United Kingdom Medical Device Contract Manufacturing Market Size & Forecast:
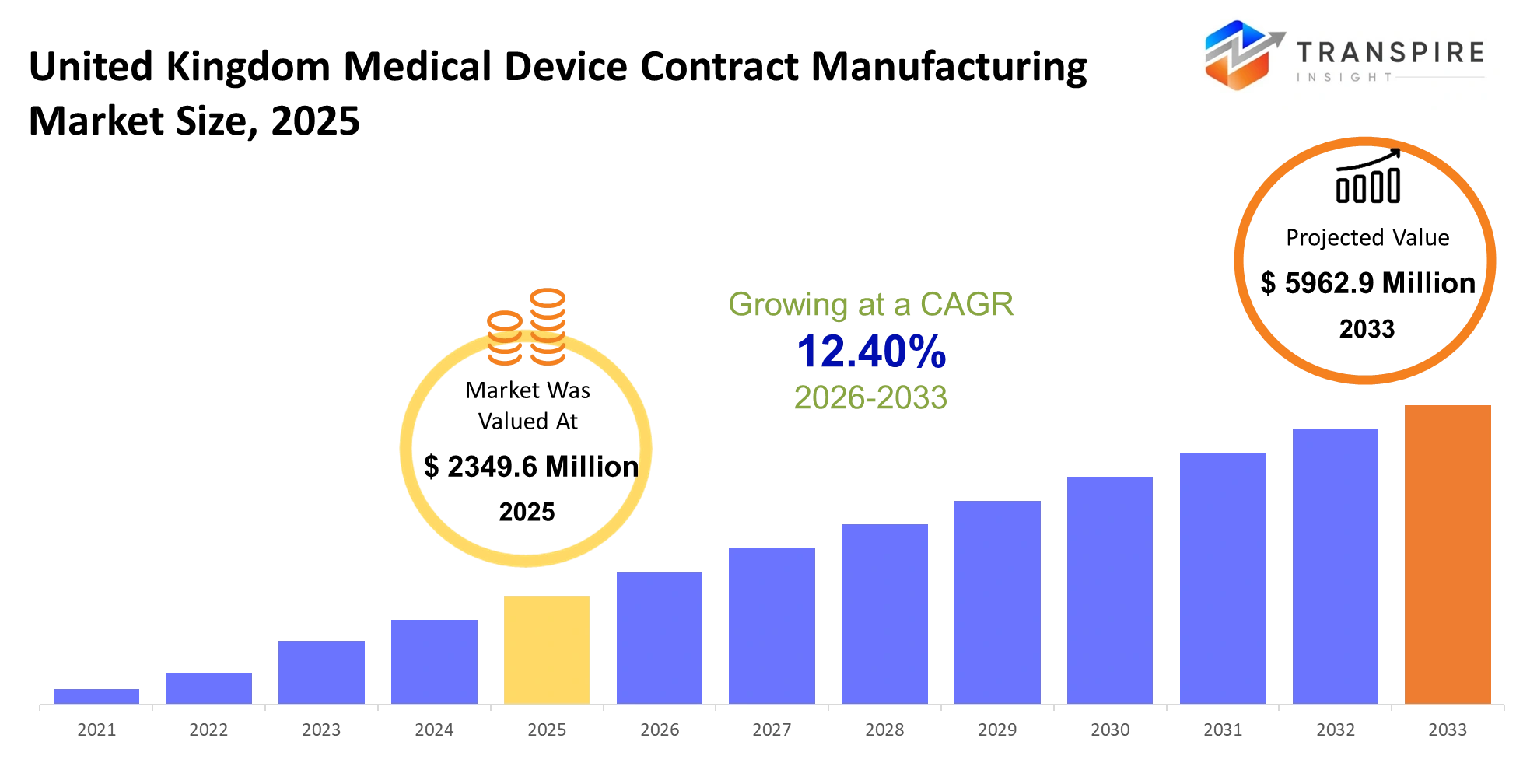
- United Kingdom Medical Device Contract Manufacturing Market Size 2025: USD 2349.6 Million
- United Kingdom Medical Device Contract Manufacturing Market Size 2033: USD 5962.9 Million
- United Kingdom Medical Device Contract Manufacturing Market CAGR: 12.40%
- United Kingdom Medical Device Contract Manufacturing Market Segments: By Service Type (Device Development & Manufacturing, Assembly Services, Packaging Services), b (Diagnostic Devices, Therapeutic Devices, Monitoring Devices), By End-User (Medical Device Companies, Pharmaceutical Companies).
To learn more about this report, Download Free Sample Report
United Kingdom Medical Device Contract Manufacturing Market Summary:
The United Kingdom Medical Device Contract Manufacturing Market size is estimated at USD 2349.6 Million in 2025 and is anticipated to reach USD 5962.9 Million by 2033, growing at a CAGR of 12.40% from 2026 to 2033. The united kingdom medical device contract manufacturing market within the healthcare and medical technology industry will reflect a steady shift toward outsourcing complex production tasks to specialized partners. Medical device companies will rely on external manufacturers to meet strict quality and compliance standards set by national regulations. The combination of automation technologies and precision engineering solutions will create a reliable production system that minimizes mistakes during operations. The need for dependable and on-schedule products will increase manufacturing demands according to changing expectations from healthcare providers. Regular updates to regulations will establish safety requirements and traceability standards together with documentation practices that must be followed throughout manufacturing processes and product lifecycle management.
Key Market Trends & Insights:
The market needs specialized manufacturing services because advanced production facilities which produce accurate results must meet compliance requirements. Medical device companies will require contract manufacturing services to obtain their advanced components and superior production standards. The shift will decrease internal work demands while maintaining constant observance of all healthcare requirements.
The manufacturing industry needs to adopt new technologies through which companies will develop their operations using automation and robotics and additive manufacturing. The technologies will enhance operational efficiency while decreasing mistakes and enabling speedier production processes. The manufacturers who invest in modern systems will achieve their production goals by meeting specific requirements while maintaining high-quality output throughout their large-scale operations.
The organization needs to handle regulatory compliance while executing its quality assurance processes. The organization will continue to operate according to established regulatory requirements. The contract manufacturers will establish better quality control systems which they will use to create comprehensive records. The organization will use regular audits together with updated guidelines to ensure product safety and maintain healthcare sector approval.
The medical device industry shows a growing trend of companies outsourcing production processes. Companies will choose to outsource their production requirements because it provides them with greater operational flexibility while enabling them to control expenses. The companies will delegate their production responsibilities to external partners which will enable them to concentrate on their research and product development activities. The method enables businesses to achieve rapid growth through expansion without requiring major construction expenses.
Service providers will provide complete service packages which include all aspects of product development from design to prototyping to manufacturing and packaging. The business model will streamline operational processes while creating less complicated supply chain networks. Companies will gain advantages by partnering with a single organization which will improve their interdepartmental collaboration and enable them to bring products to market more quickly.
United Kingdom Medical Device Contract Manufacturing Market Segmentation
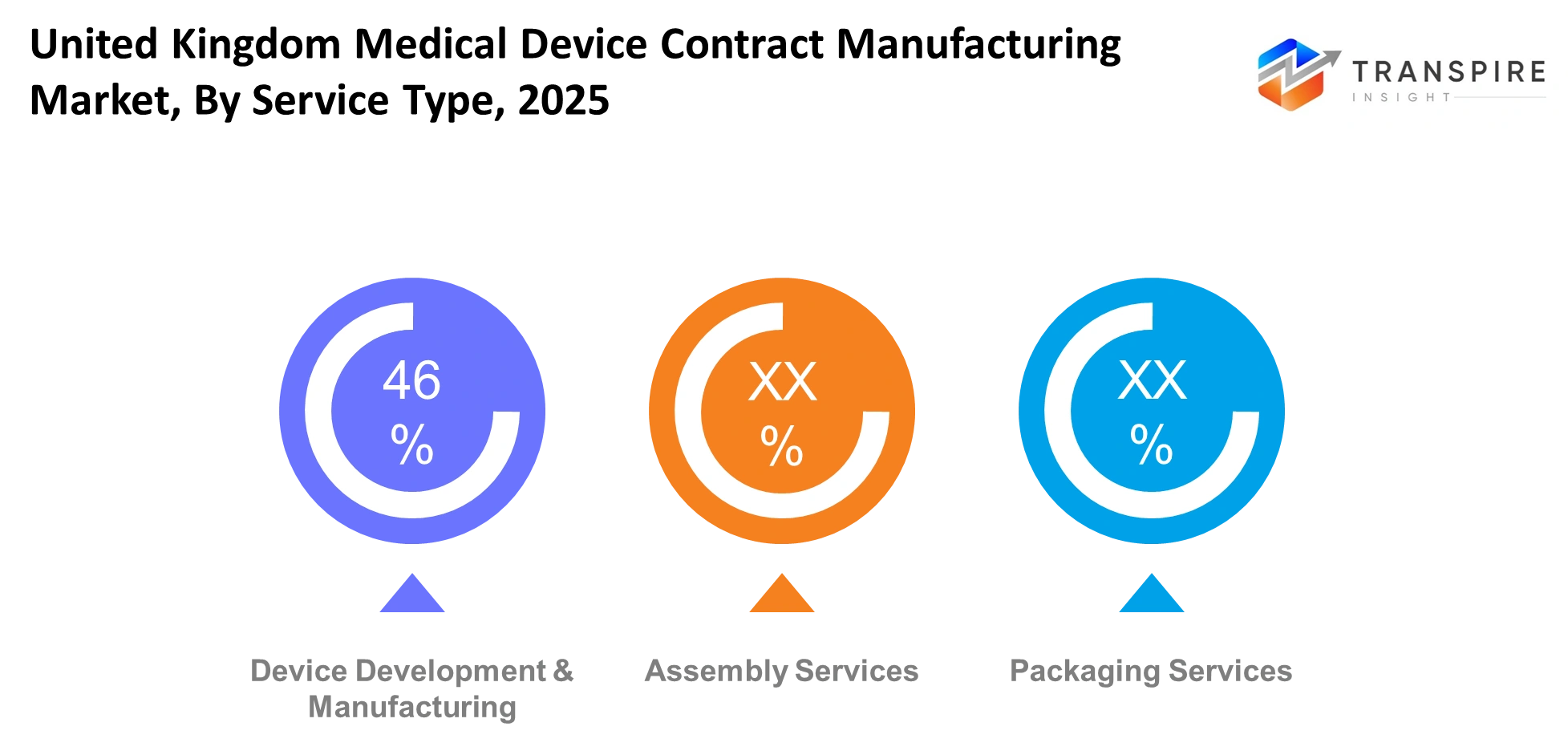
By Service Type
- Device Development & Manufacturing: This service type will cover the full process from design support to final production of medical devices. Manufacturers will work closely with companies to refine product concepts and ensure compliance with regulatory standards. Advanced engineering and precision tools will support consistent output and high-quality performance across complex devices.
- Assembly Services: Assembly services will focus on combining various components into finished or semi-finished medical devices. Cleanroom environments and strict quality checks will ensure safety and reliability. This service will help companies streamline production by outsourcing detailed assembly tasks to experienced providers with specialized capabilities.
- Packaging Services: Packaging services will ensure that medical devices are safely packed, labeled, and prepared for distribution. Compliance with healthcare regulations will guide packaging standards, including sterilization and traceability. Proper packaging will protect product integrity and support safe transportation and storage across different healthcare settings.
To learn more about this report, Download Free Sample Report
By Device Type
- Diagnostic Devices: This category includes equipment which operates to discover and determine medical conditions. Contract manufacturers will focus on precision components together with sensor integration and they will work to achieve accurate performance results. The production process will follow strict quality control standards and compliance requirements which will produce dependable results for laboratories and hospitals and point-of-care facilities.
- Therapeutic Devices: The development of therapeutic devices will focus on creating medical products which treat patients and assist with their medical needs. The production process demands that all products must achieve minimum safety requirements together with maximum product longevity and continuous operational performance. Companies will depend on their specialized partners to build advanced medical equipment which will meet all required safety standards while maintaining its operational effectiveness in multiple medical procedures.
- Monitoring Devices: The category of monitoring devices includes all equipment which tracks changes in patient health status throughout multiple time periods. The production process will create dependable systems which maintain uninterrupted connection and deliver precise data in real time. The manufacturers will develop their products through the combination of cutting-edge electronic systems and software solutions which will enable ongoing health monitoring in both medical facilities and residential settings.
By End-User
- Medical Device Companies: This segment will remain the primary user of contract manufacturing services. Companies will depend on external partners to handle complex production processes while they focus on design and innovation work. Strong emphasis will be placed on quality, compliance, and scalability to ensure that devices meet healthcare standards and reach the market efficiently.
- Pharmaceutical Companies: Pharmaceutical firms will increasingly use contract manufacturing for combination products such as drug delivery devices. The companies need both exact engineering work and complete compliance with all regulatory requirements. The special manufacturers will help the companies integrate their drugs with delivery systems which will deliver safety together with consistency and effective performance.
Country Insights
You have received training based on information which existed until the month of October in the year 2023. The northern regions of the country will gain contract manufacturing capabilities through their industrial infrastructure and their existing engineering expertise. The cities of Manchester and Sheffield together with Leeds will provide production support through their skilled workforce and their enduring manufacturing capabilities. The local facilities will specialize in creating precision components which can be produced at various scales through their partnerships with academic institutions and research facilities. The relationship will enhance technical expertise while establishing uniform quality benchmarks for medical device production throughout its manufacturing operations.
Southern regions of the country will establish operational links to both innovation centers and government regulatory facilities. The areas surrounding London Cambridge and Oxford will become business centers for companies developing high-value medical devices which use advanced technology. The manufacturing companies in these areas will use their resources to develop automation systems digital technologies and advanced materials which will help them produce complex products. The closer relationship with regulatory agencies will help organizations achieve better compliance standards while their approval procedures will become more efficient.
The two regions will use their unique strengths to execute their work while they progress toward a common mission. The northern regions of the country will focus on production efficiency together with their engineering capabilities, while the southern regions will concentrate on innovation together with their ability to comply with regulations. The system will create a manufacturing network that connects all its parts while operating in accordance with technical standards and legal requirements.
Recent Development News
In April 2026, Integer Holdings Corporation announced the acquisition of UK-based contract manufacturer Aran Biomedical to expand its European manufacturing footprint and strengthen capabilities in minimally invasive device components.
In March 2026, Nordson Corporation completed the acquisition of Atrion Corporation, enhancing its medical device contract manufacturing portfolio, including operations serving UK and European medtech customers.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 2349.6 Million |
|
Market size value in 2026 |
USD 2631.5 Million |
|
Revenue forecast in 2033 |
USD 5962.9 Million |
|
Growth rate |
CAGR of 12.40% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
United Kingdom |
|
Key company profiled |
Flex Ltd., Jabil Inc., Sanmina Corporation, Plexus Corp., Celestica Inc., TE Connectivity Ltd., Integer Holdings Corporation, Benchmark Electronics Inc., West Pharmaceutical Services Inc., Gerresheimer AG, Phillips-Medisize (Molex LLC), Nemera Development S.A., Nolato AB, Viant Medical LLC, Nortech Systems Inc. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Service Type (Device Development & Manufacturing, Assembly Services, Packaging Services), By Device Type (Diagnostic Devices, Therapeutic Devices, Monitoring Devices), By End-User (Medical Device Companies, Pharmaceutical Companies). |
Key United Kingdom Medical Device Contract Manufacturing Market Company Insights
The united kingdom medical device contract manufacturing market will see key company activities that show equal participation from established engineering companies and specialized medical production companies which develop precise products while maintaining industry standards. The major industry players will maintain their commitment to advanced manufacturing technologies through their investments in automated systems and cleanroom facilities which enable them to fulfill healthcare compliance requirements. The companies will develop their quality assurance systems and documentation methods to satisfy the changing demands of regulatory standards. The manufacturers will maintain their primary focus on collaborating with medical device developers which enables them to provide assistance during both the initial prototyping phase and the complete production phase.
Small and mid-sized companies will concentrate on developing specialized skills which include micro-engineering and custom component production and flexible manufacturing operations. The emerging medtech companies will use these companies to build strong relationships which require manufacturing solutions that can be modified according to their needs. The industrial sector will adopt better manufacturing practices through strategic alliances and technology improvements and the introduction of complete service capabilities which will allow businesses to create manufacturing systems that respond quickly to market demands while delivering advanced technological solutions.
Company List
- Flex Ltd.
- Jabil Inc
- Sanmina Corporation
- Plexus Corp.
- Celestica Inc.
- TE Connectivity Ltd.
- Integer Holdings Corporation
- Benchmark Electronics Inc.
- West Pharmaceutical Services Inc.
- Gerresheimer AG
- Phillips-Medisize
- Molex LLC
- Nemera Development S.A.
- Nolato AB
- Viant Medical LLC
- Nortech Systems Inc
United Kingdom Medical Device Contract Manufacturing Market Report Segmentation
By Service Type
- Device Development & Manufacturing
- Assembly Services
- Packaging Services
By Device Type
- Diagnostic Devices
- Therapeutic Devices
- Monitoring Devices
By End-User
- Medical Device Companies
- Pharmaceutical Companies
Frequently Asked Questions
Find quick answers to common questions.
The approximate United Kingdom Medical Device Contract Manufacturing Market size for the market will be USD 5962.9 Million in 2033.
Key segments for the United Kingdom Medical Device Contract Manufacturing Market By Service Type (Device Development & Manufacturing, Assembly Services, Packaging Services), By Device Type (Diagnostic Devices, Therapeutic Devices, Monitoring Devices), By End-User (Medical Device Companies, Pharmaceutical Companies).
Major United Kingdom Medical Device Contract Manufacturing Market players are Flex Ltd., Jabil Inc., Sanmina Corporation, Plexus Corp., Celestica Inc., TE Connectivity Ltd., Integer Holdings Corporation, Benchmark Electronics Inc., West Pharmaceutical Services Inc., Gerresheimer AG, Phillips-Medisize (Molex LLC), Nemera Development S.A., Nolato AB, Viant Medical LLC, Nortech Systems Inc.
The United Kingdom Medical Device Contract Manufacturing Market size is USD 2349.6 Million in 2025.
The United Kingdom Medical Device Contract Manufacturing Market CAGR is 12.40%.
- Flex Ltd.
- Jabil Inc
- Sanmina Corporation
- Plexus Corp.
- Celestica Inc.
- TE Connectivity Ltd.
- Integer Holdings Corporation
- Benchmark Electronics Inc.
- West Pharmaceutical Services Inc.
- Gerresheimer AG
- Phillips-Medisize
- Molex LLC
- Nemera Development S.A.
- Nolato AB
- Viant Medical LLC
- Nortech Systems Inc
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
May 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









