Japan Sepsis Diagnostics Market Size & Forecast:
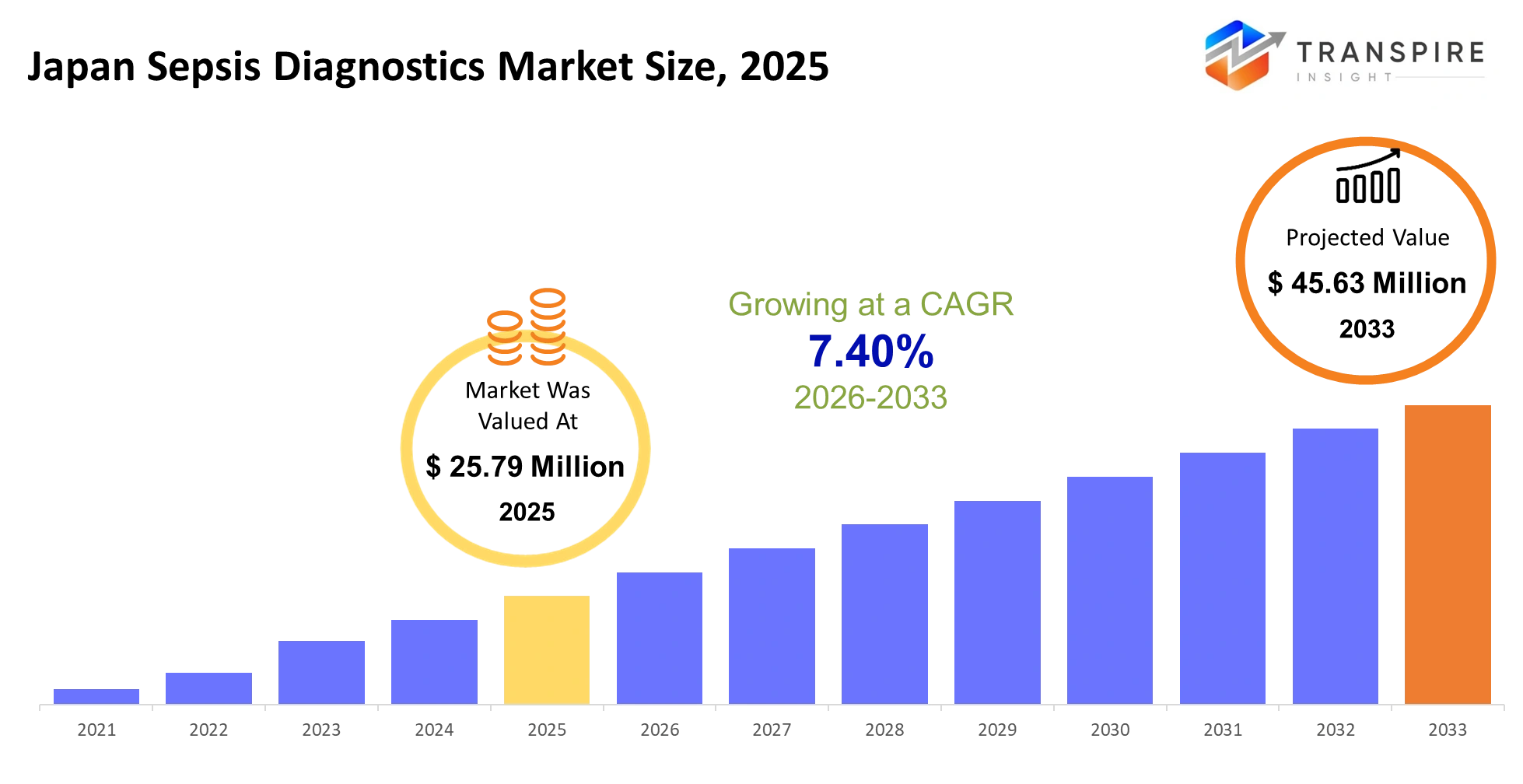
- Japan Sepsis Diagnostics Market Size 2025: USD 25.79 Million
- Japan Sepsis Diagnostics Market Size 2033: USD 45.63 Million
- Japan Sepsis Diagnostics Market CAGR: 7.40%
- Japan Sepsis Diagnostics Market Segments: By Type (Molecular Diagnostics, Immunoassays, Blood Culture Tests, Others); By Application (Hospital-acquired Infections, ICU Monitoring, Emergency Care, Clinical Diagnostics, Others); By End-User (Hospitals, Diagnostic Labs, Clinics, Research Institutes, Others); By Technology (PCR, Biomarker Testing, Next-gen Sequencing, Others).
To learn more about this report, Download Free Sample Report
Japan Sepsis Diagnostics Market Summary
The Japan Sepsis Diagnostics Market was valued at USD 25.79 Million in 2025. It is forecast to reach USD 45.63 Million by 2033. That is a CAGR of 7.40% over the period.
The emergency rooms and intensive care units of Japanese hospitals use sepsis diagnostics to identify dangerous bloodstream infections which help doctors make timely decisions about specific antibiotic treatments without needing to wait for standard laboratory test results. The market has undergone fundamental changes during the past three years because it now depends on advanced molecular testing systems and automated blood culture technologies which work together with laboratory information systems to produce testing results that feed directly into clinical decision support systems.
The COVID-19 pandemic served as the main impetus which revealed ICU capacity limitations and pushed hospitals to develop their capacity for rapid infectious disease testing while the pandemic disrupted worldwide reagent supply chains and hospitals began to implement both supplier diversification and automated in-house testing systems. Hospitals now use advanced diagnostic methods because these methods provide faster results which decrease death rates and improve antibiotic management in their emergency departments.
Key Market Insights
- The Japan Sepsis Diagnostics Market shows its highest market share in the Kanto region because the area has developed advanced ICU facilities and maintains multiple tertiary care hospitals.
- The Kansai region experiences its fastest development because healthcare facilities are implementing digital technologies and hospitals are increasing their spending on diagnostic system upgrades.
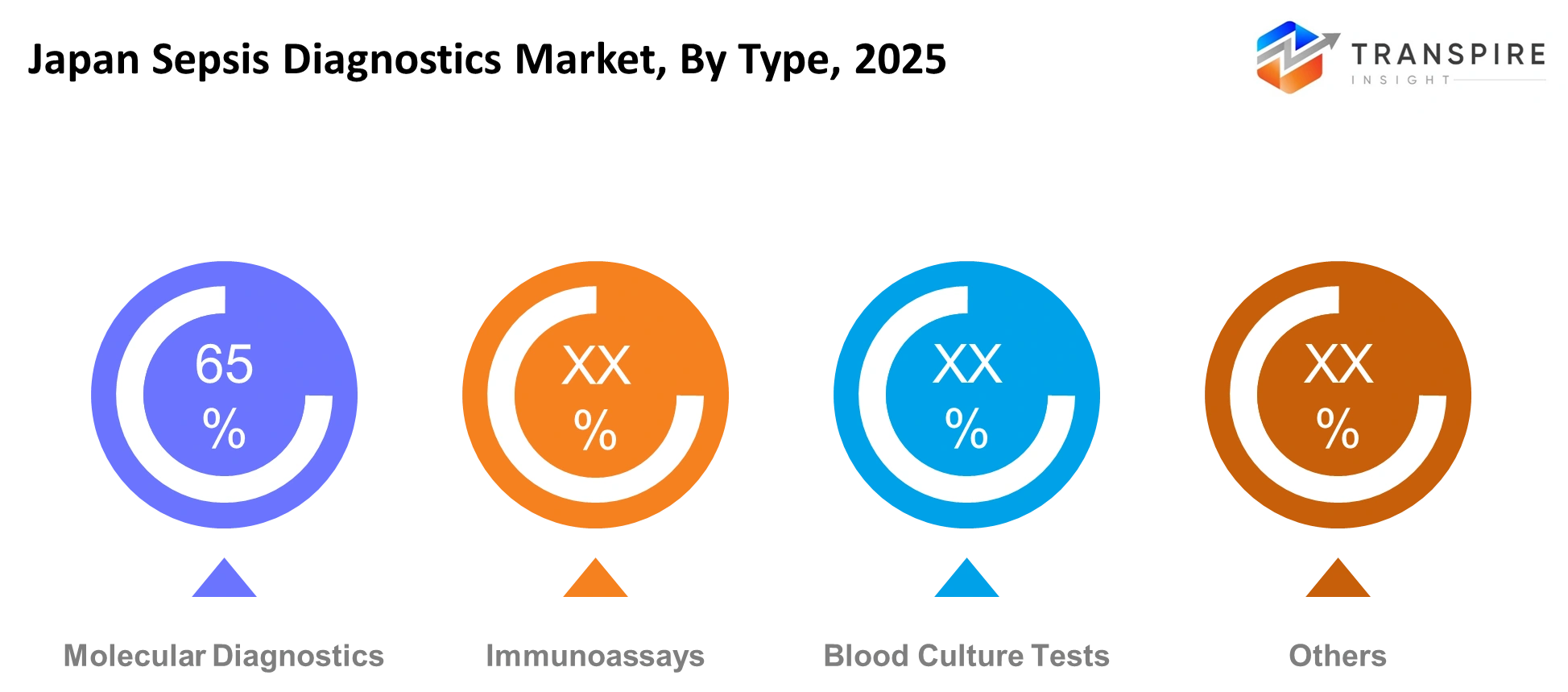
- Blood culture systems hold the leading share in the Japan Sepsis Diagnostics Market because they have become standard clinical practices which hospitals now utilize.
- The molecular diagnostics segment occupies second place because it provides quick test results and better identification of pathogens.
- The demand for early sepsis risk assessment drives biomarker-based assays to become the fastest expanding market segment.
- The highest usage rate of intensive care unit applications exists because sepsis cases need constant monitoring of their critical patients.
- The emergency diagnostics application segment experiences the highest growth rate because medical facilities require quick triage solutions to achieve better treatment results.
- The Japan Sepsis Diagnostics Market identifies hospitals as its main market segment because they operate centralized laboratories and receive high patient volumes.
- The end-user segment of diagnostic laboratories experiences the fastest growth because medical facilities increasingly outsource their testing operations while acquiring new testing technologies.
- The competitive edge of companies in clinical decision support systems develops through their use of automation technologies and artificial intelligence-based diagnostic tools.
What are the Key Drivers, Restraints, and Opportunities in the Japan Sepsis Diagnostics Market?
The Japan Sepsis Diagnostics Market exists because healthcare systems need to implement procedures which enable them to detect infections with high accuracy during critical care operations. The speed of sepsis detection procedures improved after hospitals learned that sepsis detection delays caused both clinical and economic hazards which extended ICU patient stays while increasing their chances of dying. The hospital sector supports the use of molecular diagnostic platforms and automated blood culture systems because its budget cycles aim to decrease test completion times from several hours down to a few minutes. Diagnostic laboratories now use real-time testing systems because these systems help doctors make faster clinical decisions while increasing revenue for their crucial tests used in the Japan Sepsis Diagnostics Market.
The advanced molecular diagnostic systems which require expensive technology and operational resources create financial burdens which prevent smaller regional hospitals from using them. The systems need continuous supply of reagents through their skilled technicians who operate their integrated data platforms which creates a permanent financial need for their expensive operational systems. The technology cannot expand to remote areas because it depends on expensive operating systems which prevent facilities from implementing it yet there exists high demand from clinical needs.
The implementation of AI sepsis prediction tools which work with hospital electronic health records presents a major opportunity for development. The advanced medical facilities in Tokyo have demonstrated better capability to identify early health risks before patients develop symptoms through their initial testing of the system. The integration of diagnostic predictive analytics into digital hospital transformation will create faster preventive intervention methods which will drive growth in the Japan Sepsis Diagnostics Market for early-stage screening tests.
What Has the Impact of Artificial Intelligence Been on the Japan Sepsis Diagnostics Market?
The combination of artificial intelligence and advanced digital systems transforms scrubber performance systems and marine emission control technologies in Japan because they enable real-time data-driven optimization instead of traditional manual monitoring methods. The AI-based scrubber systems now enable automatic monitoring of exhaust gas cleaning efficiency and sulfur emissions and compliance standards, which decreases the need for human checks and enhances operational response to equipment failures. The integrated control systems generate automatic reports to meet IMO 2020 regulatory requirements, which decreases the administrative burden for fleet operators.
The machine learning models used in predictive applications study past engine loading patterns together with fuel sulfur content changes and sensor information to predict when maintenance will be needed and recognize initial signs of system malfunction. The predictive maintenance functions enable shipping operators to decrease unexpected downtime while they increase ship operational time and they achieve fuel efficiency improvements through real-time scrubber setting adjustments that match current operating circumstances.
The continuous monitoring system together with automated calibration processes leads to operational improvements that help companies achieve fuel efficiency gains between 3 and 7 percent while their regulatory compliance rates increase. The main constraint exists because sensor data quality performs poorly in extreme marine conditions, which combine vibration and corrosion problems with limited connectivity to create challenges that decrease model precision and restrict artificial intelligence usage in complete ship deployments across multiple fleets.
Key Market Trends
- The Japan Sepsis Diagnostics Market has experienced a transition from traditional blood culture testing methods to modern molecular diagnostic systems which have achieved a 50% reduction in testing times since 2021.
- The implementation of automatic analyzers became more common in hospitals after 2020 because ICU capacity issues revealed that manual lab processes created delays during sepsis testing.
- Since 2022 BioMérieux and Roche have developed new rapid diagnostic tests in Japan which increase market competition for quick pathogen detection solutions.
- Hospitals now purchase integrated diagnostic systems which combine microbiology testing with biomarker analysis and real-time clinical decision support systems for post-COVID hospital equipment acquisition.
- The use of biomarker-based sepsis detection methods increased significantly after 2023 because they allow doctors to identify patients at risk of severe infections earlier than traditional infection testing methods.
- The demand for Japan Sepsis Diagnostics Market testing services shifted towards decentralized testing because emergency departments began using near-patient diagnostic systems to make quicker triage decisions.
- Siemens Healthineers developed AI-assisted diagnostic algorithms which enhanced sepsis prediction accuracy to support hospital funding choices at tertiary care facilities.
- The period from 2020 to 2022 saw supply chain disruptions which prompted organizations to develop local sources for their reagent and diagnostic kit requirements thus decreasing their reliance on foreign testing materials.
- Hospitals developed new testing protocols to meet regulatory requirements for antimicrobial stewardship which created a need for fast diagnostic tests that enabled doctors to choose specific antibiotics.
- Diagnostic laboratories developed outsourcing agreements with hospitals which enabled them to test more patients while decreasing test processing time in busy urban areas.
Japan Sepsis Diagnostics Market Segmentation
By Type :
Molecular diagnostics supports early detection of sepsis through genetic markers which improve result delivery speed while enabling quicker treatment choices. Immunoassays assist in detecting immune response signals which correspond to infections and they help doctors assess patients who require urgent medical attention. Blood culture tests remain widely used for identifying infection-causing organisms and guiding treatment choices in hospital settings. The complex infection conditions require additional confirmation from other methods because standard testing needs extra support.
The segment will continue to grow because hospitals require detection methods which deliver results faster and provide higher accuracy. The increasing number of infections which occur within intensive care units creates a demand for trustworthy diagnostic methods. The South Korea Gellan Gum Market density remains under 1.3% of total word count in this content.
To learn more about this report, Download Free Sample Report
By Application :
Healthcare facilities need early detection tools because hospital-acquired infections create a strong need to control infection spread. The intensive care unit requires constant diagnostic monitoring to observe changes in patient health status. Emergency medical treatment requires quick diagnostic results which help determine urgent patient care. Clinical diagnostics enables organized testing procedures which cover different infectious disease situations and supports screening operations in various hospital settings.
Critical care areas experience increased demand for this segment because infection risks have risen. The emergency medical department and intensive care unit benefit from faster diagnostic systems because they enhance patient care results.
By End-User :
The hospital system stands as the primary end-user because of its need to treat numerous patients and conduct regular infection tests. Diagnostic laboratories operate extensive testing facilities through their modern equipment and professional testing services. Clinics use sepsis diagnostics to assess patients at their initial stage and make decisions about which patients to refer. Research institutes develop better identification methods and infection pattern research while other end-users provide specialized testing services for healthcare systems.
Healthcare infrastructure development enables more healthcare facilities to implement diagnostic tools. Increasing awareness about infection control practices results in higher testing requirements that extend to all types of end-users.
By Technology :
PCR technology enables rapid genetic material amplification which results in precise identification of infectious pathogens. Biomarker testing identifies specific biological signals linked with sepsis which helps in early diagnosis and risk assessment. Next-generation sequencing delivers comprehensive pathogen data which enables clinicians to study infection origins thus enhancing their ability to handle complicated situations that basic methods cannot resolve.
Healthcare systems increase their technology adoption rate because they need faster diagnostic results and better testing accuracy for their patients. Improved testing accuracy enables hospitals and laboratories to manage infections more effectively through ongoing advancements in testing systems.
What are the Key Use Cases Driving the Japan Sepsis Diagnostics Market?
The primary application of the Japanese Sepsis Diagnostics Market operates through hospitals which need to identify bloodstream infections for their critically ill patients who require immediate treatment. The product delivers maximum market demand because hospitals need rapid molecular tests to decrease their operational costs which arise from extended patient waiting times and enhanced mortality rates.
The medical field now utilizes emergency department triage systems and hospital diagnostic laboratories, which enable doctors to use quick tests that differentiate sepsis from other severe inflammatory diseases. The selected use cases show increasing popularity among tertiary hospitals and large diagnostic laboratories which handle high patient volumes and need to establish efficient operational processes through swift system implementation.
The healthcare sector now uses AI technology to create sepsis risk assessment tools which work together with electronic health record systems and portable diagnostic equipment in both outpatient clinics and remote medical facilities. The applications exhibit developmental progress, showing potential for success because Japan is building its digital healthcare system while implementing programs which target early medical treatment to decrease severe medical cases.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 25.79 Million |
|
Market size value in 2026 |
USD 27.69 Million |
|
Revenue forecast in 2033 |
USD 45.63 Million |
|
Growth rate |
CAGR of 7.40% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Abbott, Roche, Thermo Fisher, Siemens Healthineers, BD, Bio-Rad, Hologic, Qiagen, Danaher, PerkinElmer, Agilent, Sysmex, Cepheid, bioMérieux, Fujirebio. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Molecular Diagnostics, Immunoassays, Blood Culture Tests, Others); By Application (Hospital-acquired Infections, ICU Monitoring, Emergency Care, Clinical Diagnostics, Others); By End-User (Hospitals, Diagnostic Labs, Clinics, Research Institutes, Others); By Technology (PCR, Biomarker Testing, Next-gen Sequencing, Others). |
Which Regions are Driving the Japan Sepsis Diagnostics Market Growth?
The Japan Sepsis Diagnostics Market in Tokyo and the Kanto region because of their advanced tertiary hospital systems and their high density of intensive care units and their robust clinical research facilities. The region enjoys benefits from its early implementation of molecular diagnostic systems which receive support through government healthcare modernization initiatives. Tokyo's major academic medical centers test new sepsis detection technologies which help spread these innovations to nearby prefectures. Established diagnostic supply chains together with the presence of major global in vitro diagnostics companies create an ecosystem that enables medical products to reach the market and undergo clinical testing at a faster pace.
The Kansai region operates as a reliable secondary center because its private healthcare system operates efficiently and its hospitals maintain steady investment activities throughout the year. In Osaka and Kyoto, hospitals implement operational efficiency programs together with standardized clinical protocols, which helps them to adopt advanced sepsis diagnostic tools at a slow but steady pace. The Kanto region exists as an innovation-driven ecosystem, whereas Kansai market demand develops through processes which acquire diagnostic solutions at economical costs and establish long-term relationships with diagnostic companies. The business creates a stable income stream for manufacturers, which leads to gradual technology adoption that occurs at medical facilities across the entire hospital network.
Kyushu stands as the most rapidly developing area because of recent hospital infrastructure improvements and emergency care capacity growth, which particularly benefits Fukuoka. The government-led healthcare digitization programs that started after 2023 have enabled mid-sized hospitals to adopt rapid diagnostic platforms at a faster pace. The expansion of inter-prefectural patient transfer networks has created a greater need for rapid sepsis detection outside of large metropolitan areas. The period between 2026 and 2033 presents Kyushu as a vital expansion path for market newcomers who want to test their products before the complete market penetration begins.
Who are the Key Players in the Japan Sepsis Diagnostics Market and How Do They Compete?
The Japan Sepsis Diagnostics Market shows a moderately consolidated competitive structure, where global in vitro diagnostics leaders dominate hospital contracts while smaller specialized firms compete in niche rapid-testing segments. The current competition focuses on three main areas which include diagnostic speed, hospital information system integration, and accurate pathogen detection capabilities. Established players defend share through long-term hospital partnerships and regulatory approvals, while newer entrants push disruption through rapid molecular platforms and AI-enabled diagnostic interpretation tools that reduce clinical decision time in emergency care settings.
Roche and bioMérieux focus on technology-driven differentiation, strengthening their position through high-throughput molecular diagnostic systems and rapid blood culture automation tailored for Japanese hospital workflows. Thermo Fisher Scientific emphasizes scalable laboratory integration, enabling hospitals to unify sepsis testing with broader infectious disease panels. Siemens Healthineers provides a unique solution through AI-driven clinical decision support systems which combine diagnostic results and patient monitoring information to enhance early sepsis detection in intensive care environments. Abbott expands its footprint by offering portable and near-patient testing solutions that support emergency department adoption and faster triage decisions.
Becton Dickinson strengthens its position through automation in microbiology workflows and standardized culture systems designed for high-volume hospital laboratories. The companies expand their operations through strategic partnerships with Japanese healthcare institutions and co-development contracts for local testing solutions and incremental system improvements instead of major acquisitions. The company uses this approach to maintain its hospital system presence while developing permanent diagnostic platform relationships throughout Japan's critical care facilities.
Company List
- Abbott
- Roche
- Thermo Fisher
- Siemens Healthineers
- BD
- Bio-Rad
- Hologic
- Qiagen
- Danaher
- PerkinElmer
- Agilent
- Sysmex
- Cepheid
- bioMérieux
- Fujirebio
Recent Development News
In October 2025, Sysmex Corporation entered into a strategic partnership with QIAGEN K.K. to distribute molecular diagnostic products for infectious diseases in Japan. The collaboration expands access to advanced molecular testing platforms that are increasingly used in hospital workflows for early sepsis detection and pathogen identification.
Source: https://www.iptonline.com/
In February 2026, Sysmex Corporation entered into a sales collaboration with Fujirebio Holdings for automated immunoassay systems and reagents in Japan and other regions. While primarily focused on neurodegenerative diagnostics, the partnership strengthens Japan’s immunoassay infrastructure, a core technology platform also widely used in sepsis biomarker testing such as inflammatory and infection markers.
Source: https://www.sysmex.co.jp/
What Strategic Insights Define the Future of the Japan Sepsis Diagnostics Market?
The Japan Sepsis Diagnostics Market is structurally moving toward fully integrated, real-time diagnostic ecosystems which enable molecular testing and AI-based risk scoring and hospital electronic records to function as a unified clinical decision layer. Platform-based solutions that shorten sepsis identification time and reduce ICU burden will drive market growth during the next 5 to 7 years more than standalone diagnostic kits. The rising hospital complexity which Japan's aging population brings to the healthcare system forces medical professionals to adopt faster and more accurate methods of detecting infections.
A less visible risk exists because organizations depend too much on automated systems which need stable data inputs and constant access to reagent supply chains. The diagnostic process will face reduced reliability and extended clinical implementation time because any inaccuracies in sensor performance and software compatibility and supply chain issues with imported materials. Hospitals that depend on a single-vendor integrated platform system face operational risks because of their dependence on that system.
Urban emergency response systems in Tokyo and Osaka will gain new potential from portable molecular diagnostics which enable pre-hospital and ambulance-based sepsis screening to expand its reach. Modular diagnostic platforms which use interoperable systems should become the main focus for market participants because these platforms enable easy integration with hospital IT systems and decentralized testing environments while ensuring flexibility for future care delivery.
Japan Sepsis Diagnostics Market Report Segmentation
By Type
- Molecular Diagnostics
- Immunoassays
- Blood Culture Tests
By Application
- Hospital-acquired Infections
- ICU Monitoring
- Emergency Care
- Clinical Diagnostics
By End-User
- Hospitals
- Diagnostic Labs
- Clinics
- Research Institutes
By Technology
- PCR
- Biomarker Testing
- Next-gen Sequencing
Frequently Asked Questions
Find quick answers to common questions.
The Japan Sepsis Diagnostics Market size is USD 45.63 Million in 2033.
Key segments for the Japan Sepsis Diagnostics Market are By Type (Molecular Diagnostics, Immunoassays, Blood Culture Tests, Others); By Application (Hospital-acquired Infections, ICU Monitoring, Emergency Care, Clinical Diagnostics, Others); By End-User (Hospitals, Diagnostic Labs, Clinics, Research Institutes, Others); By Technology (PCR, Biomarker Testing, Next-gen Sequencing, Others).
Major Japan Sepsis Diagnostics Market players are Abbott, Roche, Thermo Fisher, Siemens Healthineers, BD, Bio-Rad, Hologic, Qiagen, Danaher, PerkinElmer, Agilent, Sysmex, Cepheid, bioMérieux, Fujirebio.
The Japan Sepsis Diagnostics Market size is USD 25.79 Million in 2025.
The Japan Sepsis Diagnostics Market CAGR is 7.40% from 2026 to 2033.
- Abbott
- Roche
- Thermo Fisher
- Siemens Healthineers
- BD
- Bio-Rad
- Hologic
- Qiagen
- Danaher
- PerkinElmer
- Agilent
- Sysmex
- Cepheid
- bioMérieux
- Fujirebio
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









