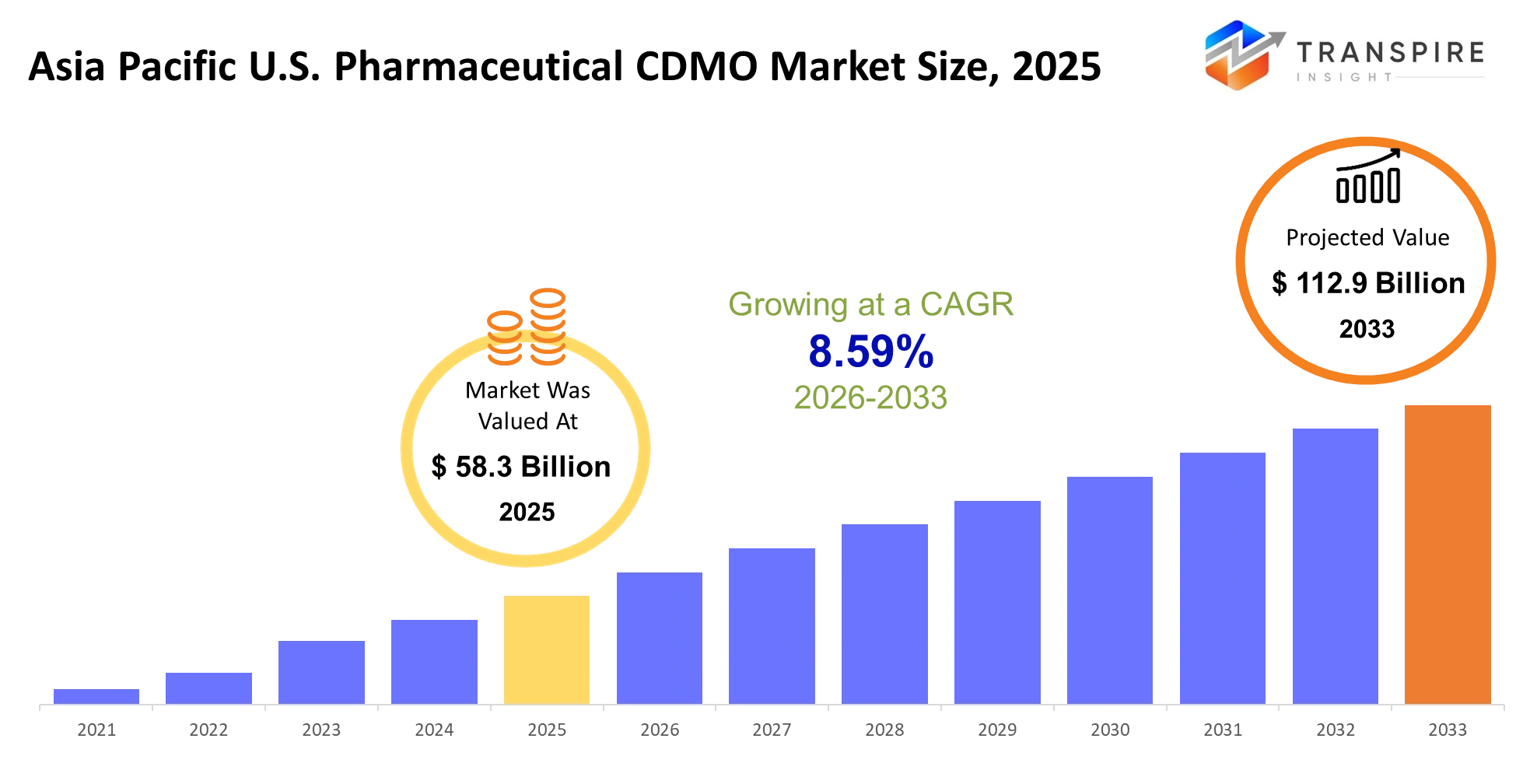
Asia Pacific U.S. Pharmaceutical CDMO Market Size & Forecast:
- Asia Pacific U.S. Pharmaceutical CDMO Market Size 2025: USD 58.3 Billion
- Asia Pacific U.S. Pharmaceutical CDMO Market Size 2033: USD 112.9 Billion
- Asia Pacific U.S. Pharmaceutical CDMO Market CAGR: 8.59%
- Asia Pacific U.S. Pharmaceutical CDMO Market Segments: By Type (Biologics CDMO, Small Molecule CDMO, Cell & Gene Therapy CDMO, Others); By Application (Drug Development, Manufacturing, Clinical Trials, Commercial Production, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Startups, Others); By Service (Process Development, Analytical Testing, Packaging, Regulatory Support, Others).
To learn more about this report, Download Free Sample Report
Asia Pacific U.S. Pharmaceutical CDMO Market Summary
The Asia Pacific U.S. Pharmaceutical CDMO Market was valued at USD 58.3 Billion in 2025. It is forecast to reach USD 112.9 Billion by 2033. That is a CAGR of 8.59% over the period.
The market for U.S. Pharmaceutical CDMO services in the Asia Pacific region enables drug developers to transform laboratory developed products into commercial manufacturing operations. These organizations perform complex operations which include producing active pharmaceutical ingredients and developing drug formulations and manufacturing clinical trial batches and providing large-scale commercial products to pharmaceutical companies.
The market has experienced a transformation within the last five years which evolved from basic outsourcing services into specialized partnerships that focus on biologics and cell and gene therapies and advanced sterile manufacturing capabilities. The transition occurred because post-pandemic supply chain disruptions and the implementation of stricter U.S. sourcing requirements forced pharmaceutical companies to establish production sites throughout the Asia Pacific region while they remained compliant with regulations. Contract manufacturers from Singapore and South Korea and India increased their capacity to operate high-compliance production sites which include digital quality control systems. The adoption of flexible manufacturing systems which reduce operational risks has become a priority for pharmaceutical companies because it leads to increased multi-year outsourcing agreements and improved revenue projections for regional CDMO providers.
Key Market Insights
- The Asia Pacific U.S. Pharmaceutical CDMO Market experiences growth because pharmaceutical companies outsource biologics production which enables faster product delivery and decreases their capital investment needs.
- The post-2020 supply chain disruptions required U.S. pharmaceutical companies to develop compliance strategies which resulted in a shift toward production facilities located in the Asia Pacific region.
- The Asia Pacific U.S. Pharmaceutical CDMO Market experiences a transformation because advanced therapies which include cell and gene therapy manufacturing create a demand for high-value specialized production.
- The APAC hubs which link to North America control approximately 38% market share because they provide U.S. companies with regulatory-compliant manufacturing systems and outsourcing options.
- Southeast Asia will experience the fastest growth between 2024 and 2030 because of its cost-effective solutions and growing skilled workforce and ongoing infrastructure development.
- Drug substance manufacturing leads with approximately 40% share because both biologics and active pharmaceutical ingredient outsourcing create high demand.
- The Asia Pacific U.S. Pharmaceutical CDMO Market grows through 2030 because cell and gene therapy services become its fastest expanding sector.
- Oncology applications dominate with nearly 32% share because global biologics pipelines and precision medicine adoption continue to increase.
- Rare disease therapeutics become the fastest expanding application area because regulatory approvals and orphan drug incentives have increased.
- The major pharmaceutical companies control about 55% market share because they use CDMO partnerships to improve their worldwide production capacity.
What are the Key Drivers, Restraints, and Opportunities in the Asia Pacific U.S. Pharmaceutical CDMO Market?
The Asia Pacific U.S. Pharmaceutical CDMO Market growth is driven by pharmaceutical companies now outsourcing their complex biologics and advanced therapy drug development and manufacturing needs. In-house production became more expensive because U.S. regulations established tighter quality control and supply chain tracking standards. Pharmaceutical companies turn to Asia Pacific CDMOs because these facilities possess FDA-approved operations at their sites in India and Singapore and South Korea. The number of long-term contract manufacturing agreements increased which helped service providers achieve better revenue forecasting while their production capacity at advanced manufacturing sites increased.
The market encounters a structural barrier because different jurisdictions impose strict regulatory requirements that must be harmonized. Most Asia Pacific facilities undergo U.S.-FDA inspections but continuous compliance with changing regulations needs ongoing financial resources for quality system development and documentation system building and employee education. The system creates operational challenges for mid-sized CDMOs because their lack of international regulatory knowledge leads to increased operational costs and work delays. The need for new capacity to generate revenue extends existing expansion timelines.
The rapid growth of cell and gene therapy production presents a major business opportunity. The biomanufacturing sector is experiencing increased investment for modular and flexible production facilities, which governments in Singapore and Japan are supporting through their advanced therapy ecosystem development initiatives. Lonza and other companies are developing their specialized production facilities, which will help the Asia Pacific U.S. Pharmaceutical CDMO Market transition to high-value precision manufacturing services during its upcoming growth period.
What Has the Impact of Artificial Intelligence Been on the Asia Pacific U.S. Pharmaceutical CDMO Market?
The Asia Pacific U.S. Pharmaceutical CDMO Market experiences operational changes because artificial intelligence and advanced digital technologies create intelligent systems which manufacturers use to modify their production processes and quality control methods. AI-powered automation systems currently monitor cleanroom environments and control operational systems and track equipment calibration in various industries. CDMO facilities deploy machine vision and sensor analytics systems to maintain constant monitoring of sterile production areas which decreases the need for manual inspections and enhances readiness for regulatory compliance at FDA-approved facilities.
Biotechnology researchers increasingly adopt predictive models to enhance bioreactor performance and maintain chromatography systems and forecast utility loads. Manufacturers use machine learning algorithms to study historical batch data which enables them to predict when their equipment will start to fail and plan maintenance activities accordingly. The company achieved higher production uptime through this solution which minimized unplanned downtime and reduced operational expenses by optimizing energy usage and decreasing batch rejection rates.
Digital twin platforms together with AI-powered manufacturing execution systems enable organizations to achieve consistent operational performance while they reduce the time needed to transition from clinical testing to commercial manufacturing. Asia Pacific CDMO facilities show measurable productivity improvements which occur mainly through better yield consistency and improved compliance documentation precision.
The main obstacle facing pharmaceutical companies today lies in the expensive and complicated process of implementing artificial intelligence systems throughout their existing production facilities. The majority of manufacturing sites maintain separate data systems which hinder their ability to create accurate models and make instantaneous decisions. The digital transformation process in mid-sized CDMO operators faces challenges because there exists a divide between their advanced digital systems and actual business operations.
Key Market Trends
- CDMO outsourcing shifted from small-molecule production pre-2020 to biologics and cell therapy dominance after 2022, increasing high-value contract manufacturing demand.
- U.S. pharmaceutical firms reduced single-source dependency by 35% since 2021, which led to their increased manufacturing operations at FDA-compliant facilities throughout the Asia Pacific region.
- Continuous manufacturing adoption increased post-2023 because it replaced batch-based production methods which enabled better waste management and shorter biologics production times.
- WuXi AppTec developed its global biologics capacity from 2022 to 2025 because customers wanted complete CDMO services that covered all aspects of production.
- Digital quality management systems replaced manual compliance tracking in major facilities after 2023, which increased audit readiness while reducing delays that stemmed from regulatory requirements.
- Vaccine manufacturing capacity expanded significantly during 2021–2024, but production operations gradually transitioned toward mRNA and personalized therapeutics production platforms.
- Samsung Biologics expanded its bioreactor capacity by more than 50% in 2022 to meet international pharmaceutical contract obligations.
- Supply chain disruption from COVID-19 resulted in companies using multiple manufacturing sites to decrease their dependence on manufacturing operations located in one country.
- Process optimization through AI technology became more common after 2024 which resulted in improved yield stability and decreased production failures for advanced therapy manufacturing systems.
- Lonza together with other global contract development and manufacturing organizations expanded their partnerships in Asia-Pacific countries which indicates their strategic shift toward complex pharmaceutical manufacturing systems that match U.S. operational standards.
Asia Pacific U.S. Pharmaceutical CDMO Market Segmentation
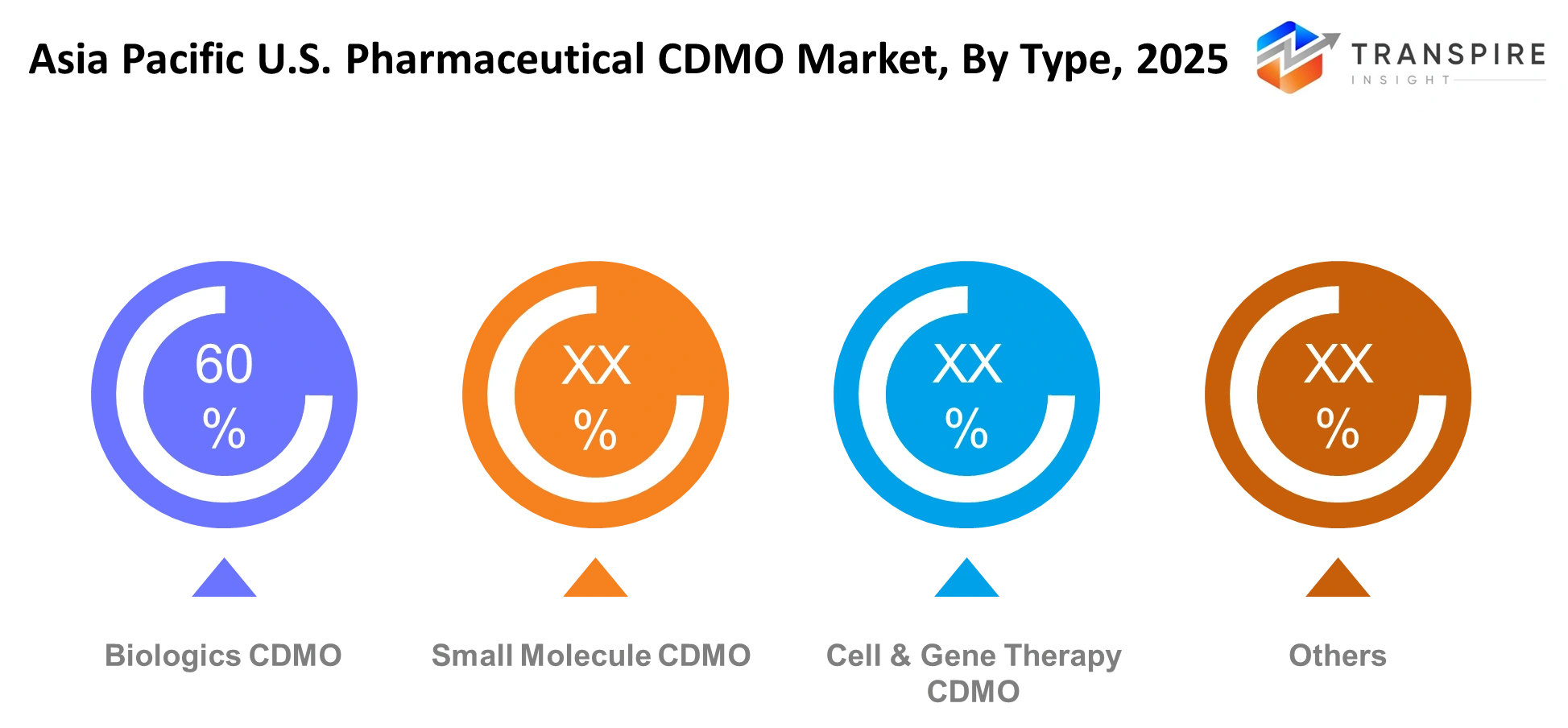
By Type :
The demand for biologics CDMO services will experience growth because of rising interest in antibody-based and protein-based therapies. The demand for small molecule CDMO services will not change because these services are commonly used in standard treatment procedures. The demand for cell and gene therapy CDMO services will increase because of the need for better therapeutic solutions. The remaining services will enable production through both specialized and combined production methods.
Biologics production requires facilities with advanced technology and systems that provide strict operational control. The small molecule services maintain their business focus on providing affordable services which can expand according to client needs. Cell and gene therapy processes require facilities that operate with specialized methods and maintain their environmental conditions. The other category will contain mixed manufacturing platforms which enable flexible production operations throughout different stages of therapeutic development.
To learn more about this report, Download Free Sample Report
By Application :
Companies will continue to use drug development as their main research area because they delegate their initial research activities and formulation tasks to outside partners. The manufacturing sector will experience strong demand because of the requirements for large-scale production operations. Clinical trials will increase because of the growing number of worldwide research studies and commercial production will provide the necessary supply for authorized pharmaceutical products.
The practice of drug development outsourcing enables organizations to decrease their internal responsibilities while accelerating their product development activities. The manufacturing services of the company will prioritize operational productivity and the delivery of high-quality products at all times. The organization will provide clinical trial support to expedite patient recruitment and shorten testing durations. The organization will use commercial production to maintain a constant supply after product approvals while using other resources to meet specific project requirements.
By End-User :
The pharmaceutical industry will continue to use most resources because of their product development requirements and their worldwide distribution demands. Biotech companies will depend on external partners to develop their high-level medical treatments. Research institutes will use CDMO services for study support, while CROs and startups will depend on flexible and cost-efficient solutions.
Pharmaceutical companies will use their internal capacity to increase production while decreasing their manufacturing requirements. Biotech companies will require precise measurements together with specialized technical assistance. Research institutes will use services for experimental work. CROs will manage outsourced clinical activities, and startups will depend on CDMO partners for early-stage development and production support.
By Service :
The process development will provide support for both the initial formulation development and the subsequent scale-up process. The analytical testing process will verify that products maintain safety standards and quality requirements and legal compliance from their initial development through their entire lifecycle. The packaging services will protect drugs through secure handling operations while the regulatory support system will assist with meeting market-specific approval requirements and document standards.
The process development activities will create better productivity improvement from laboratory operations through to full-scale manufacturing. Analytical testing will confirm stability and quality standards. The packaging system will protect medicines during transportation and storage. The support system for regulatory matters will assist companies in their approval process while ensuring compliance with regulations which will help them achieve market availability across different areas.
What are the Key Use Cases Driving the Asia Pacific U.S. Pharmaceutical CDMO Market?
The primary application of the Asia Pacific U.S. Pharmaceutical CDMO Market centers on biologics and active pharmaceutical ingredient production which serves the needs of large pharmaceutical companies. The market demand arises from three factors which include the high complexity of product development and the requirement for exact FDA compliance and the demand for production processes which can expand without needing extensive in-house facility expenses.
Biotech companies which focus on oncology and rare disease research markets their clinical trial batch manufacturing and sterile injectable production services for their experimental pipelines. The demand for these use cases has increased because smaller companies now depend on CDMOs to assist them with their development process and their need to comply with regulatory submission deadlines.
The new applications focus on developing cell and gene therapy products and creating personalized medical solutions. These solutions exist in their initial adoption phase but are becoming more popular because regulatory systems develop and dedicated production facilities establish themselves throughout Singapore and South Korea.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 58.3 Billion |
|
Market size value in 2026 |
USD 63.4 Billion |
|
Revenue forecast in 2033 |
USD 112.9 Billion |
|
Growth rate |
CAGR of 8.59% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Asia Pacific (China, India, Japan, South Korea, Australia, Rest of Asia Pacific) |
|
Key company profiled |
Lonza, Catalent, Thermo Fisher, WuXi AppTec, Samsung Biologics, Recipharm, Siegfried, Cambrex, Evotec, Charles River, Boehringer Ingelheim, Fujifilm Diosynth, Piramal Pharma, Alcami, Jubilant Biosys. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Biologics CDMO, Small Molecule CDMO, Cell & Gene Therapy CDMO, Others); By Application (Drug Development, Manufacturing, Clinical Trials, Commercial Production, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Startups, Others); By Service (Process Development, Analytical Testing, Packaging, Regulatory Support, Others). |
Which Regions are Driving the Asia Pacific U.S. Pharmaceutical CDMO Market Growth?
North America leads the Asia Pacific U.S. Pharmaceutical CDMO Market because it has strict regulatory requirements and needs industrial facilities from outside sources. The U.S. FDA enforcement standards require pharmaceutical companies to work with Asia-based CDMOs which operate inspection-ready facilities that meet compliance standards. The existing situation exists because companies have active outsourcing agreements and they possess advanced biologics development programs and they maintain strong protection for their intellectual property assets. The existing supply chain connections between U.S. pharmaceutical companies and Asia Pacific manufacturing centers enable facilities to operate at their full production capacity. The region experiences stable demand because high-value drug development programs remain active throughout the area.
Europe functions as a stable secondary market for pharmaceutical outsourcing while North American markets prefer aggressive outsourcing strategies. Regional pharmaceutical companies select multiple CDMO partners through risk-balanced sourcing methods instead of relying on a single supplier. The combination of strong healthcare funding systems and consistent demand for biologics drives the market for outsourcing services. The EMA and other regulatory bodies implement regular compliance updates which promote safe but ongoing usage of CDMO services from Asia. The system establishes stable income streams which maintain steady demand for contract manufacturing services.
Southeast Asia has become the fastest-growing area because of recent massive investments which have developed pharmaceutical manufacturing facilities throughout Singapore, Malaysia, and Vietnam. The implementation of incentives by the government after 2023 has led to faster development of compliant facilities and increased foreign direct investment. The development of cold chain logistics together with the modernization of ports has improved the export capacity for biologics and sterile injectables. The region provides expansion opportunities to investors who enter between 2026 and 2033 because of its cost advantages and its regulatory framework which is developing to match US standards.
Who are the Key Players in the Asia Pacific U.S. Pharmaceutical CDMO Market and How Do They Compete?
The Asia Pacific U.S. Pharmaceutical CDMO Market shows moderate consolidation at its top level while mid-tier providers display fragmented competition. The leading companies in the market compete through their ability to produce biologics and their adherence to regulatory standards and their provision of complete service packages. Pharmaceutical companies use digital manufacturing and sterile production technology to gain competitive advantages because they value reliability more than saving money through outsourcing.
The company Samsung Biologics aims to expand its South Korean bioreactor capacity through large-scale operations which will enable it to produce high volumes of biologics and establish long-term production contracts. WuXi AppTec provides U.S. biotech companies with complete development services because it combines its discovery capabilities with its commercial production functions. Thermo Fisher Scientific expands its business through acquisitions while developing new biologics and analytical services that provide customers with complete development-to-manufacturing solutions which reduce project completion times.
Catalent uses its expertise in advanced drug delivery systems and clinical-scale manufacturing to strengthen its capability in developing complex formulations. Dr. Reddy's Laboratories develops cost-effective CDMO operations in India which include regulatory improvements needed to support U.S.-based pharmaceutical customers. Lonza establishes its position in next-generation therapeutic development through its global collaborations and capacity development that enable it to manufacture high-value biologics and cell therapies.
Company List
- Lonza
- Catalent
- Thermo Fisher
- WuXi AppTec
- Samsung Biologics
- Recipharm
- Siegfried
- Cambrex
- Evotec
- Charles River
- Boehringer Ingelheim
- Fujifilm Diosynth
- Piramal Pharma
- Alcami
- Jubilant Biosys
Recent Development News
In April 2026, Andelyn Biosciences and ENCell Co., Ltd. established a partnership to create a gene therapy manufacturing network which will connect the United States with the Asia Pacific region. The partnership establishes a dual-hemisphere contract development and manufacturing organization which links manufacturing sites in the United States with South Korea to support worldwide gene therapy development through efficient GMP production and clinical supply operations while simplifying both regulatory and logistical processes.
Source: https://pharmasource.global/
In March 2026, Merck KGaA signed a collaboration with Cyto-Facto Inc. for cell and gene therapy manufacturing support in the Asia-Pacific region. The partnership focuses on lentiviral vector production, process scale-up, and technical training across Japan and India, which helps regional CDMO facilities to develop their advanced therapy capabilities.
Source: https://pharma.economictimes.indiatimes.com/
What Strategic Insights Define the Future of the Asia Pacific U.S. Pharmaceutical CDMO Market?
The Asia Pacific U.S. Pharmaceutical CDMO Market is moving toward manufacturing systems that require advanced manufacturing capabilities, which produce innovative products through complete development and production systems. The market development occurs because companies need to meet rising biologics demand, while the U.S. government implements stricter supply chain regulations, and pharmaceutical companies experience continuous financial losses from their internal manufacturing operations. Businesses will achieve competitive advantages through their technical expertise and ability to meet regulatory requirements during the upcoming five to seven years.
A less visible risk lies in growing concentration among a small group of large CDMOs controlling high-end biologics capacity. U.S. pharmaceutical companies face dependency risks because global supply chains will experience sudden disruptions when these hubs encounter capacity bottlenecks or regulatory non-compliance issues. The concentration of CDMOs in the market limits smaller biotech clients from negotiating better terms.
The key emerging opportunity shows potential through the expansion of decentralized modular manufacturing facilities, which produce personalized therapies, especially in Singapore and South Korea, because these countries are implementing modern regulatory frameworks. The new production system will enable flexible facilities to produce late-stage clinical and commercial products. Market participants need to invest in modular facilities that combine digital technology and upgrade FDA compliance systems to obtain outsourcing contracts and meet future requirements for biologics products.
Asia Pacific U.S. Pharmaceutical CDMO Market Report Segmentation
By Type
- Biologics CDMO
- Small Molecule CDMO
- Cell & Gene Therapy CDMO
By Application
- Drug Development
- Manufacturing
- Clinical Trials
- Commercial Production
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- CROs
- Startups
By Service
- Process Development
- Analytical Testing
- Packaging
- Regulatory Support
Frequently Asked Questions
Find quick answers to common questions.
The Asia Pacific U.S. Pharmaceutical CDMO Market size is USD 112.9 Billion in 2033.
Key segments for the Asia Pacific U.S. Pharmaceutical CDMO Market are By Type (Biologics CDMO, Small Molecule CDMO, Cell & Gene Therapy CDMO, Others); By Application (Drug Development, Manufacturing, Clinical Trials, Commercial Production, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Startups, Others); By Service (Process Development, Analytical Testing, Packaging, Regulatory Support, Others).
Major Asia Pacific U.S. Pharmaceutical CDMO Market players are Lonza, Catalent, Thermo Fisher, WuXi AppTec, Samsung Biologics, Recipharm, Siegfried, Cambrex, Evotec, Charles River, Boehringer Ingelheim, Fujifilm Diosynth, Piramal Pharma, Alcami, Jubilant Biosys.
The Asia Pacific U.S. Pharmaceutical CDMO Market size is USD 58.3 Billion in 2025.
The Asia Pacific U.S. Pharmaceutical CDMO Market CAGR is 8.59% from 2026 to 2033.
- Lonza
- Catalent
- Thermo Fisher
- WuXi AppTec
- Samsung Biologics
- Recipharm
- Siegfried
- Cambrex
- Evotec
- Charles River
- Boehringer Ingelheim
- Fujifilm Diosynth
- Piramal Pharma
- Alcami
- Jubilant Biosys
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


