Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Size & Forecast:
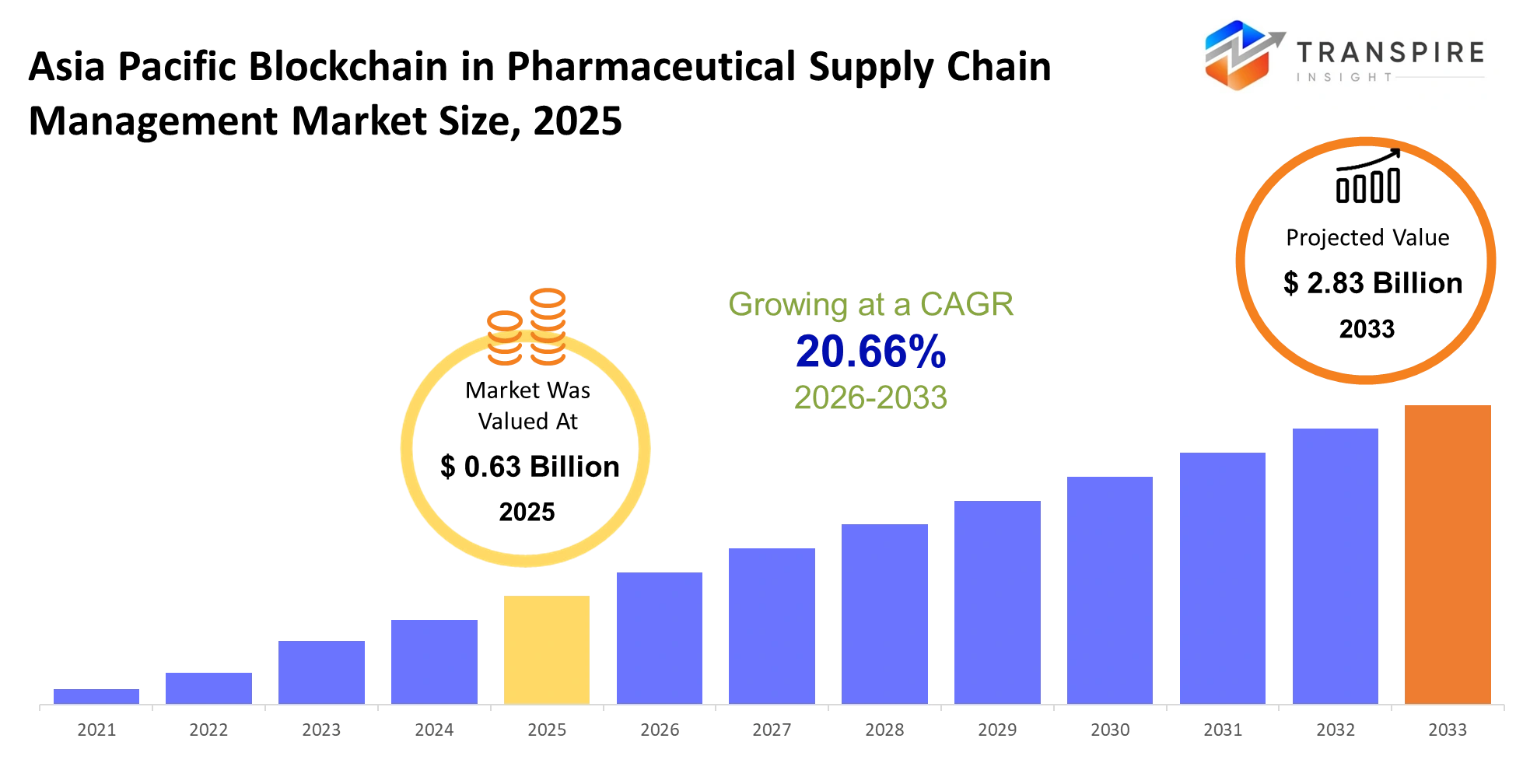
- Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Size 2025: USD 0.63 Billion
- Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Size 2033: USD 2.83 Billion
- Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market CAGR: 20.66%
- Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Segments: By Type (Private Blockchain, Public Blockchain, Consortium Blockchain, Others); By Application (Drug Traceability, Supply Chain Transparency, Anti-counterfeiting, Inventory Management, Others); By End-User (Pharma Companies, Distributors, Hospitals, Government, Logistics Providers, Others); By Deployment (Cloud, On-premise, Hybrid, Others).
To learn more about this report, Download Free Sample Report
Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Summary
The Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market was valued at USD 0.63 Billion in 2025. It is forecast to reach USD 2.83 Billion by 2033. That is a CAGR of 20.66% over the period.
The Asia Pacific blockchain in pharmaceutical supply chain management market addresses one of the industry’s most persistent operational challenges: ensuring that every drug shipment can be tracked, verified, and authenticated as it moves from manufacturers to distributors, hospitals, and pharmacies. The platforms enable pharmaceutical companies to use their tools for preventing counterfeit medicines while they enhance their ability to trace product batches and conduct their required process for product recalls and compliance audits throughout their various operational areas.
The market has moved from its original testing phase into complete system integration which operates through cloud-based ERP together with serialization technology during the past three to five years. The structural transition occurred because countries like China and India and Japan implemented stricter drug traceability requirements which made digital verification systems mandatory. The COVID-19 pandemic supply disruptions showed companies their inventory problems making them more visible and their supplier problems harder to control. Pharmaceutical manufacturers use blockchain technology because it improves their business operations through better system security and their ability to manage essential information system across various locations which leads to increased platform spending and bigger implementation projects and ongoing software revenue streams throughout the entire area.
Key Market Insights
- China controls the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market which will reach 34% market share by 2026 because of its dedicated efforts towards digitizing its pharmaceutical industry.
- The pharmaceutical export compliance modernization will drive India to become the most rapidly expanding regional market which will continue to grow until 2026 through 2032.
- Japan maintains a major market position because its healthcare IT systems are highly developed and its pharmaceutical distribution networks use blockchain technology in pilot projects.
- Pharmaceutical companies focus on real-time transaction verification which allows blockchain platform solutions to secure approximately 46% of market share in 2026.
- The cloud-based blockchain deployment market will grow at the fastest rate until 2032 because of its affordable implementation and ability to expand.
- The primary application of drug serialization and product authentication functions as the main driver for 41 percent of Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market revenue which will reach its peak in 2026.
- The fastest expanding application market exists in cold chain monitoring because its growth is driven by the increasing distribution of biologics throughout Asia Pacific pharmaceutical networks.
- The market for recall management solutions is experiencing growth because regulators require organizations to establish complete supply chain tracking systems while responding to product recalls at greater speed.
- Pharmaceutical manufacturers control the market with their 49 percent share because they use blockchain technology to improve their compliance efforts and decrease their risk of counterfeit products.
- Healthcare systems are increasingly adopting hospital procurement networks because they need methods to verify medicine sourcing through transparent processes.
What are the Key Drivers, Restraints, and Opportunities in the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market?
The pharmaceutical supply chain management market for Asia Pacific blockchain technology experiences its highest growth rate because Asian countries which include China India Japan and South Korea have established stricter regulations for drug traceability that pharmaceutical manufacturers must follow. Governments have strengthened serialization mandates and digital reporting requirements after repeated incidents involving counterfeit medicines and compliance failures in export markets. Pharmaceutical manufacturers must now dedicate their resources to develop blockchain-based verification systems which produce permanent record systems and enable real-time product tracking. The implementation process progresses from pilot stage to enterprise-wide adoption while vendors generate higher software license revenue and ongoing managed service income.
The most significant barrier remains the fragmented digital infrastructure across pharmaceutical supply chains in emerging Asia Pacific economies. The regional manufacturing and distribution networks together with third-party logistics providers continue to face high cost challenges regarding blockchain technology integration because their current systems use outdated ERP software which does not support modern standards. The process of upgrading entire digital ecosystems creates structural constraints because it requires organizations to invest significant funds while training their workforce and establishing new industry standards. The result is slower deployment cycles, postponed procurement decisions, and lost revenue opportunities for technology providers.
The company can succeed through its expansion plan which connects blockchain technology with artificial intelligence prediction systems for monitoring vaccine and temperature-sensitive biological products. The digital health infrastructure investments of Singapore and Australia create opportunities for intelligent supply chain platforms to operate in their respective nations. Pharmaceutical companies can achieve three benefits through the combined use of blockchain traceability and automated anomaly detection, which includes preventing product spoilage and improving inventory control and accessing high-value service contracts for specialty drug markets.
What Has the Impact of Artificial Intelligence Been on the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market?
The development of pharmaceutical blockchain supply chain platforms through artificial intelligence benefits the Asia Pacific region by using automated systems that validate data and detect anomalies while monitoring compliance across intricate drug distribution systems. The AI-based control systems, which operate with blockchain technology, automatically verify shipment records and serialization data while they identify inconsistencies to reduce the necessity of manual audits by 25 to 35 percent throughout major pharmaceutical distribution networks.
The automation functions enable manufacturers and distributors to achieve precise inventory records because they streamline the process of gathering necessary information for regulatory compliance. Machine learning is also enhancing predictive capabilities within pharmaceutical traceability systems. The advanced models examine past logistics information together with temperature anomalies and supplier performance data and routing interruptions to assess potential product integrity threats that exist before medicines reach hospitals and pharmacies.
The predictive intelligence system enables inventory to be redirected while product recalls are stopped which results in better cold-chain performance and decreased spoilage costs for biologics and vaccines. The digital systems create operational advantages because they speed up compliance responses and decrease recall costs through their ability to trace products through decentralized supply chains. Regional implementations show that they successfully decrease counterfeit risks while they enable quicker authentication of products at the batch level.
The pharmaceutical systems in Asia Pacific countries face two main challenges which prevent them from using artificial intelligence technology because their data sources are incomplete and their enterprise resource planning systems need better connection to their specific operational requirements. The insufficient high-quality historical datasets required to develop accurate predictive models restrict decision-making capabilities for enterprises while hindering their ability to implement enterprise-wide systems.
Key Market Trends
- Pharmaceutical companies in China and India have progressed from conducting separate blockchain pilot projects since 2022 to establishing complete supply chain systems that use serialization technology.
- The period between 2023 and 2026 saw authorities enhance their regulatory enforcement methods, which resulted in more than 40% of major regional manufacturers adopting digital systems for their batch-level verification operations.
- The market trend shows that buyers now prefer blockchain platforms which provide interoperability, which has led IBM and Oracle to develop their API-based system architectures since 2024.
- Pharmaceutical companies started to prioritize cold-chain tracking systems after vaccination distribution problems in Southeast Asian logistics networks showed they needed better tracking systems between 2021 and 2023.
- Since 2023, pharmaceutical exporters have adopted permissioned blockchain systems to meet the import authentication standards required by European and North American markets.
- Mid-sized drug manufacturers who wanted to implement their systems faster began choosing cloud-native deployment models after 2024, which replaced their expensive on-premise infrastructure.
- The emergence of counterfeit drug incidents in regional supply chains led to increased regulator engagement, which resulted in India and Japan introducing new digital audit requirements for their operations.
- The strategic partnerships between SAP, Accenture, and regional pharmaceutical distributors experienced rapid growth beginning in 2024.
- AI-based anomaly detection systems have transformed into crucial elements for competitive advantage which blockchain companies utilize to enhance their recall processes while decreasing their manual verification tasks.
- The implementation of complete compliance automation systems has become the main purchasing factor for pharmaceutical buyers, who now prioritize it over basic ledger functions as they assess various regions for their implementation progress.
Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Segmentation
By Type :
Pharmaceutical companies implement private blockchain systems to create controlled access points which enable secure data management and internal system monitoring. The public blockchain system allows users to see specific supply chain activities at selected times. Pharmaceutical companies and distributors and regulators use consortium blockchain technology because it enables them to share system control which improves their operational coordination and data sharing accuracy.
The various supply chain types increase trust levels for pharmaceutical supply networks. Organizations use private environments to protect their internal systems from breaches while they restrict information access. The public systems provide controlled system visibility which operates according to specific requirements. Consortium models continue gaining preference due to shared governance and improved cooperation between multiple stakeholders involved in drug production, storage, and distribution activities.
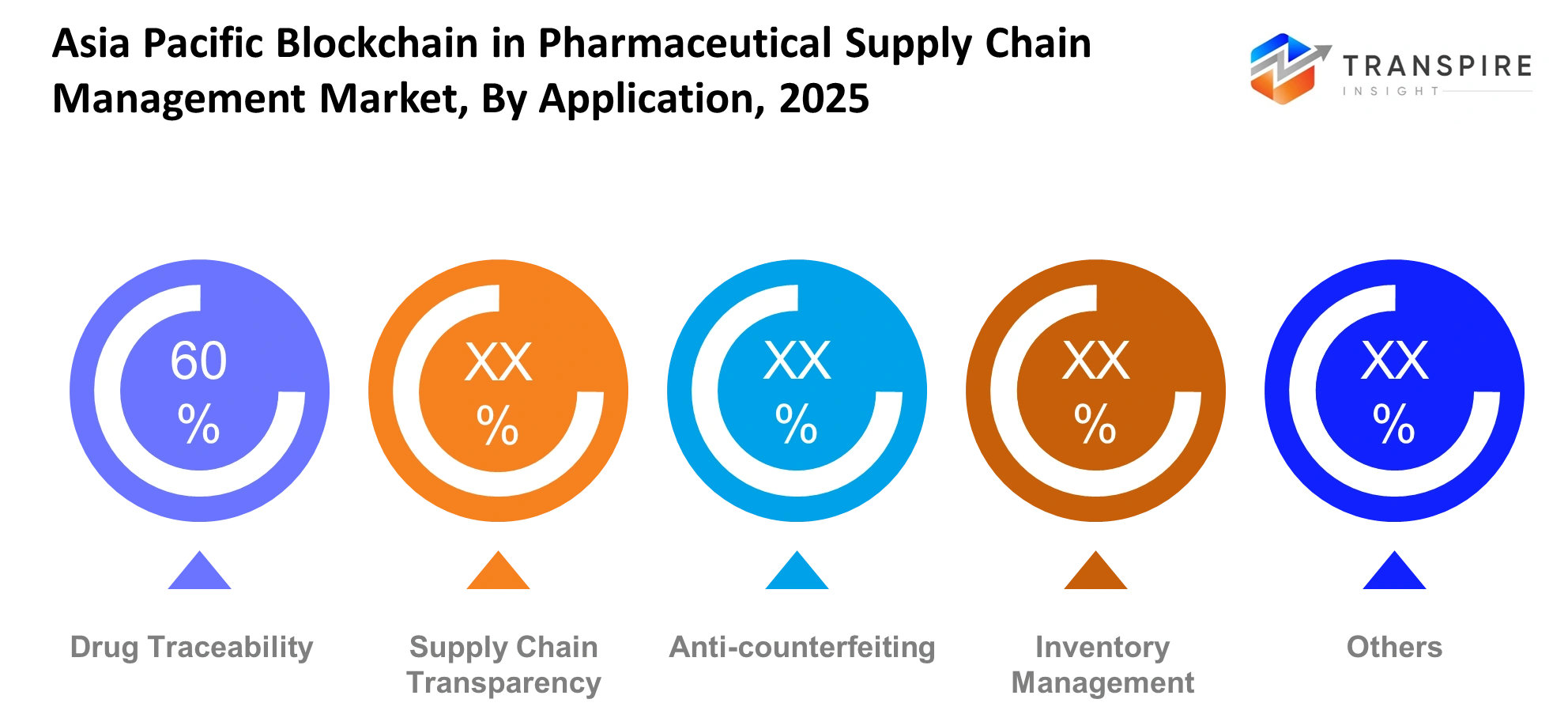
By Application :
The production process of medicines until their delivery remains the primary application for drug traceability. Supply chain transparency improves visibility across multiple stages. Anti-counterfeiting solutions help reduce fake drugs. The inventory management applications enable better stock management which prevents shortages in hospitals and pharmacies and distribution centers throughout the Asia Pacific pharmaceutical distribution system.
These applications enhanceboth operational efficiencyand medicine handling security. Every batch undergoes traceability which establishes clear records of its movement. Transparency builds trust between different groups that have vested interests. The anti-counterfeiting functions protect businesses from financial harm caused by counterfeit products. Inventory systems help maintain balance between demand and supply which enables smoother pharmaceutical operations throughout the region.
To learn more about this report, Download Free Sample Report
By End-User :
Pharmaceutical companies implement blockchain technology to track their manufacturing processes and ensure they meet regulatory requirements. Distributors use this system to verify shipment details and share essential information. Hospitals use this system to secure their medicine acquisition process. Government authorities utilize this system to track their regulatory enforcement activities. Pharmaceutical companies use this system to monitor their shipment status throughout their entire supply chain.
The coordination among different user groups leads to better results and fewer mistakes. Pharmaceutical companies achieve improved tracking capabilities for their inventory. The accuracy of deliveries improves for both distribution companies and logistics organizations. Hospitals create processes which help protect patients from dangerous drug usage. Government agencies enforce regulations which enhance monitoring practices that enable pharmaceutical distribution centers to maintain quality standards while minimizing distribution hazards.
By Deployment :
The adoption of cloud deployment has become common because it offers users simple access and scalable solutions while requiring minimal infrastructure. Organizations that need complete data protection use on-premise systems to manage their information. Companies can use hybrid deployment to operate both secure internal systems and allow external partners to access their systems.
Organizations select their deployment methods based on their security requirements and their need to run operations at various sizes. Cloud solutions enable organizations to implement systems more quickly while facilitating data exchange. Organizations using on-premise systems maintain complete control over their protected pharmaceutical data. The hybrid system enables organizations to manage their internal operations while working with outside parties within the pharmaceutical supply chain.
What are the Key Use Cases Driving the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market?
The primary application of blockchain technology in the Asia Pacific pharmaceutical supply chain market uses drug serialization and product authentication methods. The pharmaceutical manufacturing sector drives market demand because companies need to track their products across complex distribution systems while meeting anti-counterfeit regulations which have become more stringent in countries like India and China.
Cold-chain monitoring and recall management systems receive increasing adoption from vaccine manufacturers and biologics producers and third-party pharmaceutical distribution companies. The use cases enable healthcare distributors to respond quickly when temperature changes or shipment errors occur which helps them decrease spoilage costs and strengthen their regulatory reporting operations.
The market now offers emerging applications which include smart-contract-enabled supplier payments and decentralized clinical trial material tracking solutions. The initial phase of these solutions displays high market potential because pharmaceutical contract manufacturers and research organizations in Singapore and Japan and South Korea develop digital systems which enable automatic compliance management and protected data sharing among multiple organizations.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 0.63 Billion |
|
Market size value in 2026 |
USD 0.76 Billion |
|
Revenue forecast in 2033 |
USD 2.83 Billion |
|
Growth rate |
CAGR of 20.66% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Asia Pacific (India, China, Japan, Australia,South Korea, Rest of Asia Pacific) |
|
Key company profiled |
IBM, Microsoft, Oracle, SAP, Amazon, Deloitte, Accenture, Infosys, TCS, Wipro, Guardtime, Chronicled, Modum, FarmaTrust, VeChain. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Private Blockchain, Public Blockchain, Consortium Blockchain, Others); By Application (Drug Traceability, Supply Chain Transparency, Anti-counterfeiting, Inventory Management, Others); By End-User (Pharma Companies, Distributors, Hospitals, Government, Logistics Providers, Others); By Deployment (Cloud, On-premise, Hybrid, Others). |
Which Regions are Driving the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Growth?
The Asia Pacific blockchain market for pharmaceutical supply chain management operates under Chinese leadership because the country implements aggressive digitalization regulations for pharmaceuticals and maintains centralized drug traceability control through its regulatory system. The pharmaceutical industry adopted blockchain-backed verification systems because national serialization requirements and healthcare regulatory agencies executed strict monitoring procedures.
Japan serves as the second most important regional contributor because the country maintains operational consistency which operates independently from regulatory processes. Japan's market expansion depends on businesses which invest over extended periods while healthcare IT modernization projects maintain their steady progress according to Japanese business practices. Japanese pharmaceutical companies implement blockchain technology through system reliability and cybersecurity and ERP system integration which they consider essential for their operations. Japanese businesses establish their revenue base through controlled development which makes Japan a sustainable market for solution providers who develop high-value pharmaceutical compliance software.
The pharmaceutical industry of India has emerged as the fastest-growing market because it adopts modern technologies and expands its digital compliance requirements. Domestic manufacturers have increased their blockchain investments because they need to improve drug authentication systems and meet international pharmaceutical traceability standards. The market expansion receives additional support from contract manufacturing companies and the rising need for clear export documentation in North American and European trade routes. The period of 2026 to 2033 presents strong business growth opportunities for market entrants and investors because India will move from compliance-based systems to complete supply chain intelligence platforms.
Who are the Key Players in the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market and How Do They Compete?
The Asia Pacific blockchain pharmaceutical supply chain management market exhibits moderate consolidation because global enterprise software and consulting providers control its competition. The existing companies use blockchain technology for their ERP and compliance systems which results in high switching costs that protect their market share. The current competitive landscape now focuses on three key areas which are interoperability and regulatory integration and implementation services instead of technology solutions that operate independently.
IBM uses Hyperledger frameworks to achieve real-time pharmaceutical traceability in international supply chains. Oracle uses blockchain technology to connect procurement and compliance data in its cloud ERP system. SAP integrates supply chain analytics into its S/4HANA system to support regulated pharmaceutical processes. Accenture provides transformation solutions together with regulatory blockchain implementation services to pharmaceutical clients across the Asia Pacific region.
Infosys uses its India-based delivery centers to provide cost-effective blockchain solutions which help mid-tier pharmaceutical companies. VeChain uses its blockchain technology for product authentication and logistics partnerships to create pharmaceutical traceability solutions in China and Southeast Asia. The two companies use pilot projects and regional partnerships to help exporters achieve complete supply chain visibility while maintaining compliance.
Company List
- IBM
- Microsoft
- Oracle
- SAP
- Amazon
- Deloitte
- Accenture
- Infosys
- TCS
- Wipro
- Guardtime
- Chronicled
- Modum
- FarmaTrust
- VeChain
Recent Development News
In March 2026, Wellgistics Health announced selection of Hyperledger Fabric, Ethereum/Polygon, VeChain, Quorum/Besu, and R3 Corda to develop its blockchain-enabled PharmacyChain pharmaceutical traceability platform. The initiative aims to strengthen end-to-end drug serialization and DSCSA-compliant supply chain transparency across its pharmacy network.
Source: https://www.nasdaq.com/
In May 2026, SAP entered a strategic partnership with Chronicled to integrate the MediLedger blockchain network into its pharmaceutical supply chain compliance systems. The collaboration is designed to improve prescription drug verification and strengthen DSCSA compliance by enabling secure, shared blockchain-based drug authentication across stakeholders.
Source: https://erpnews.com/
What Strategic Insights Define the Future of the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market?
The Asia Pacific blockchain in the pharmaceutical supply chain management market is moving toward complete systems that use artificial intelligence for compliance but currently operates with separate traceability systems. The upcoming 5-7 years will see growth through stricter international pharmaceutical identity verification rules and the rising requirement for instant data sharing between multiple partners throughout diverse regional distribution networks. Blockchain technology will develop into the core trust component of digital health systems which will connect to enterprise resource planning systems and logistics operations and compliance tracking systems.
The pharmaceutical industry faces an unrecognized danger because major global enterprise vendors are currently dominating platform development which will reduce system compatibility and force drug manufacturers into vendor dependency. The tightening of pricing frameworks together with the introduction of highly restrictive platform standards will result in decreased operational flexibility among users even though regulatory bodies require system adoption.
Operators should develop decentralized clinical trial supply tracking systems because Singapore and South Korea currently provide digital regulatory approval for clinical trials through their active digital trial development programs. The drug development process will gain from blockchain technology when this use case introduces a new application that connects to logistics operations. The pharmaceutical industry in Asia Pacific should develop blockchain systems that use modular API-first designs which allow AI analytics and regulatory databases to function together for fast expansion across different pharmaceutical systems.
Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market Report Segmentation
By Type
- Private Blockchain
- Public Blockchain
- Consortium Blockchain
By Application
- Drug Traceability
- Supply Chain Transparency
- Anti-counterfeiting
- Inventory Management
By End-User
- Pharma Companies
- Distributors
- Hospitals
- Government
- Logistics Providers
By Deployment
- Cloud
- On-premise
- Hybrid
Frequently Asked Questions
Find quick answers to common questions.
The Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market size is USD 2.83 Billion in 2033.
Key segments for the Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market are By Type (Private Blockchain, Public Blockchain, Consortium Blockchain, Others); By Application (Drug Traceability, Supply Chain Transparency, Anti-counterfeiting, Inventory Management, Others); By End-User (Pharma Companies, Distributors, Hospitals, Government, Logistics Providers, Others); By Deployment (Cloud, On-premise, Hybrid, Others).
Major Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market players are IBM, Microsoft, Oracle, SAP, Amazon, Deloitte, Accenture, Infosys, TCS, Wipro, Guardtime, Chronicled, Modum, FarmaTrust, VeChain.
The Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market size is USD 0.63 Billion in 2025.
The Asia Pacific Blockchain in Pharmaceutical Supply Chain Management Market CAGR is 20.66% from 2026 to 2033.
- IBM
- Microsoft
- Oracle
- SAP
- Amazon
- Deloitte
- Accenture
- Infosys
- TCS
- Wipro
- Guardtime
- Chronicled
- Modum
- FarmaTrust
- VeChain
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


