing Organization Market Segments: ByService Type (API Development, Formulation Development, Clinical & Commercial Manufacturing, Packaging & Labeling), By Drug Type (Small Molecule, Biologics), By End User (Pharma Companies, Biotech Firms)
To learn more about this report, Download Free Sample Report
Germany Healthcare Contract Development And Manufacturing Organization Market Summary:
The Germany Healthcare Contract Development And Manufacturing Organization Market size is estimated at USD 6698.3 Million in 2025 and is anticipated to reach USD 9491.8 Million by 2033, growing at a CAGR of 4.48% from 2026 to 2033. The German healthcare contract development and manufacturing organization market operates within the pharmaceutical and biotechnology services sector which helps drug developers who need flexible production agreements. The increasing diversity of drug pipelines together with shortened development periods will lead German sponsors to outsource their formulation trials and sterile filling and small-batch biologics manufacturing needs. Customers require businesses to provide them with open access to quality information along with digital batch documentation and more efficient procedures for scaling production from laboratory-sized quantities to full commercial output. The introduction of automation and continuous processing equipment and advanced analytics will transform plant operations which enable partners to control intricate therapies that include cell and gene treatments. German regulators will establish new requirements for documentation and sustainability practices and systems which track product movement throughout the supply chain.
Key Market Trends & Insights:
The pharmaceutical industry in Germany will choose their outsourcing partners because they require specialized abilities to handle drugs that need advanced molecule extraction methods and sterile production environments and manufacturing control rules which must comply with European standards and documentation requirements.
The workflow of contract development and production will undergo a digital transformation because of manufacturing technologies. The facilities will implement systems which include automated quality monitoring and electronic batch records and predictive maintenance technology. The new technologies will enhance operational transparency while diminishing production downtime and enabling rapid regulatory documentation processes which will establish better collaboration pathways between pharmaceutical companies and their manufacturing partners.
Outsourcing partnerships will start evaluating sustainability requirements as primary decision-making factors. Pharmaceutical companies will seek manufacturers that adopt energy-efficient facilities, waste-reduction systems and environmentally responsible sourcing practices. German service providers will invest in greener operations to align with European environmental policies and the growing corporate focus on responsible pharmaceutical production.
Pharmaceutical companies will require flexible manufacturing capacity because their product pipelines are now expanding into new areas. The need for personalized medicines and smaller clinical batches with rapid development times will create demand for production facilities that can quickly change between different products. German manufacturers will increase their capacity through modular production lines while providing specialized development services to meet the needs of new therapeutic methods emerging in the market.
Germany Healthcare Contract Development And Manufacturing Organization Market Segmentation
By Service Type
API Development-API development services will support pharmaceutical innovators during early-stage drug creation. German service providers will offer synthesis optimization, analytical testing, and scalable process design. Clients will use specialized laboratories to develop active ingredients which require both regulatory documentation maintenance and quality control procedures throughout all development stages.
Formulation Development-Formulation development will focus on transforming active ingredients into stable and effective dosage forms. German service providers will assist with solubility improvement, stability studies, and delivery system design. Pharmaceutical companies will increasingly seek partners who can streamline their formulation optimization process while adhering to European safety and manufacturing requirements.
Clinical & Commercial Manufacturing- Clinical and commercial manufacturing services will enable pharmaceutical developers to scale their production processes from trial batches to full commercial production. German facilities will use modern production methods and sterile processing spaces together with adaptable production capacities to help clinical studies and new treatment introductions.
Packaging & Labeling- European pharmaceutical distribution operations will meet their essential regulatory requirements through packaging and labeling services. The German manufacturers will supply tamper-evident packaging together with serialization systems and multilingual labeling solutions. Pharmaceutical companies will depend on specialized partners to achieve safety standard compliance while they distribute products through domestic and international markets. 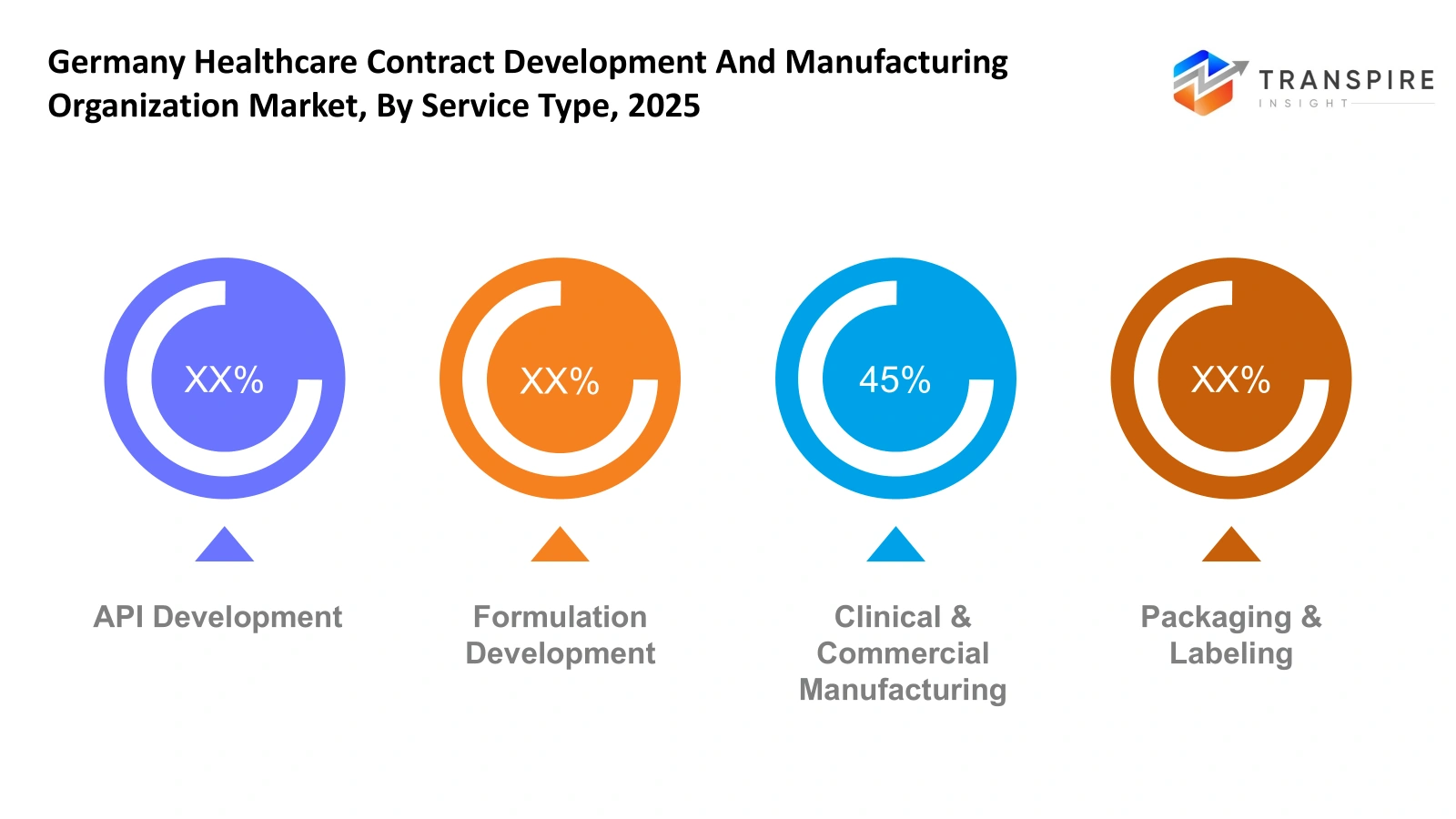
To learn more about this report, Download Free Sample Report
By Drug Type
Small Molecule-Chemical synthesis and industrial production facilities will remain essential for continued development of small molecule medications. The German development team will deliver solutions for process improvement and purification methods and they will establish uniformity in their quality evaluation procedures. Pharmaceutical companies will rely on trustworthy manufacturers who possess industry expertise to maintain production processes and achieve regulatory requirements throughout international pharmaceutical distribution networks.
Biologics-Biologics production will require sophisticated manufacturing techniques that utilize living organisms and cutting-edge biotechnological equipment. German research institutions will allocate funding towards creating dedicated bioreactor systems and controlled research laboratories and testing equipment for scientific investigations. Pharmaceutical and biotechnology organizations will partner with established companies to create and verify and manufacture biologic treatments for market distribution.
By End User
Pharma Companies-Pharmaceutical companies will increasingly outsource development and manufacturing activities to specialized partners in Germany. The partnerships will develop operational capacities that enable quicker drug development processes. Service providers will offer integrated support, from early research stages to commercial-scale production, which enables pharmaceutical companies to concentrate on developing new ideas and building their market presence.
Biotech Firms-Biotechnology companies will require outside assistance for their development and production needs because their internal production capabilities are insufficient. German service providers will offer technical support together with regulatory expertise and adaptable manufacturing space. The partnership will help new biotech companies progress their research projects toward obtaining clinical testing and market approval.
Country Insights
The pharmaceutical manufacturing sector in Germany receives its advantages from established research institutions and modern infrastructure and its adherence to stringent quality standards. Drug innovators in Germany will use outsourcing partnerships to achieve their goals of developing new drugs within shorter time frames while maintaining dependable manufacturing processes.
The German regulatory bodies will exercise their power to oversee pharmaceutical manufacturing operations which will motivate companies to work together with dedicated service providers. Manufacturing partners must uphold their documentation requirements and traceability systems and validated production processes because of European Medicines Agency compliance standards.
The specialized development services will experience increased demand because German research clusters continue to develop new biotechnology innovations. The combination of universities and research hospitals and private laboratories will develop new therapeutic candidates which need external partners to assist with formulation design and testing programs and manufacturing at scale.
The pharmaceutical industry in Germany will establish manufacturing partnerships which will be determined by the country's strong export orientation. German service providers will support international drug developers by offering high-quality production environments and reliable logistics networks and expertise which complies with European and global pharmaceutical regulations.
Recent Development News
German contract manufacturer Lyocontract and equipment partner Syntegon have started their expansion project which will create a new facility that produces sterile freeze-dried pharmaceuticals. The Ilsenburg project will establish an automated vial filling and lyophilisation system which meets different container size requirements and regulatory standards, with its main equipment scheduled for delivery during the summer of 2027.
Vetter, a CDMO with a long history, has reported its plans to invest in multiple stages for a new production facility in Saarlouis, Germany, which will receive from EU state aid. The first construction phase which will begin in mid-2026, will establish commercial injectable manufacturing capacity to provide advanced aseptic fill-finish services for global clients. This development will create approximately 2000 employment opportunities that will last until 2031.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 6698.3 Million |
|
Market size value in 2026 |
USD 6982.1 Million |
|
Revenue forecast in 2033 |
USD 9491.8 Million |
|
Growth rate |
CAGR of 4.48% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Germany |
|
Key company profiled |
Lonza Group, Catalent Inc., Boehringer Ingelheim, Siegfried Holding AG, Thermo Fisher Scientific (Patheon), Recipharm AB, WuXi AppTec, Samsung Biologics, Piramal Pharma Solutions, CordenPharma, Baxter BioPharma Solutions, Fareva Group, Aenova Group, Delpharm. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Service Type (API Development, Formulation Development, Clinical & Commercial Manufacturing, Packaging & Labeling), By Drug Type (Small Molecule, Biologics), By End User (Pharma Companies, Biotech Firms) |
Key Germany Healthcare Contract Development And Manufacturing Organization Market Insights
The Germany Healthcare Contract Development And Manufacturing Organization Market creates an ongoing impact on the country's developing wellness economy because people choose to practice mindful movement throughout their daily activities. The yoga industry will develop three types of practice spaces including urban studios, digital platforms and wellness retreats which will meet the increasing demand for flexible practice options. The yoga industry in Germany will develop through three main factors which include technological integration and professional instructor certification standards and sustainable product preferences.
Company List
- Lonza Group
- Catalent Inc.
- Boehringer Ingelheim
- Siegfried Holding AG
- Thermo Fisher Scientific (Patheon)
- Recipharm AB
- WuXi AppTec
- Samsung Biologics
- Piramal Pharma Solutions
- CordenPharma
- Baxter BioPharma Solutions
- Fareva Group
- Aenova Group
- Delpharm
Germany Healthcare Contract Development And Manufacturing Organization Market Report Segmentation
By Service Type
- API Development
- Formulation Development
- Clinical & Commercial Manufacturing
- Packaging & Labeling
By Drug Type
- Small Molecule
- Biologics
By End User
- Pharma Companies
- Biotech Firms
Frequently Asked Questions
Find quick answers to common questions.
The approximate Germany Healthcare Contract Development And Manufacturing Organization Market size for the market will be USD 9491.8 Million in 2033.
Key segments for the Germany Healthcare Contract Development And Manufacturing Organization Market By Service Type (API Development, Formulation Development, Clinical & Commercial Manufacturing, Packaging & Labeling), By Drug Type (Small Molecule, Biologics), By End User (Pharma Companies, Biotech Firms)
Major players in the Germany Healthcare Contract Development And Manufacturing Organization Market are Lonza Group, Catalent Inc., Boehringer Ingelheim, Siegfried Holding AG, Thermo Fisher Scientific (Patheon), Recipharm AB, WuXi AppTec, Samsung Biologics, Piramal Pharma Solutions, CordenPharma, Baxter BioPharma Solutions, Fareva Group, Aenova Group, Delpharm.
The Germany Healthcare Contract Development And Manufacturing Organization Market size is USD 6698.3 Million in 2025.
The Germany Healthcare Contract Development And Manufacturing Organization Market CAGR is 4.48%.
- Lonza Group
- Catalent Inc.
- Boehringer Ingelheim
- Siegfried Holding AG
- Thermo Fisher Scientific (Patheon)
- Recipharm AB
- WuXi AppTec
- Samsung Biologics
- Piramal Pharma Solutions
- CordenPharma
- Baxter BioPharma Solutions
- Fareva Group
- Aenova Group
- Delpharm
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


