United States Point-Of-Care Testing (POCT) Market Size & Forecast:
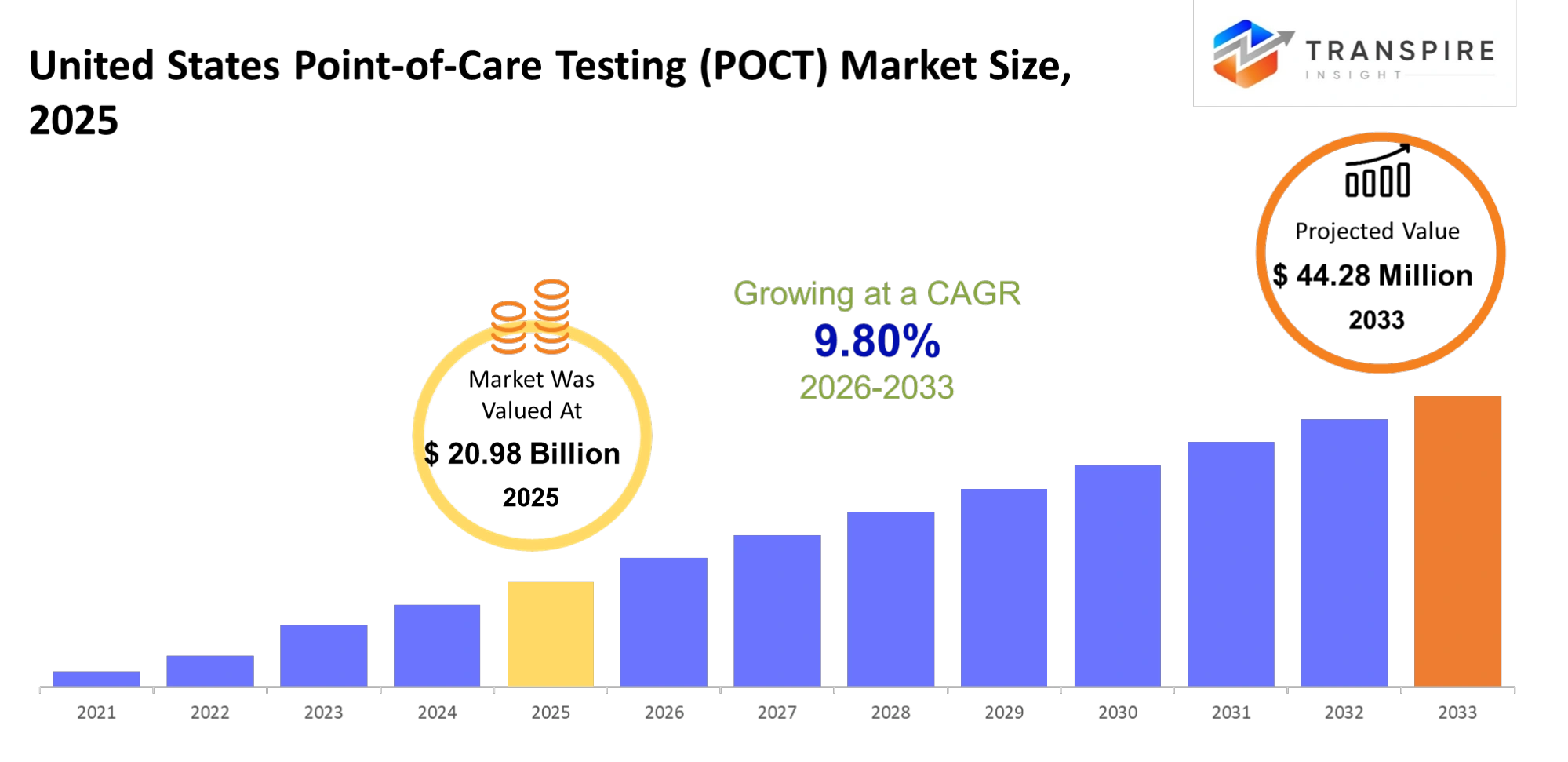
- United States Point-Of-Care-Testing (POCT) Market Size 2025: USD 20.98 Billion
- United States Point-Of-Care-Testing (POCT) Market Size 2033: USD 44.28 Billion
- United States Point-Of-Care-Testing (POCT) Market CAGR: 9.80%
- United States Point-Of-Care Testing (POCT) Market Segments: By Product Type (Glucose Testing Kits, Infectious Disease Testing Kits, Cardiac Marker Testing Kits, Pregnancy & Fertility Testing Kits), By End User (Hospitals, Clinics, Homecare Settings).
To learn more about this report, Download Free Sample Report
United States Point-Of-Care-Testing (POCT) Market Summary:
The United States Point-Of-Care Testing (POCT) Market size is estimated at USD 20.98 Billion in 2025 and is anticipated to reach USD 44.28 Billion by 2033, growing at a CAGR of 9.80% from 2026 to 2033. The United States Point-of-Care Testing (POCT) market is evolving rapidly because there is an increasing demand for fast and precise medical tests which can be performed at the patient's location. POCT solutions enable healthcare providers to deliver faster medical tests which patients can access from clinics and home environments. The industry experiences growth because of technological advancements and increasing cases of chronic diseases and the need for personalised medical treatments. The healthcare industry adopts point-of-care testing because it provides practical solutions which enable healthcare providers to deliver immediate medical care while improving patient satisfaction and treatment results.
Key Market Trends & Insights:
- Healthcare providers require test results to arrive more quickly so they can make their clinical decisions at a faster rate. The use of POCT devices in emergency and critical care environments leads to shorter waiting periods, which helps hospitals achieve better patient management.
- Patients are becoming more comfortable with self-testing at home. This trend, which gained momentum during the pandemic, has created a need for more user-friendly and portable POCT devices which require fewer testing facilities.
- New POCT devices now provide higher precision measurements while being smaller and having digital connectivity capabilities. The integration of smartphones with health applications enables users to track their health status and share their data instantly.
- POCT serves as a diagnostic tool which enables healthcare workers to provide medical services to people living in remote areas. Basic diagnostic testing becomes accessible to remote communities because of its ability to function without laboratories and its simple operation.
- The current regulatory environment provides POCT products with rapid market entry because it streamlines product evaluation processes. This process enables companies to bring their new products into the market at a faster pace.
- POCT technology has expanded beyond hospitals to become an essential part of various medical settings, which include clinics, pharmacies and workplace environments. Healthcare systems now use this technology because it offers flexible applications and shows increasing value in current medical practices.
United States Point-of-Care Testing (POCT) Market Segmentation
By Product Type
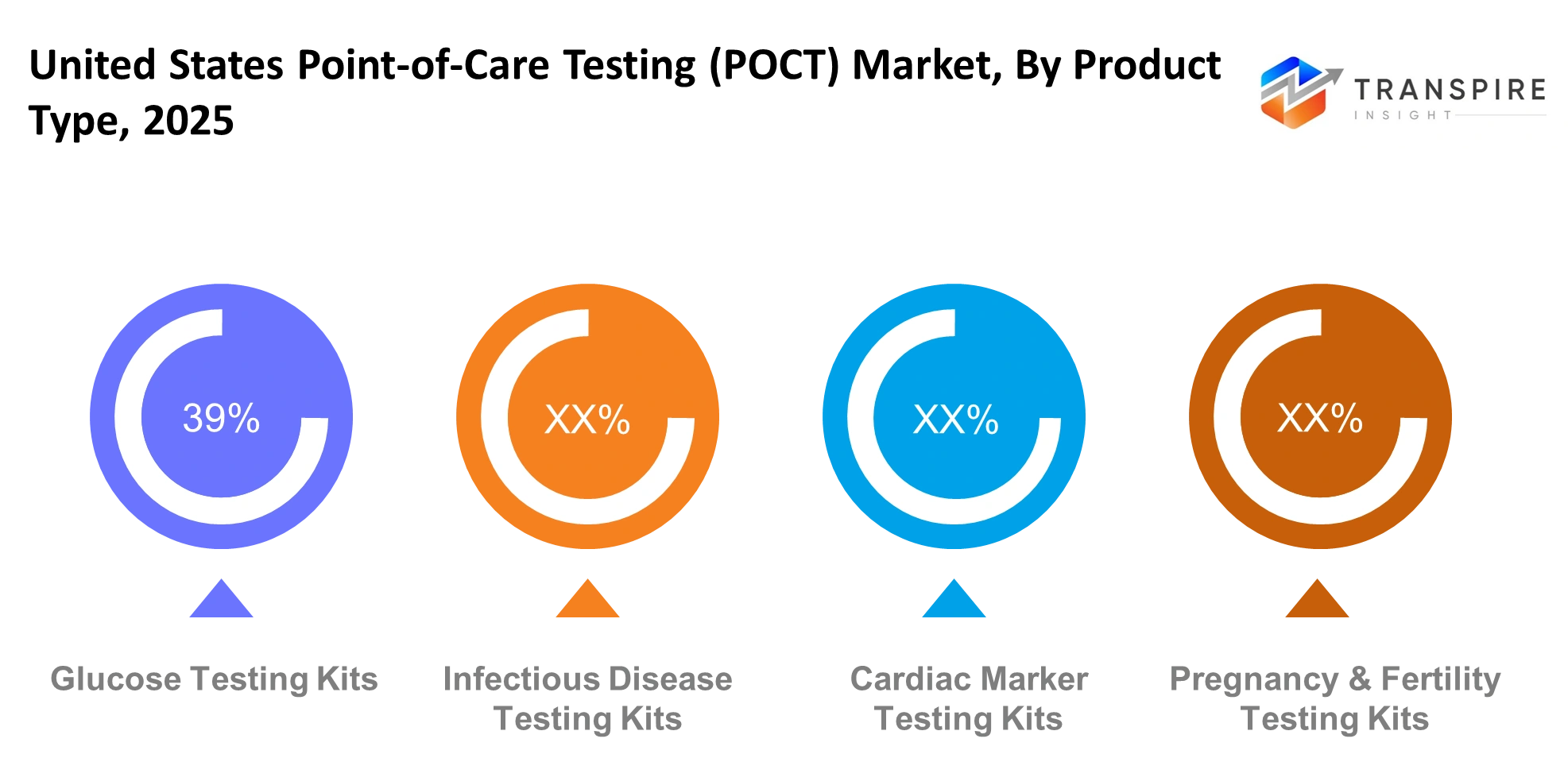
- Glucose Testing Kits: Glucose testing kits remain one of the most widely used POCT products across the United States. These kits enable patients to monitor their blood glucose levels at home and in clinical settings because diabetes rates continue to increase. The product becomes necessary for daily disease management because it provides fast results and affordable pricing and simple operation.
- Infectious Disease Testing Kits: Infectious disease testing kits have gained strong momentum because people now understand the need for early diagnosis. The testing kits provide health professionals with the ability to quickly identify various diseases, which include COVID-19 and influenza and other infections. The system provides fast results which enable doctors to make treatment choices while preventing infectious diseases from spreading.
- Cardiac Marker Testing Kits: Cardiac marker testing kits enable doctors to identify heart-related diseases in patients with high efficiency. The emergency room and ambulatory care settings use the kits to identify markers which doctors link to heart attacks and cardiac stress conditions. The system provides quick results which doctors use to treat patients while improving their chances of surviving medical emergencies.
- Pregnancy & Fertility Testing Kits: Pregnancy and fertility testing kits offer users three main benefits through their ability to provide results in complete privacy during testing. The testing kits which customers can purchase in stores give people the ability to track their reproductive health from their home environment. The increasing need for self-care products has led more people into using these products, which businesses now sell to different customer segments.
To learn more about this report, Download Free Sample Report
By End User
- Hospitals: As the main institution for using POCT devices, hospitals require these tools because they need instant access to diagnostic results. Rapid testing in these environments supports emergency medical treatment and intensive medical care and surgical operations. Through faster clinical decision-making the POCT system improves patient results while decreasing laboratory needs and streamlining operational processes.
- Clinics: Clinics increasingly adopt POCT solutions to provide quicker consultations and treatments. The ability to conduct immediate testing enables clinics to optimise their operational processes while delivering superior services to patients. Healthcare professionals use these devices to diagnose and treat patients during their first appointment.
- Homecare Settings: The POCT market has identified homecare environments as a developing market segment. Patients choose to control their health from their home environments by utilising simple-to-use medical equipment. The current trend enables individuals to maintain self-sufficiency while decreasing their need for hospital treatment, which supports the movement toward personalised and patient-focused healthcare delivery methods.
Country Insight
The United States Point-of-Care Testing (POCT) market shows a changing healthcare environment which resulted from three factors: new medical technologies, improved testing options, and the development of services that prioritise patient care. The country has quickly adopted point-of-care testing solutions, which medical systems established through their existing infrastructure at hospitals, clinics, and residential facilities. The increasing demand for early disease detection together with preventive health measures is driving more people to use these fast-testing solutions. Because healthcare expenses keep rising, medical providers need to find diagnostic methods which maintain accuracy while decreasing expenses, which drives them to choose point-of-care testing as their preferred option.
POCT provides essential diagnostic services to rural and underserved regions of the United States because it enables patients to access testing services which require no advanced laboratory facilities. The market expansion receives support through digital system integration and the development of portable technology solutions. The existing regulatory frameworks, together with the continuous financial support for healthcare research, provide businesses with sufficient resources to create new products. The demand for testing solutions which provide quick results and improved patient results has led to the U.S. POCT market, which has developed into a major testing solution.
Recent Development News
FDA Expands Access to Self-Collected Cancer Screening: The U.S. government is promoting self-collection methods for cervical cancer testing, making screening more accessible and convenient. This move supports wider adoption of point-of-care and at-home diagnostic solutions.
Growing Adoption of Multi-Disease At-Home Testing Kits: New combination tests for multiple infections are gaining traction in the U.S., offering convenience and faster diagnosis. These tools reflect a shift toward integrated POCT solutions.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 20.98 Billion |
|
Market size value in 2026 |
USD 23.02 Billion |
|
Revenue forecast in 2033 |
USD 44.28 Billion |
|
Growth rate |
CAGR of 9.80% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
United States |
|
Key company profiled |
Abbott Laboratories, Roche Diagnostics, Siemens Healthineers AG, Danaher Corporation, Thermo Fisher Scientific Inc., Becton Dickinson and Company, Bio-Rad Laboratories Inc., QuidelOrtho Corporation, Nova Biomedical Corporation, EKF Diagnostics Holdings PLC, Trinity Biotech PLC, OraSure Technologies Inc., Meridian Bioscience Inc., Chembio Diagnostics Inc., HemoCue AB. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product Type (Glucose Testing Kits, Infectious Disease Testing Kits, Cardiac Marker Testing Kits, Pregnancy & Fertility Testing Kits), By End User (Hospitals, Clinics, Homecare Settings). |
Key United States Point-Of-Care-Testing (POCT) Company Insights
The United States Point-of-Care Testing (POCT) market operates with intense competition because companies must create innovative products which deliver accurate results while being easy for users to operate. Leading players are investing in advanced technologies through digital connectivity and portable diagnostics systems to satisfy increasing demand from healthcare facilities. Companies use strategic partnerships and product launches which combine with their research efforts to create their competitive environment. Companies are also prioritising affordability and accessibility to expand their reach. Businesses must develop faster and more trustworthy solutions because patient demands are changing, which will improve healthcare delivery and result in superior medical results.
Company List
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers AG
- Danaher Corporation
- Thermo Fisher Scientific Inc.
- Becton Dickinson and Company
- Bio-Rad Laboratories Inc.
- QuidelOrtho Corporation
- Nova Biomedical Corporation
- EKF Diagnostics Holdings PLC
- Trinity Biotech PLC
- OraSure Technologies Inc.
- Meridian Bioscience Inc.
- Chembio Diagnostics Inc.
- HemoCue AB
United States Point-Of-Care-Testing (POCT) Market Report Segmentation
By Product Type
- Glucose Testing Kits
- Infectious Disease Testing Kits
- Cardiac Marker Testing Kits
- Pregnancy & Fertility Testing Kits
By End User
- Hospitals
- Clinics
- Homecare Settings
Frequently Asked Questions
Find quick answers to common questions.
The approximate United States Point-Of-Care-Testing (POCT) Market size for the market will be USD 44.28 Billion in 2033.
The key segments of the United States Point-Of-Care-Testing (POCT) Market are By Product Type (Glucose Testing Kits, Infectious Disease Testing Kits, Cardiac Marker Testing Kits, Pregnancy & Fertility Testing Kits), By End User (Hospitals, Clinics, Homecare Settings).
Major players in the United States Point-Of-Care-Testing (POCT) Market are Abbott Laboratories, Roche Diagnostics, Siemens Healthineers AG, Danaher Corporation, Thermo Fisher Scientific Inc., Becton Dickinson and Company, Bio-Rad Laboratories Inc., QuidelOrtho Corporation, Nova Biomedical Corporation, EKF Diagnostics Holdings PLC, Trinity Biotech PLC, OraSure Technologies Inc., Meridian Bioscience Inc., Chembio Diagnostics Inc., HemoCue AB.
The current market size of the United States Point-Of-Care-Testing (POCT) Market is USD 20.98 Billion in 2025.
The United States Point-Of-Care-Testing (POCT) Market CAGR is 9.80%.
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers AG
- Danaher Corporation
- Thermo Fisher Scientific Inc.
- Becton Dickinson and Company
- Bio-Rad Laboratories Inc.
- QuidelOrtho Corporation
- Nova Biomedical Corporation
- EKF Diagnostics Holdings PLC
- Trinity Biotech PLC
- OraSure Technologies Inc.
- Meridian Bioscience Inc.
- Chembio Diagnostics Inc.
- HemoCue AB
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









