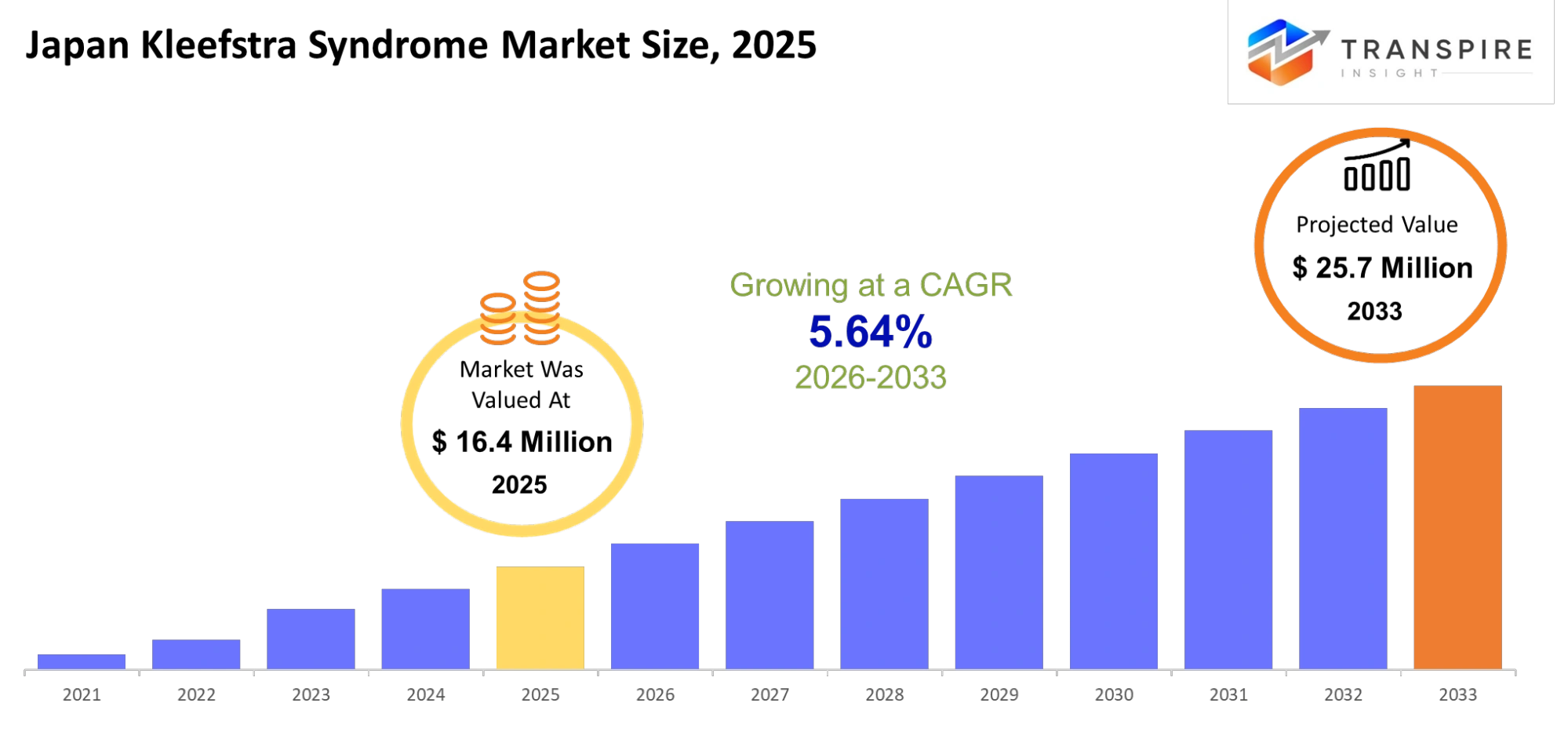
Japan Kleefstra Syndrome Market Size & Forecast:
- Japan Kleefstra Syndrome Market Size 2025: USD 16.4 Million
- Japan Kleefstra Syndrome Market Size 2033: USD 25.7 Million
- Japan Kleefstra Syndrome Market CAGR: 5.64%
- Japan Kleefstra Syndrome Market Segments: By Treatment (Supportive Therapy, Behavioral Therapy, Medication, Genetic Counseling), By Diagnosis (Genetic Testing, Clinical Evaluation, Imaging, Screening), By End-User (Hospitals, Clinics, Research Institutes, Patients)
To learn more about this report, Download Free Sample Report
Japan Kleefstra Syndrome Market Summary:
The Japan Kleefstra Syndrome Market size is estimated at USD 16.4 Million in 2025 and is anticipated to reach USD 25.7 Million by 2033, growing at a CAGR of 5.64% from 2026 to 2033. The Japan Kleefstra Syndrome Market which belongs to the rare genetic disorder healthcare industry will show changes because Japanese medical research now focuses on extremely uncommon neurodevelopmental disorders. Genomic screening will become more essential for clinicians and caregivers throughout upcoming years because registry mapping will improve and policy frameworks will develop whereas Japanese healthcare systems will build toward their objectives of quick diagnosis and custom treatment pathways.
Family expectations will create demand for improved coordination between hospitals and research institutes while Japan's regulatory framework enables thorough assessment of gene therapies and data monitoring technologies. The development of sequencing technology will enhance medical practitioners ability to make specific clinical decisions through direct patient analysis at later points in their career.
What Has the Impact of Artificial Intelligence Been on the Japan Kleefstra Syndrome Market?
The healthcare industry is undergoing major changes as artificial intelligence develops new technologies which are now being deployed to treat specific medical conditions. AI in Japan Kleefstra Syndrome Market is enabling advanced analysis of rare disease datasets which improves the speed and accuracy of genetic testing and patient identification processes.
Artificial intelligence in Japan Kleefstra Syndrome Market uses market analysis tools to identify existing clinical gaps and assist researchers who develop new studies. The Japan Kleefstra Syndrome Market stakeholders use machine learning together with predictive analytics to discover disease progression patterns and treatment options which improve their research accuracy and decision-making process.
The Japanese market for Kleefstra Syndrome treatment undergoes operational changes because smart automation and predictive analytics create new operational possibilities. The Japan Kleefstra Syndrome Market relies on demand forecasting models which help pharmaceutical and research organizations predict treatment needs while they manage their resource distribution. AI-powered supply chain optimization decreases operational expenses while it enhances the delivery process for specialized treatments used to treat uncommon medical conditions.
Artificial intelligence in the Japan Kleefstra Syndrome Market enables personalized medicine approaches to improve patient treatment methods and creates new possibilities for medical research. AI implementation in the Japan Kleefstra Syndrome Market ecosystem enhances competitive strength while it speeds up research processes and increases data trustworthiness.
Key Market Trends & Insights:
- The Japan Kleefstra Syndrome Market is expanding because healthcare institutions now conduct more genetic tests and their understanding of rare diseases has improved.
- The Japan Kleefstra Syndrome Market growth outlook benefits from artificial intelligence integration which produces more accurate diagnoses and detects diseases at earlier stages.
- The Kanto region controls the Japan Kleefstra Syndrome Market because it will possess 55-60% market share in 2025 through its advanced hospital systems.
- The Kansai region experiences rapid growth because research funding will increase during the period from 2024 to 2030.
- The Japan Kleefstra Syndrome Market identifies genetic testing services as its most significant market segment because people increasingly demand accurate diagnostic tests.
- The second highest market segment of therapeutic development solutions maintains its expansion through ongoing clinical pipeline development.
- The Japanese market for Kleefstra Syndrome diagnosis systems shows its fastest growth through AI-powered diagnostic tools which use machine learning integration.
- The Japan Kleefstra Syndrome Market shows its primary market share through diagnostic applications which maintain a 45 to 50 percent market share and support early disease detection efforts.
- The research institutes receive funding support which enables them to expand their genomic sequencing programs, which makes them the fastest growing end-user market segment.
- The main companies that determine market trends in Japan for Kleefstra Syndrome include Roche, Pfizer, Novartis, Takeda, Astellas Pharma, and Illumina.
Japan Kleefstra Syndrome Market Segmentation
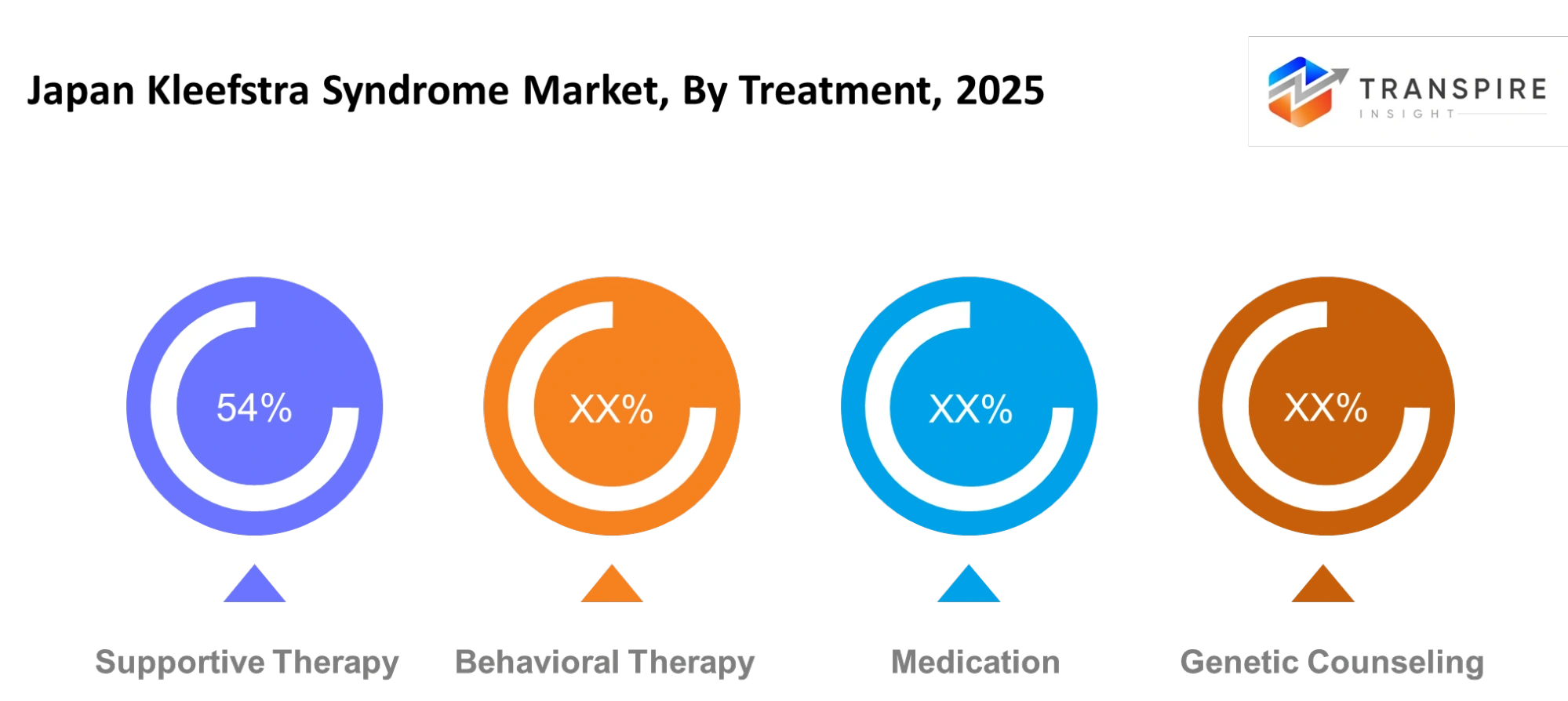
By Treatment :
The therapy treatment method uses daily assistance to treat Kleefstra Syndrome symptoms which affect the patient's daily life. The care plan uses physical therapy and speech therapy and daily living support to assist the patient. The method seeks to enhance comfort and enable basic daily activities for the individual. The therapy method helps patients develop better communication abilities while decreasing their problematic conduct.
The doctor can prescribe medication to treat sleep disorders and mood fluctuations and associated symptoms. Genetic counseling enables families to learn about the condition and its inheritance patterns and how to plan for the future. The different treatments provide various methods to support the patient requirements of long-term medical care.
To learn more about this report, Download Free Sample Report
By Diagnosis :
Kleefstra Syndrome confirmation through genetic testing shows partial success because it detects specific gene mutations. The test delivers medical results when doctors face difficulties diagnosing their patients. The clinical evaluation process requires assessment of the patient's growth and conduct together with the medical signs that show their current state. The research uses imaging techniques to examine brain or structural differences which become necessary for specific cases.
Early detection of developmental delays occurs through screening procedures that medical professionals implement during initial patient evaluations. The combination of these testing techniques enables healthcare professionals to identify conditions early while establishing accurate diagnoses which lead to immediate treatment plans and improved patient outcomes.
By End-User :
Diagnostic and treatment services together with specialist care and advanced testing capabilities operate as the primary medical facilities of hospitals. Clinics offer their patients both routine medical assessments and therapeutic treatments and continuous medical assistance. These facilities help manage long-term care needs for individuals with Kleefstra Syndrome.
Research institutes focus on studying genetic causes and improving diagnostic methods. Patients and their families directly participate in treatment administration through their home support activities and therapy programs. Each end-user contributes to better understanding, diagnosis, and long-term care support for the condition.
What are the Main Challenges for the Japan Kleefstra Syndrome Market Growth?
The Japan Kleefstra Syndrome Market is facing multiple technical and operational difficulties which impede its entire growth process. The Japan Kleefstra Syndrome Market diagnosis process faces major difficulties because rare diseases require multiple medical tests which become impossible when doctors have access to only limited patient information and incomplete genomic databases.
The healthcare system experiences workflow disruptions because advanced AI-based diagnostic systems cannot effectively work with current medical systems. The Japan Kleefstra Syndrome Market faces multiple challenges that prevent research centers from establishing standardized methods which operate in various medical settings.
The Japan Kleefstra Syndrome Market must deal with expensive product development and market entry costs which arise from strict regulatory frameworks that govern its manufacturing and distribution operations. The implementation of strict healthcare regulations which include both clinical validation requirements and quality assurance standards increases the time required to introduce new treatments into the market.
The ultra-rare nature of Kleefstra syndrome creates limited commercial potential for the Japan Kleefstra Syndrome Market which results in decreased funding from investors at major businesses. The companies face challenges in achieving equilibrium between their research and development costs and the anticipated financial benefits from their projects.
The Japan Kleefstra Syndrome Market faces major adoption obstacles because healthcare professionals lack sufficient understanding of the condition and small medical facilities lack adequate specialized treatment centers. The effective use of advanced diagnostic solutions remains limited because there is a lack of trained genetic experts and data science professionals. The Japan Kleefstra Syndrome Market experiences funding shortages which hinder early-stage research and impact the development of new clinical programs.
Country Insights
The market for Japan Kleefstra Syndrome shows gradual progress through increased understanding of rare genetic disorders and their treatment methods. The three factors of clinical awareness combined with diagnostic access and supportive healthcare systems create progress within this medical area. The healthcare system in Japan dedicates its resources to discovering diseases at their earliest stage while establishing organized patient care methods and advancing research capabilities through its genetic research facilities.
Clinical diagnosis and identification framework: The evolution of clinical diagnosis in Japan advances through two major improvements which include expanded genetic testing options and better hospital-based knowledge. Medical institutions focus on recognizing developmental delay patterns and linking them with rare genetic markers. Pediatric and neurology departments now conduct more screening tests which helps them discover and accurately identify Kleefstra syndrome patients at an earlier stage.
The healthcare system in Japan uses its existing medical facilities to create dedicated medical routes which help patients with rare genetic disorders. Multidisciplinary care approaches assist in long-term management of developmental and cognitive challenges. Government-supported healthcare coverage strengthens accessibility to consultations, therapies, and continuous monitoring services for affected individuals and families.
Recent Development News
In April 2026, Nature Genetics published a correction related to a neurodevelopmental disorder gene discovery study involving pathways overlapping Kleefstra syndrome biology. The correction refined genetic findings associated with recessive neurodevelopmental disorders that are clinically relevant to EHMT1-linked conditions such as Kleefstra syndrome.Source https://www.nature.com
In March 2026, international clinical research groups continued updating evidence-based guidelines for Kleefstra syndrome management used by Japanese pediatric neurology centers. The updated guideline framework strengthened standardized diagnosis and long-term care practices for EHMT1-related disorders across global and Japanese clinical settings.Source https://www.kleefstrasyndrome.org
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 16.4 Million |
|
Market size value in 2026 |
USD 17.5 Million |
|
Revenue forecast in 2033 |
USD 25.7 Million |
|
Growth rate |
CAGR of 5.64% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Pfizer, Novartis, Roche, Takeda, Astellas, Daiichi Sankyo, Otsuka, Sanofi, GSK, AstraZeneca, Eli Lilly, AbbVie, Teva, Bristol Myers Squibb, Bayer |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Treatment (Supportive Therapy, Behavioral Therapy, Medication, Genetic Counseling), By Diagnosis (Genetic Testing, Clinical Evaluation, Imaging, Screening), By End-User (Hospitals, Clinics, Research Institutes, Patients) |
How Can New Companies Establish a Strong Foothold in the Japan Kleefstra Syndrome Market?
New entrants can establish a strong position in the Japan Kleefstra Syndrome Market by focusing on niche specialization and precision-driven innovation. The Japan Kleefstra Syndrome Market needs continuous research work because companies that develop advanced genomic diagnostic methods and AI-based screening technologies and create patient-specific data models will achieve competitive benefits. Artificial intelligence integration in Japan Kleefstra Syndrome Market solutions helps new firms differentiate by improving diagnostic speed and accuracy while addressing critical gaps in rare disease identification.
Strategic partnerships serve as important entry routes into the Japan Kleefstra Syndrome Market because businesses can establish partnerships with hospitals and genetic research institutes and biotech accelerators to overcome entry obstacles. Startups of Genomix Care Japan and RareGene Analytics demonstrate how AI-powered sequencing platforms and cloud-based genomic analysis tools can enhance early diagnosis efficiency. These innovation strategies in the Japan Kleefstra Syndrome Market allow new companies to build credibility while leveraging established clinical networks for faster adoption.
The Japanese Kleefstra Syndrome Market requires companies to create smart automation systems together with adaptable data infrastructure systems for their long-term market presence. The solution to healthcare issues that cause slow diagnosis and create separate patient records while showing insufficient knowledge about rare diseases will result in substantial business value. Organizations that develop predictive analytics solutions with machine learning capabilities for medical workflows will achieve better success in their business operations within the Japanese Kleefstra Syndrome Market.
The Japan Kleefstra Syndrome Market requires companies to develop unique products through their ability to meet regulations while creating affordable new solutions. Organizations that establish compliance with rules from the beginning while using flexible pricing strategies will achieve better success in navigating market challenges. Through their combination of technology leadership and strategic partnerships and their focus on patient-oriented innovations, new companies can establish a strong presence in the Japanese Kleefstra Syndrome Market.
Key Japan Kleefstra Syndrome Market Company Insights
The genetic disorder Kleefstra syndrome shows low occurrence rates in Japan although more people are being diagnosed with the condition because of increased awareness. The implementation of genetic testing has become more widespread in both hospitals and research institutions. The organization provides support to people with disabilities while operating rare disease support programs which are currently in the process of expanding their services. The market experiences growth because of development in healthcare systems and implementation of early detection programs within pediatric healthcare facilities and genetic testing services.
The market advances through the development of genomic sequencing technologies and the expansion of newborn screening programs and the enhancement of clinical genetics education programs. Government support for rare disease identification programs enhances detection rates. Hospitals are increasing their spending on molecular diagnostics which leads to better treatment planning and patient evaluation in pediatric neurology and genetic counseling throughout the country while creating better diagnostic pathways.
The high costs required for diagnosis together with the lack of knowledge in primary care facilities and the slow process of diagnosis creation make it difficult for patients to receive treatment at the right time. The small patient pool limits large-scale clinical studies. The different ways patients show their symptoms make it hard to create standardized procedures for diagnosis. The existence of healthcare facility access disparities between urban and rural areas creates challenges for consistent disease management which in turn leads to unequal medical treatment across different regions.
The competitive market receives its supply from genetic testing laboratories and sequencing technology providers and specialized rare disease clinics. The hospitals establish strategic partnerships with biotech companies to achieve their research objectives. The healthcare sector in Japan develops its clinical laboratories through two main areas of focus which include better diagnostic methods and quick sequencing technology and expanded testing capabilities for rare diseases.
Company List
- Pfizer
- Novartis
- Roche
- Takeda
- Astellas
- Daiichi Sankyo
- Otsuka
- Sanofi
- GSK
- AstraZeneca
- Eli Lilly
- AbbVie
- Teva
- Bristol Myers Squibb
- Bayer
What are the Key Use-Cases Driving the Growth of Japan Kleefstra Syndrome Market?
The market for Kleefstra Syndrome in Japan operates through diagnostic improvements and genetic sequencing applications which help detect rare neurological disorders at earlier stages. The hospital laboratories in Japan's Kleefstra Syndrome Market now use next-generation sequencing (NGS) technology and artificial intelligence-based genomic analysis tools to study EHMT1 gene mutations which cause Kleefstra syndrome.
The Japan Kleefstra Syndrome Market uses precision medicine as its main application, as pharmaceutical and biotech companies develop targeted treatments using patient-specific genetic information. Researchers use artificial intelligence in Japan Kleefstra Syndrome Market operations to discover how diseases progress and enhance their drug development processes. The application of predictive analytics in clinical trial optimization has produced better patient stratification results which enable researchers to conduct studies at lower costs while achieving greater efficiency.
The Japan Kleefstra Syndrome Market depends on two key components which are healthcare data integration and rare disease registries. The centralized databases help hospitals and research institutes to monitor patient outcomes which results in better disease tracking and streamlined treatment processes.
The Japan Kleefstra Syndrome Market receives advanced accessibility through emerging use-cases which include AI-driven telemedicine and remote genetic counseling. The innovations enable more patients to receive diagnostic testing especially in areas which have few available specialists. The Japan Kleefstra Syndrome Market will experience substantial growth through digital health adoption because scalable applications will drive its expansion.
Japan Kleefstra Syndrome Market Report Segmentation
By Treatment
- Supportive Therapy
- Behavioral Therapy
- Medication
- Genetic Counseling
By Diagnosis
- Genetic Testing
- Clinical Evaluation
- Imaging
- Screening
By End-User
- Hospitals
- Clinics
- Research Institutes
- Patients
Frequently Asked Questions
Find quick answers to common questions.
The approximate Japan Kleefstra Syndrome Market size for the market will be USD 25.7 Million in 2033.
Key segments for the Japan Kleefstra Syndrome Market are By Treatment (Supportive Therapy, Behavioral Therapy, Medication, Genetic Counseling), By Diagnosis (Genetic Testing, Clinical Evaluation, Imaging, Screening), By End-User (Hospitals, Clinics, Research Institutes, Patients).
Major Japan Kleefstra Syndrome Market players are Pfizer, Novartis, Roche, Takeda, Astellas, Daiichi Sankyo, Otsuka, Sanofi, GSK, AstraZeneca, Eli Lilly, AbbVie, Teva, Bristol Myers Squibb, Bayer.
The Japan Kleefstra Syndrome Market size is USD 16.4 Million in 2025.
The Japan Kleefstra Syndrome Market CAGR is 5.64%.
- Pfizer
- Novartis
- Roche
- Takeda
- Astellas
- Daiichi Sankyo
- Otsuka
- Sanofi
- GSK
- AstraZeneca
- Eli Lilly
- AbbVie
- Teva
- Bristol Myers Squibb
- Bayer
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









