France Bioregenerative Aesthetic Injectable Market Size & Forecast:
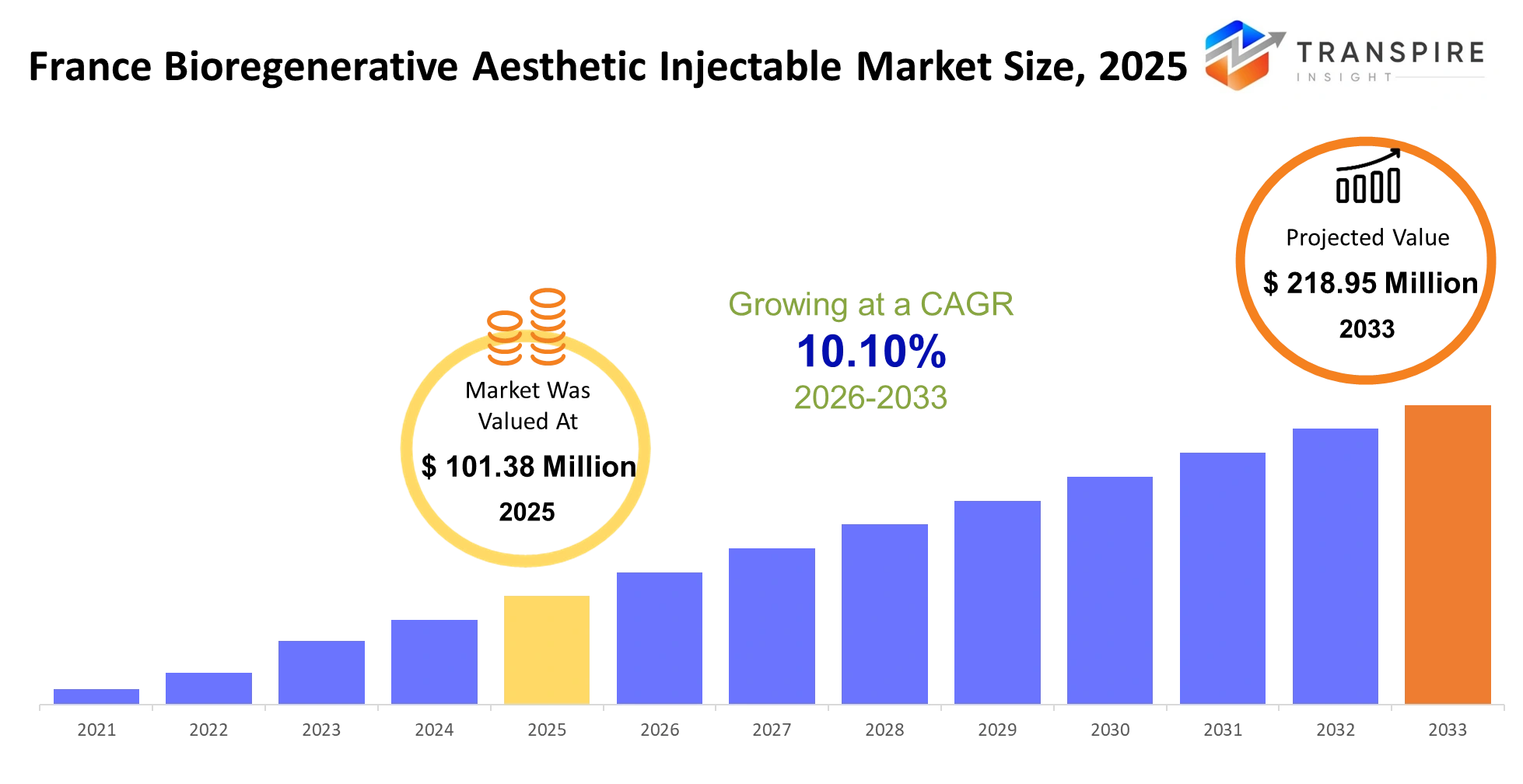
- France Bioregenerative Aesthetic Injectable Market Size 2025: USD 101.38 Million
- France Bioregenerative Aesthetic Injectable Market Size 2033: USD 218.95 Million
- France Bioregenerative Aesthetic Injectable Market CAGR: 10.10%
- France Bioregenerative Aesthetic Injectable Market Segments: By Type (Dermal Fillers, Collagen Stimulators, PRP Injectables, Others); By Application (Anti-aging, Wrinkle Reduction, Skin Rejuvenation, Facial Contouring, Others); By End-User (Dermatology Clinics, Hospitals, Cosmetic Centers, Aesthetic Clinics, Others); By Distribution (Clinics, Online Channels, Others).
To learn more about this report, Download Free Sample Report
France Bioregenerative Aesthetic Injectable Market Summary
The France Bioregenerative Aesthetic Injectable Market was valued at USD 101.38 Million in 2025. It is forecast to reach USD 218.95 Million by 2033. That is a CAGR of 10.10% over the period.
The market enables dermatology clinics and medical aesthetic practices to achieve skin restoration through injectable treatments which include hyaluronic acid boosters and collagen-stimulating compounds and platelet-rich plasma and new bio-derived exosome therapies. The solutions do not serve as volume enhancers because they activate the body to initiate its natural healing processes which result in better skin elasticity and texture and sustained dermal health.
The industry has experienced a complete structural change during the last three to five years because customers now demand natural-looking results that last longer which has replaced traditional volumizing fillers with regenerative biostimulatory injectables. The recovery process for elective aesthetic procedures after COVID-19 combined with hyaluronic acid import restrictions in 2021-2022 and stricter EU medical aesthetic compliance regulations served as the main catalyst for this change. The two forces compelled clinics to expand their treatment options while they increased their use of biologically active injectables which helped them maintain revenue stability and deliver premium services.
Key Market Insights
- The France Bioregenerative Aesthetic Injectable Market exists to serve Île-de-France which controls 40 to 45 percent of market share through its extensive network of cosmetic dermatology facilities.
- The region of Occitanie will experience its strongest growth until 2033 because biotech incubators operate in both Toulouse and Montpellier.
- The expansion of regional markets gets supported by the growing number of aesthetic clinics which operate in cities throughout France.
- Hyaluronic acid-based biostimulatory injectables dominate with the largest share due to widespread clinical adoption and predictable outcomes.
- The second largest segment of the market consists of collagen-stimulating injectables which customers prefer because they produce long-lasting regenerative results.
- Exosome and peptide-based injectables represent the fastest expanding market segment because France Bioregenerative Aesthetic Injectable Market research and development activities accelerate.
- Over half of the market share goes to facial rejuvenation applications because consumers demand anti-aging treatments and skin texture enhancement procedures.
- The dermatology-led aesthetic procedures have created a growing demand for skin regeneration and scar repair applications.
- Dermatology clinics maintain their dominant market position through their superior treatment capabilities and advanced aesthetic treatment facilities.
- The end-user segment of medical spas and cosmetic centers shows the highest growth rate because more consumers choose outpatient medical procedures.
- The main participants in the market include Allergan Aesthetics which operates under AbbVie Galderma Merz Pharmaceuticals Teoxane and Ipsen.
- Companies are strengthening the France Bioregenerative Aesthetic Injectable Market through advanced R&D in bioactive formulations.
- Strategic collaborations with dermatology networks are expanding clinical adoption and brand penetration.
What are the Key Drivers, Restraints, and Opportunities in the France Bioregenerative Aesthetic Injectable Market?
The France Bioregenerative Aesthetic Injectable Market experiences its primary market drive through the dermatology field's movement toward regenerative treatments which now attract patients who want procedures to create permanent skin enhancements through collagen and tissue regeneration.
The EU medical device and biologics regulation system creates a fundamental obstacle because it requires extensive testing through its approval system which delays new injectable products from reaching the market. The first barrier requires long clinical testing periods plus safety data collection and post-market monitoring obligations. The France Bioregenerative Aesthetic Injectable Market suffers from market entry delays which affect multiple new product launches thus decreasing product variety while hindering revenue growth for manufacturers. The restriction of product options at clinics leads to treatment limitations which prevent healthcare providers from creating personalized treatment strategies.
The establishment of biotech-led aesthetic innovation hubs in Île-de-France and Occitanie provides startups with a major business opportunity to create exosome-based and peptide-engineered injectables. The European life sciences industry is increasing its investments while dermatology clinics form partnerships with biotech companies to boost their clinical research activities.
What Has the Impact of Artificial Intelligence Been on the France Bioregenerative Aesthetic Injectable Market?
The France Bioregenerative Aesthetic Injectable Market experiences transformation through Artificial Intelligence which enhances treatment accuracy and clinic operational capacity and provides dermatology and aesthetic clinics with better patient outcome projections. The current operational capacity of AI facial analysis systems enables clinics to utilize these technologies for their skin assessment needs which include determining facial volume loss and creating custom injectable treatment plans. The process enhances patient flow through the clinic while optimizing revenue generation during each patient visit through its ability to decrease consultation durations and create uniform treatment methods.
The use of machine learning models has expanded to predict how patients will respond to biostimulatory injectables which include collagen-stimulating fillers and regenerative skin boosters. The systems use historical treatment outcome data to enable practitioners to select optimal dosage levels which will minimize overcorrection risk while achieving better long-term aesthetic outcomes. Clinics use AI to enhance their inventory management systems because it enables them to create accurate demand forecasts which result in better management of high-value injectable products while decreasing product waste.
Early adopters report that their operational performance improves through AI-powered patient visualization technologies, which increase scheduling efficiency and boost treatment conversion rates by creating standardized consultation processes. The process of establishing standardized clinical databases together with European Union data protection regulations creates obstacles which prevent researchers from conducting extensive model development activities. The high expenses associated with implementing advanced imaging technologies create obstacles that hinder small aesthetic clinics from using these systems which results in restricted AI adoption throughout the France Bioregenerative Aesthetic Injectable Market.
Key Market Trends
- Clinics across France increased adoption of regenerative injectables post-2022, which resulted in their replacement of traditional fillers with collagen-stimulating formulations that deliver longer-lasting results.
- Dermatology practices began to use exosome-based injectables as their main skin regeneration treatment method after 2023 because they wanted to use biologically active skin regeneration therapies.
- Île-de-France clinics shifted over 40% of treatment portfolios toward biostimulatory injectables between 2021 and 2025 because of rising demand for premium aesthetic treatments.
- The EU imposed stricter regulations between 2022 and 2024, which led to increased use of domestically available regenerative injectable products that met regulatory standards instead of imported cosmetic fillers.
- COVID-19 recovery brought increased demand for minimally invasive anti-aging procedures, which resulted in higher outpatient aesthetic procedure volumes throughout France.
- The 2024 expansion of regenerative injectable pipelines by Galderma and Merz Pharmaceuticals created an intense competitive environment for collagen-boosting formulation development.
- Dermatology clinics began providing their doctors with AI-assisted facial mapping tools in 2023 as a method to create accurate treatment plans for injectable procedures while handling patient requirements.
- Clinics started to create multiple bioregenerative product sourcing methods after supply chain problems caused hyaluronic acid imports to halt during the period from 2021 to 2022.
- Medical spas developed swiftly in tier-2 French cities after 2023, which allowed more people to access advanced injectable treatments that were previously only available in major urban areas.
- The French research institutes established biotech partnerships which started to accelerate their development process for injectable products that contain peptide and exosome-based technology after 2024.
France Bioregenerative Aesthetic Injectable Market Segmentation
By Type
- The France Bioregenerative Aesthetic Injectable Market has its biggest market share from dermal fillers because dermatology clinics use these products for facial volume restoration and facial shape correction. The combination of high practitioner experience with the procedures and predictable aesthetic results has enabled their continued success in both premium clinics and mid-tier clinics. Their established market leadership remains strong because elective procedures require no reimbursement and their clinical protocols are widely recognized which increases their market strength against emerging regenerative therapies.
- Collagen stimulators are experiencing rapid market growth because more people now prefer permanent body enhancements that last over temporary body volume increases. Clinics increasingly use these injectables for progressive skin quality improvement, driven by patient demand for natural-looking results and reduced retreatment frequency. PRP injectables show steady adoption in niche dermatology and hair restoration applications, although variability in clinical outcomes limits broader penetration.
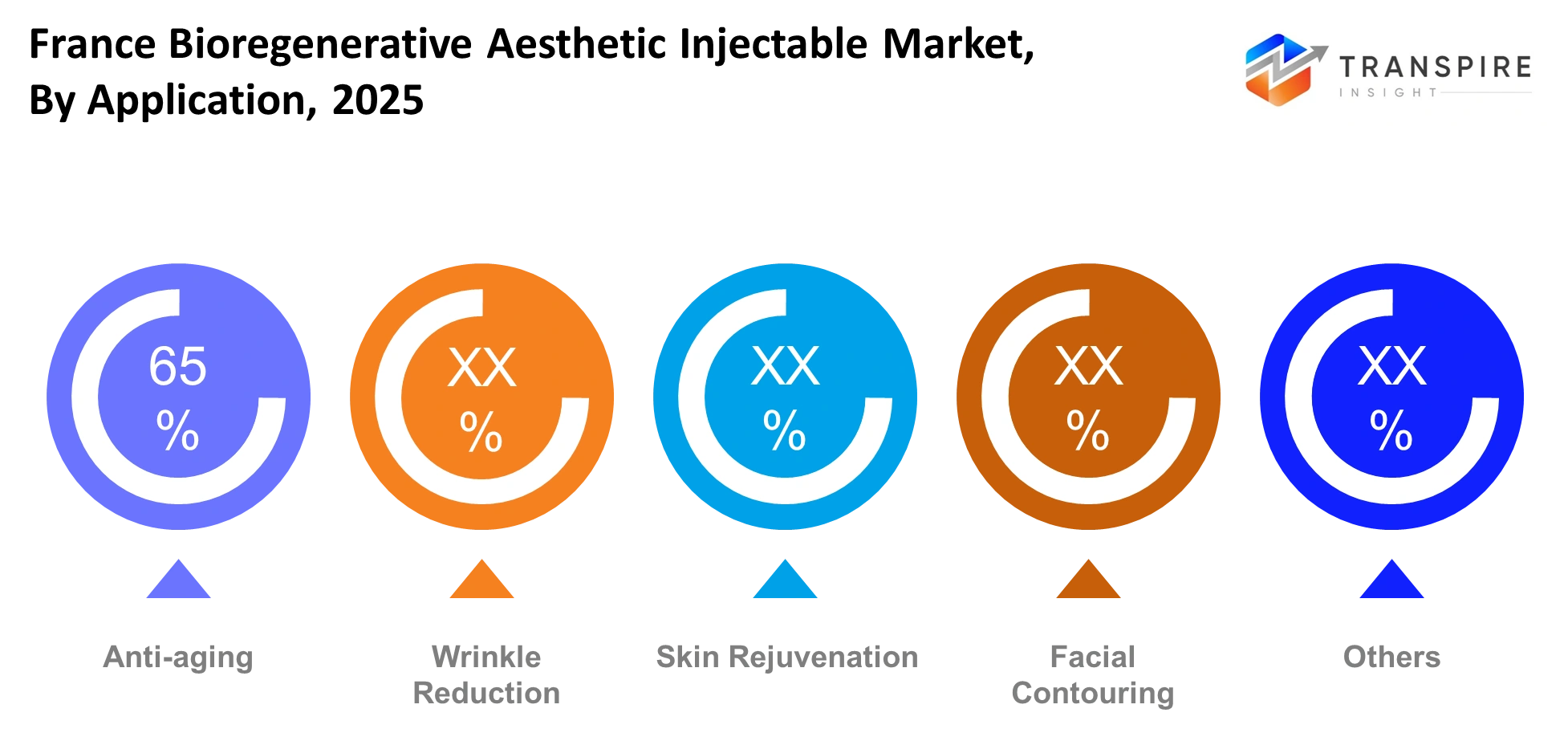
By Application
- The France Bioregenerative Aesthetic Injectable Market currently uses anti-aging treatments as its primary application because the aging population in cities continuously seeks facial rejuvenation and they frequently visit medical clinics. Dermatology centers prioritize these procedures because they deliver consistent revenue cycles and repeat treatment schedules. Urban France people maintain strong cultural values which make physical appearance more important than other aspects of life.
- The market for wrinkle reduction and skin rejuvenation treatments experiences rapid growth because younger patients start their preventive aesthetic care habits earlier than their forebears. The applications receive advantages from better injectable safety systems and reduced recovery durations which make first-time users more likely to try the products. The popularity of facial contouring has grown because premium clinics use combination therapies to achieve both structural improvements and facial symmetry restoration.
To learn more about this report, Download Free Sample Report
By End-User
- The France Bioregenerative Aesthetic Injectable Market shows its largest market share through dermatology clinics which operate as the main distribution channel for injectable aesthetic treatments. The clinics maintain constant patient flow because they possess particular expertise and patients trust them and they have access to advanced regenerative medical treatments. The organizations achieve greater power because they use physician-driven treatment methods to build their patient referral systems.
- The rising demand for less invasive outpatient cosmetic procedures has led to rapid growth in cosmetic and aesthetic clinics. These centers attract younger and mid-income patient groups through their combination of flexible pricing options and quick appointment scheduling. The hospital system maintains its market share through its focus on medically necessary reconstructive and post-trauma procedures while keeping elective cosmetic services to a minimum.
By Distribution
- The France Bioregenerative Aesthetic Injectable Market mainly operates through direct clinic-based distribution because medical facilities need to conduct most procedures using certified doctors. The system establishes regulations through its controlled procedures for handling active biological injectable products which need direct doctor supervision to administer treatments. The strong partnerships between manufacturers and clinics help to maintain the channel's position as the leading distribution method.
- The online channels face restrictions on direct injectable sales because the European Union enforces strict regulations that control prescription-based aesthetic products. The digital platforms now help patients to book consultations which enables medical staff to educate patients through clinical interactions that drive product demand. The distribution networks which include authorized distributors and specialty medical suppliers ensure that smaller cities and new clinics maintain their supply chain operations.
What are the Key Use Cases Driving the France Bioregenerative Aesthetic Injectable Market?
The France Bioregenerative Aesthetic Injectable Market shows facial rejuvenation and skin quality restoration work as its primary application because clinics administer collagen-stimulating injectables and biostimulatory fillers to diminish wrinkles and restore facial volume and enhance skin elasticity. The application generates maximum demand because patients prefer to achieve natural-looking results that last longer than temporary volumizing treatments.
The post-acne scar correction application together with early skin aging treatment solution has gained wide acceptance across dermatology clinics and medical spas. The use cases are expanding because younger patients now prefer preventive aesthetic treatments instead of doing corrective procedures which results in more treatment visits and higher revenue for clinics.
The new use cases involve regenerative skin repair through exosome-based injectables which help with wound healing and post-procedure recovery. The premium clinics test combination therapies which use injectables together with laser and microneedling treatments to demonstrate strong potential for future adoption throughout advanced aesthetic centers across France.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 101.38 Million |
|
Market size value in 2026 |
USD 111.62 Million |
|
Revenue forecast in 2033 |
USD 218.95 Million |
|
Growth rate |
CAGR of 10.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Allergan, Galderma, Merz Pharma, Sinclair Pharma, Teoxane, Ipsen, Revance, Hugel, Medytox, Bloomage Biotech, LG Chem, Caregen, Prollenium, BioPlus, IBSA. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Dermal Fillers, Collagen Stimulators, PRP Injectables, Others); By Application (Anti-aging, Wrinkle Reduction, Skin Rejuvenation, Facial Contouring, Others); By End-User (Dermatology Clinics, Hospitals, Cosmetic Centers, Aesthetic Clinics, Others); By Distribution (Clinics, Online Channels, Others). |
Which Regions are Driving the France Bioregenerative Aesthetic Injectable Market Growth?
The France Bioregenerative Aesthetic Injectable Market designates Île-de-France as its main market area because the region contains numerous top-tier dermatology facilities and modern aesthetic medical hospitals. The region benefits from strong purchasing power and early adoption of innovative regenerative injectables, particularly in Paris-based cosmetic hubs. A well-established ecosystem of medical universities, biotech startups, and cosmetic research institutes supports continuous clinical innovation. High patient awareness of anti-aging treatments and frequent exposure to luxury aesthetic services further reinforce its dominant position.
The wellness tourism sector provides a stable foundation for Provence-Alpes-Côte d’Azur which maintains constant patient demand from both domestic and international visitors. The growth in this region exists because existing private clinic networks and seasonal aesthetic tourism create demand at medical facilities which operate throughout the year. The region maintains steady revenue contribution because clinics consistently invest in mid-to-high-end injectable treatments targeting skin rejuvenation and preventive aging. The system functions effectively because both the developed healthcare system and the established reputation for cosmetic dermatology services sustain its operations.
Occitanie has reached its fastest growth stage because biotech funding has increased in Toulouse and Montpellier while medical aesthetics facilities continue to grow. The regional innovation hubs have developed new conditions which support clinical testing of next-generation regenerative injectables that local clinics will begin using in their practices. Life sciences startups now receive more funding while dermatology practitioners gain better access to training which has enhanced market growth efforts. The company shows strong growth potential through its international market expansion efforts.
Who are the Key Players in the France Bioregenerative Aesthetic Injectable Market and How Do They Compete?
The France Bioregenerative Aesthetic Injectable Market exhibits moderate market concentration at its uppermost level but maintains intense rivalry among mid-level restorative product manufacturers. Established dermatology-focused pharmaceutical firms compete with biotech-driven entrants that are accelerating innovation in exosome therapies, polynucleotides, and collagen-stimulating injectables. The main factors that drive competition between companies operate through technology differences and clinical effectiveness as premium aesthetic clinics choose to evaluate safety standards and treatment duration results instead of product costs. The regulatory requirements together with EU certification standards function as essential competitive barriers which determine how products enter markets and establish their market presence.
Galderma strengthens its position through advanced regenerative dermatology pipelines, particularly collagen-stimulating injectables designed for long-term skin quality improvement. The company operates a comprehensive dermatology clinic network throughout France which enables fast product deployment and medical professional training initiatives that help it attain better results during medical operations.
Teoxane differentiates through its expertise in hyaluronic acid-based injectable technologies developed in Switzerland and widely adopted in French aesthetic clinics. The company concentrates on innovations that clinicians create while developing formulations which enable natural tissue integration and establishing distribution agreements with private dermatology clinics.
Merz Pharma competes through biostimulatory injectable platforms that emphasize regenerative skin repair rather than volumization. The company establishes its market presence by forming strategic partnerships with aesthetic networks throughout Europe which enable it to enter medical spa platforms.Sinclair Pharma targets niche regenerative aesthetics with portfolio diversification into collagen induction therapies.
Company List
- Allergan
- Galderma
- Merz Pharma
- Sinclair Pharma
- Teoxane
- Ipsen
- Revance
- Hugel
- Medytox
- Bloomage Biotech
- LG Chem
- Caregen
- Prollenium
- BioPlus
- IBSA
Recent Development News
In November 2025, Galderma presented new clinical data on its injectable aesthetics portfolio, including Sculptra and Restylane, at ASDS 2025. The findings reinforced long-term regenerative benefits and expanded use cases for facial contouring and skin quality improvement in European dermatology clinics.
https://www.galderma.com
In January 2026, Galderma showcased expanded regenerative injectable applications at IMCAS 2026 in Paris. The company highlighted Sculptra body indications and advanced Restylane-based skin rejuvenation treatments, strengthening adoption of regenerative aesthetics across French clinics.
https://www.galderma.com
What Strategic Insights Define the Future of the France Bioregenerative Aesthetic Injectable Market?
The France Bioregenerative Aesthetic Injectable Market is now developing a new aesthetic treatment system which will start using biologics to provide aesthetic solutions through regenerative cell-signaling injectables instead of traditional volumizing methods. Aesthetic medicine is evolving into tissue regeneration treatments because dermatology and biotechnology and precision medicine have started to work together. The upcoming 5 to 7 years will show that value creation depends on two factors, which include clinically proven duration of results and doctor-led product customization, instead of product quantity.
The establishment of stricter EU biologics regulations represents a concealed danger because it will create obstacles for exosome and peptide-based injectable products to obtain regulatory approval, which will extend their time to market introduction while allowing current market leaders to strengthen their dominance.
The growth of biotech-dermatology innovation clusters in Occitanie and Île-de-France creates an opportunity, which supports human trials of regenerative injectable platforms that biomedical startups develop with strong academic funding.
These hubs will function as primary distribution centers which supply advanced treatment solutions that use next-generation therapies.
Companies should concentrate on establishing their first clinical ties with research hospitals in France because this strategy will help them obtain trial access and expedite their efforts to achieve regulatory compliance before EU harmonization measures make late-stage market entry more difficult.
France Bioregenerative Aesthetic Injectable Market Report Segmentation
By Type
- Dermal Fillers
- Collagen Stimulators
- PRP Injectables
- Others
By Application
- Anti-aging
- Wrinkle Reduction
- Skin Rejuvenation
- Facial Contouring
- Others
By End-User
- Dermatology Clinics
- Hospitals
- Cosmetic Centers
- Aesthetic Clinics
- Others
By Distribution
- Clinics
- Online Channels
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure is USD 218.95 Million.
Key segments for the France Bioregenerative Aesthetic Injectable Market are By Type (Dermal Fillers, Collagen Stimulators, PRP Injectables, Others); By Application (Anti-aging, Wrinkle Reduction, Skin Rejuvenation, Facial Contouring, Others); By End-User (Dermatology Clinics, Hospitals, Cosmetic Centers, Aesthetic Clinics, Others); By Distribution (Clinics, Online Channels, Others).
Major France Bioregenerative Aesthetic Injectable Market players are Allergan, Galderma, Merz Pharma, Sinclair Pharma, Teoxane, Ipsen, Revance, Hugel, Medytox, Bloomage Biotech, LG Chem, Caregen, Prollenium, BioPlus, IBSA.
The France Bioregenerative Aesthetic Injectable Market size is USD 101.38 Million in 2025.
The France Bioregenerative Aesthetic Injectable Market's CAGR is 10.10% from 2026 to 2033.
- Allergan
- Galderma
- Merz Pharma
- Sinclair Pharma
- Teoxane
- Ipsen
- Revance
- Hugel
- Medytox
- Bloomage Biotech
- LG Chem
- Caregen
- Prollenium
- BioPlus
- IBSA
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


