France Pharmaceutical CRO Market Size & Forecast
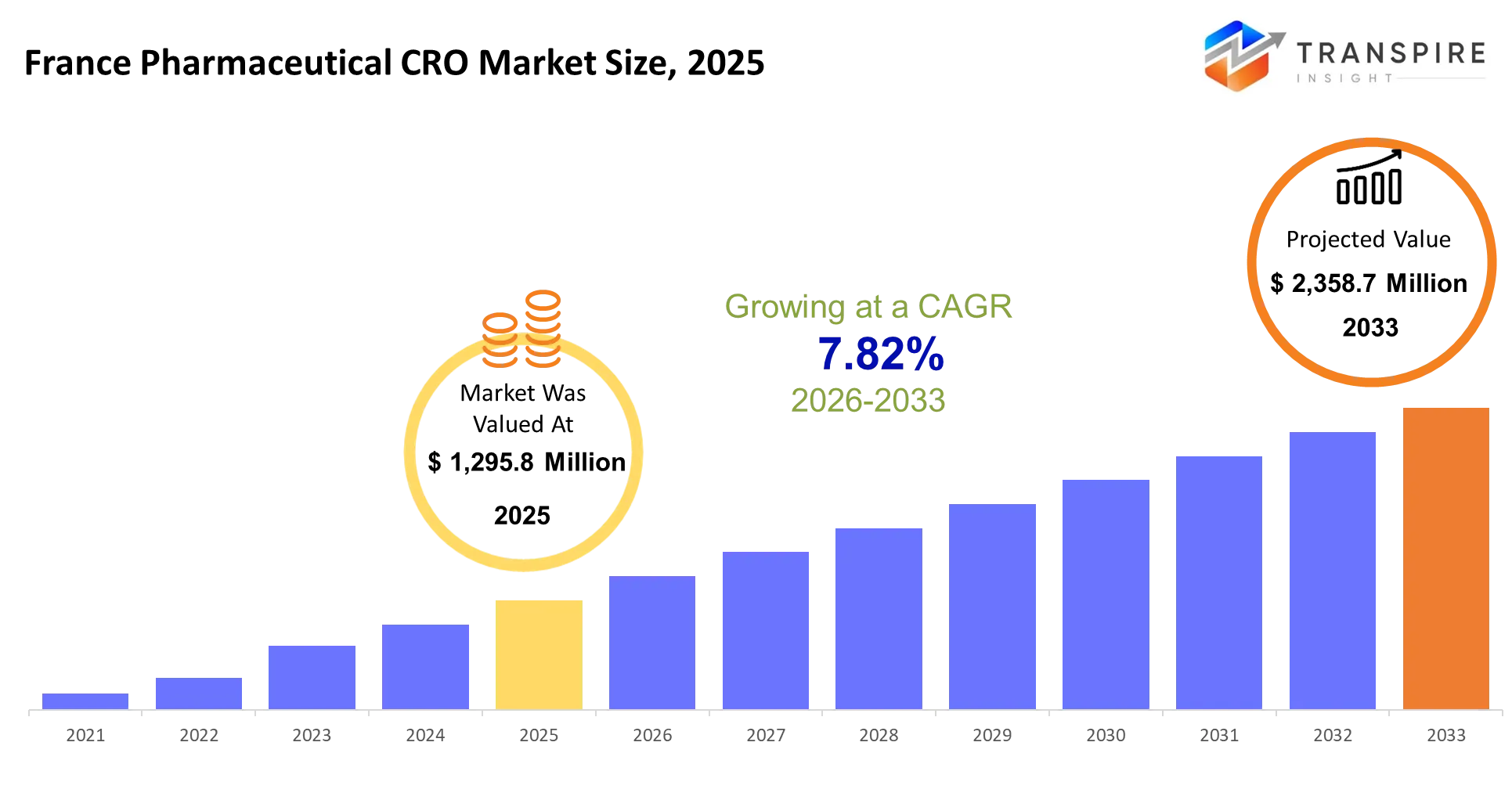
- France Pharmaceutical CRO Market Size 2025: USD 1,295.8 Million
- France Pharmaceutical CRO Market Size 2033: USD 2,358.7 Million
- France Pharmaceutical CRO Market CAGR: 7.82%
- France Pharmaceutical CRO Market Segments: By Type (Clinical Research, Preclinical Research, Laboratory Services, Others); By Application (Drug Development, Clinical Trials, Toxicology Studies, Data Management, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Phase (Phase I, Phase II, Phase III, Phase IV, Others)
To learn more about this report, Download Free Sample Report
France Pharmaceutical CRO Market Summary
The France pharmaceutical CRO market was valued at USD 1,295.8 million in 2025. It is expected to reach USD 2,358.7 million by 2033, growing at a CAGR of 7.82%. Contract research organizations provide outsourced research services to pharmaceutical and biotechnology companies, handling everything from preclinical studies to Phase IV post-market surveillance. Clinical research services account for 65% of the French market because sponsors prefer transferring complex trial management to specialized vendors rather than maintaining in-house capabilities across multiple therapeutic areas and regulatory jurisdictions.
Three regulatory changes between 2020 and 2025 reshaped how French CROs operate. First, the EU Clinical Trials Regulation introduced harmonized approval processes across member states, which reduced timeline variability but increased upfront documentation requirements. Second, France's national health data platform opened access to real-world evidence datasets that sponsors can now link to clinical trial outcomes, creating demand for CROs with bioinformatics capabilities. Third, ANSM began accepting decentralized trial protocols that allow remote patient monitoring, which shifted investigator site selection away from traditional hospital networks toward home health providers and telemedicine platforms.
Current market expansion is driven by biotech companies externalizing late-stage development work they previously handled internally. Small and mid-size biotech firms lack the regulatory expertise to navigate EMA submissions and the site network to execute multi-country Phase III trials efficiently. CROs offer these companies access to established investigator relationships, therapeutic area expertise, and regulatory affairs specialists on a project basis rather than as permanent headcount. This business model allows biotech sponsors to preserve cash while accelerating development timelines, which matters when venture funding cycles require demonstrable clinical progress every 18 to 24 months.
Key France Pharmaceutical CRO Market Insights
- Clinical research services dominate the France pharmaceutical CRO market with a 65% share in 2024, driven by increasing externalization of Phase II and Phase III trial management.
- Preclinical research services account for 22% of the market in 2024, supporting early-stage drug candidates that require toxicology testing and pharmacokinetic studies.
- Drug development applications represent 41.3% of CRO service demand in 2024, as pharmaceutical companies outsource formulation development and stability testing.
- Clinical trials applications hold 38.7% market share in 2024, concentrated in oncology, immunology, and rare disease therapeutic areas where patient recruitment is challenging.
- Pharmaceutical companies constitute 52.8% of end-user demand in 2024, outsourcing non-core clinical development activities to focus internal resources on discovery and commercialization.
- Biotech firms account for 31.4% of the market in 2024 and represent the fastest-growing end-user segment, expanding at 9.1% CAGR through 2033.
- Phase III trials generate 43.2% of clinical research revenue in 2024, requiring large patient populations and extended monitoring periods that favor full-service CROs.
- Phase II trials contribute 28.6% of revenue in 2024, with increasing demand for adaptive trial designs that require specialized statistical and data management capabilities.
- IQVIA holds approximately 15-18% of the French CRO market in 2025, leveraging its integrated data analytics and clinical trial services across multiple therapeutic areas.
- Labcorp expanded its French clinical development operations by 20% in 2024, adding investigator sites and therapeutic area specialists to serve growing biotech demand.
What are the Key Drivers, Restraints, and Opportunities in the France Pharmaceutical CRO Market?
The primary growth driver is pharmaceutical companies shifting from selective outsourcing to strategic partnerships where CROs manage entire development programs rather than individual studies. Large pharma firms now award multi-year contracts covering preclinical through Phase III work for specific therapeutic areas, which provides CROs with revenue visibility and allows them to invest in specialized capabilities like rare disease patient registries or oncology biomarker platforms. IQVIA, Syneos Health, and Parexel have all structured their French operations around these strategic partnerships, dedicating teams to specific sponsor accounts rather than operating as transactional service providers. The financial impact is substantial: strategic partnership contracts typically run three to five years and cover development portfolios worth hundreds of millions of euros, compared to single-study contracts that last 18 to 36 months.
The most significant restraint is investigator site capacity constraints in high-demand therapeutic areas. French oncology centers and immunology specialists are oversubscribed with trial protocols, which creates patient recruitment bottlenecks that delay study timelines regardless of how efficiently the CRO manages other aspects of trial execution. CROs cannot simply open new sites to resolve this problem because qualified investigators require years of training and regulatory certification. The constraint is particularly acute in rare disease trials where France might have only five to ten centers with relevant patient populations, and those centers are already committed to multiple competing protocols. This forces CROs to extend recruitment timelines or expand into less experienced investigator sites, either of which increases costs and risks to study quality.
The clearest opportunity lies in real-world evidence services that connect clinical trial data to France's national health database. ANSM now accepts real-world evidence to support regulatory submissions for certain indications, and sponsors need CROs that can design hybrid studies combining traditional randomized controlled trial methodology with observational data from electronic health records. This requires capabilities most CROs do not currently possess: data scientists who understand healthcare analytics, regulatory specialists who know how to structure real-world evidence submissions, and established relationships with French health data authorities. CROs that build these capabilities will capture high-margin work that cannot be easily replicated by competitors lacking the technical infrastructure and regulatory expertise to execute these hybrid study designs.
What Has the Impact of Artificial Intelligence Been on the France Pharmaceutical CRO Market?
AI-powered patient identification systems now screen electronic health records to find candidates who meet specific trial inclusion criteria, which accelerates recruitment timelines by identifying eligible patients before sites manually review charts. IQVIA and Labcorp have deployed machine learning models in French healthcare systems that flag patients based on diagnosis codes, lab values, medication histories, and other clinical markers. These systems reduced median screening time from weeks to days at sites with integrated electronic health records, though adoption remains limited to larger hospital networks with digitized patient data. The operational benefit is most visible in rare disease trials where manual chart review previously required months of investigator time to identify even a handful of eligible patients.
Natural language processing automates safety monitoring by extracting adverse events from clinical notes, lab reports, and patient diaries without requiring manual data entry by site coordinators. Syneos Health and ICON have implemented NLP systems that continuously scan trial documentation for safety signals, flagging potential serious adverse events that require immediate reporting to regulatory authorities. This automation reduced median adverse event reporting time from 48-72 hours to under 24 hours at sites using the technology, which matters when regulatory requirements mandate reporting serious adverse events within 24 hours of investigator awareness. The limitation is that NLP accuracy depends on documentation quality, and French clinical notes often include medical shorthand and non-standardized terminology that reduce automated extraction reliability.
The primary barrier to broader AI adoption is data fragmentation across French healthcare providers. Patient records remain siloed within individual hospital systems, regional health networks, and private practices, which prevents CROs from training AI models on comprehensive datasets that reflect real-world patient populations. Additionally, French data protection regulations require explicit patient consent for secondary use of health data in research contexts, which adds compliance overhead that many CROs find prohibitive for AI development projects. This creates a paradox where the most valuable AI applications require large, diverse datasets that French regulatory frameworks make difficult to assemble legally.
Key Market Trends
- Pharmaceutical sponsors shifted from transactional service purchasing to multi-year strategic partnerships between 2022 and 2024, consolidating CRO relationships from five or six vendors to two or three preferred providers.
- Decentralized trial adoption accelerated in 2023 when ANSM published guidance accepting remote patient monitoring and home health visits as valid alternatives to site-based assessments.
- Biotech companies increased CRO outsourcing from an average 60% of development spend in 2021 to over 75% by 2024, driven by venture capital pressure to minimize fixed costs.
- IQVIA and Syneos Health expanded their French therapeutic area specialists by 25-30% between 2023 and 2025, hiring oncology and rare disease experts to support strategic partnership contracts.
- Real-world evidence services emerged as a distinct offering in 2024, with CROs establishing dedicated teams to design hybrid studies combining trial data and observational health records.
- Patient recruitment timelines for oncology trials increased from 12 months to 16-18 months between 2022 and 2024 due to investigator site capacity constraints and competing protocols.
- Chinese CROs entered the French market in 2024, offering Phase I and early Phase II services at 20-25% below incumbent pricing to establish European operations.
- Data management services grew faster than clinical monitoring between 2023 and 2025, reflecting increased sponsor demand for advanced analytics and real-time data visualization during trials.
France Pharmaceutical CRO Market Segmentation
By Type
Clinical research services dominate the French CRO market at 65% of total revenue. This segment covers patient recruitment, site monitoring, data collection, and regulatory submission support for Phase I through Phase IV clinical trials. The dominance reflects a structural shift where pharmaceutical and biotech sponsors externalize trial management to CROs rather than maintaining internal clinical operations teams. Full-service CROs like IQVIA, Labcorp, and Syneos Health operate as the sponsor's clinical development arm, managing investigator relationships, regulatory interactions, and data quality oversight across multiple concurrent trials. The value proposition is risk transfer: sponsors pay CROs to deliver enrolled patients and clean data on schedule, while the CRO absorbs the operational complexity and timeline variability.
Preclinical research services account for 22% of the market, encompassing toxicology studies, pharmacokinetic analysis, and formulation development that occur before human trials. This segment serves early-stage biotech companies and pharmaceutical discovery teams that need specialized laboratory capabilities without building dedicated facilities. Charles River, Covance, and Eurofins dominate French preclinical services through ownership of GLP-certified laboratories equipped for regulatory toxicology studies. The segment is technically demanding because preclinical studies must meet strict regulatory standards for data quality and reproducibility, which requires validated analytical methods and experienced laboratory scientists. Margins are higher than clinical research because switching costs are substantial once a sponsor qualifies a specific laboratory for a development program.
Laboratory services and other specialized offerings together represent 13% of the market. This includes bioanalytical testing, central laboratory services for clinical trials, and niche capabilities like medical device testing or digital health validation. The segment grew faster than core clinical research between 2023 and 2025 because sponsors increasingly require integrated services that combine clinical trial management with specialized laboratory analysis. For example, oncology trials often need companion diagnostic development alongside drug testing, which requires CROs that can coordinate biomarker validation, clinical sample collection, and diagnostic assay development as a unified program. CROs that offer these integrated services capture higher per-project revenue than those providing only clinical trial management.
To learn more about this report, Download Free Sample Report
By Application
Drug development applications lead at 41.3% of market demand, covering formulation development, stability testing, analytical method development, and manufacturing support services. Pharmaceutical companies outsource these activities to CROs when internal development functions lack capacity or specialized expertise for specific formulation challenges. For instance, developing stable formulations for biologics requires specialized equipment and expertise that most sponsors do not maintain in-house. CROs like Catalent, Patheon, and Lonza provide these capabilities on a contract basis, allowing sponsors to advance candidates through development without capital investment in formulation laboratories. The application is margin-accretive because formulation development work is technically complex and requires experienced scientists who command premium billing rates.
Clinical trials applications account for 38.7% of demand, focused specifically on patient enrollment, site management, and trial execution services rather than broader development support. This segment differs from clinical research services in that it captures only the trial execution component, excluding regulatory strategy, data analysis, and post-trial activities. The application grew between 2022 and 2024 as biotech companies sought CROs that could handle patient recruitment and site monitoring while retaining other development activities in-house. This unbundling allowed sponsors to externalize the most operationally complex and timeline-sensitive aspects of clinical development while preserving control over strategic decisions like endpoint selection and trial design.
Toxicology studies, data management, and other applications together represent 20% of the market. Toxicology studies are mandatory for regulatory submissions and require GLP-certified facilities that most sponsors do not operate internally. Data management services have grown rapidly as trials generate increasing volumes of electronic data from wearables, remote monitoring devices, and patient-reported outcomes that require specialized database platforms and statistical analysis capabilities. CROs that invested in cloud-based data platforms and real-time analytics between 2020 and 2025 captured growing sponsor demand for transparent trial monitoring and adaptive trial designs that require continuous data analysis rather than traditional locked databases reviewed only at study completion.
By End-User
Pharmaceutical companies constitute 52.8% of CRO demand in France. Large pharma firms outsource specific development programs where internal capacity is constrained or therapeutic area expertise is lacking. For example, a company with strong oncology capabilities might externalize rare disease development to CROs with established patient registries and investigator networks in those indications. The outsourcing decision is economically driven: maintaining in-house clinical operations for therapeutic areas where the company runs only one or two trials per year is cost-prohibitive compared to engaging CROs on a project basis. This creates stable demand for CROs in therapeutic areas where sponsor interest is sustained but individual company portfolios are limited.
Biotech firms account for 31.4% of the market and represent the fastest-growing segment at 9.1% CAGR through 2033. These companies typically lack internal clinical development capabilities and externalize the majority of development work from preclinical studies through Phase III trials. The growth driver is venture capital funding dynamics: biotech companies are expected to demonstrate clinical progress quickly while minimizing cash burn, which makes outsourcing to CROs more financially attractive than building internal capabilities that require fixed headcount. CROs provide biotech sponsors with flexible capacity that scales with program needs, billing only for active projects rather than requiring ongoing retainer payments or salaried staff.
Research institutes and other end users contribute 15.8% of demand. Academic medical centers and government research organizations engage CROs for investigator-initiated trials and public health studies that require regulatory-grade data collection and monitoring. These customers typically operate on grant funding with fixed budgets and defined timelines, which requires CROs to deliver services within rigid cost constraints. The segment offers lower margins than commercial pharmaceutical work but provides CROs with access to novel therapeutic approaches and rare disease patient populations that later translate into commercial development opportunities when academic discoveries move toward regulatory approval and commercialization.
By Phase
Phase III trials generate 43.2% of clinical research revenue due to their large patient populations, extended monitoring periods, and complex regulatory requirements. A typical Phase III oncology trial in France might enroll 300-500 patients across 25-40 investigator sites over three to four years, requiring site monitoring, data management, safety reporting, and regulatory coordination throughout the study period. CROs provide value by managing this operational complexity while allowing sponsors to focus on strategic decisions around trial design and regulatory submissions. The revenue concentration in Phase III reflects both the direct cost of large-scale trial execution and the risk premium sponsors pay for CROs to deliver on aggressive enrollment timelines that determine product launch dates.
Phase II trials contribute 28.6% of revenue, focused on dose-finding studies and initial efficacy assessments in smaller patient populations. These trials increasingly use adaptive designs that modify enrollment criteria or dosing schedules based on interim results, which requires CROs with strong statistical and data management capabilities. The technical complexity of adaptive trials creates barriers to entry for smaller CROs that lack specialized expertise, allowing larger full-service providers to command premium pricing. Phase II work is strategically important for CROs because sponsors that successfully complete Phase II typically continue with the same CRO into Phase III, creating multi-year revenue pipelines from early-stage engagement.
Phase I, Phase IV, and other trial phases together account for 28.2% of revenue. Phase I trials are relatively small but technically demanding, requiring specialized facilities for first-in-human safety studies and intensive patient monitoring. Phase IV post-marketing studies are growing as regulators increasingly require real-world evidence to supplement pre-approval clinical data. CROs that offer both traditional Phase IV trial management and real-world evidence capabilities combining trial data with observational health records are capturing growing sponsor demand for integrated post-marketing surveillance programs that satisfy regulatory requirements while generating evidence for payer reimbursement negotiations and clinical guideline development.
What are the Key Use Cases Driving the France Pharmaceutical CRO Market?
The primary application is full-service Phase III trial management for biotech companies that lack internal clinical operations capabilities. These sponsors engage CROs to handle all aspects of trial execution from site selection through database lock, regulatory submission, and post-approval safety monitoring. A typical engagement might involve a rare disease indication where the biotech sponsor has completed Phase II proof-of-concept studies and needs a CRO to execute the pivotal Phase III program required for EMA approval. The CRO provides investigator site networks, regulatory expertise, patient recruitment strategies, and data management infrastructure on a turnkey basis, allowing the biotech company to advance toward commercialization without building permanent clinical operations headcount.
Adjacent demand comes from large pharmaceutical companies outsourcing therapeutic area-specific development programs. For example, a company with limited immunology expertise might engage a CRO to manage all clinical development for an acquired immunology asset, leveraging the CRO's established relationships with French rheumatology and dermatology investigators. These strategic partnerships typically span multiple trials across several years, providing the CRO with predictable revenue while giving the sponsor access to therapeutic area expertise without permanent staff expansion. The commercial logic is specialization: CROs that focus on specific therapeutic areas develop deep investigator relationships and protocol design experience that generalist internal development teams cannot replicate.
Emerging use cases center on hybrid trial designs combining traditional randomized controlled trials with real-world evidence from French health databases. Sponsors engage CROs to design studies where the control arm uses matched patients from electronic health records rather than enrolling a separate placebo cohort, which reduces trial costs and accelerates enrollment timelines. IQVIA and Syneos Health have established dedicated teams to execute these hybrid studies, requiring data scientists, epidemiologists, and regulatory specialists who understand both clinical trial methodology and real-world evidence standards. The application is technically demanding but offers sponsors substantial cost savings, with some hybrid designs reducing Phase III costs by 30-40% compared to traditional fully enrolled trials.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1,295.8 Million |
|
Market size value in 2026 |
USD 1,392.6 Million |
|
Revenue forecast in 2033 |
USD 2,358.7 Million |
|
Growth rate |
CAGR of 7.82% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key companies profiled |
IQVIA, Labcorp, Syneos Health, Parexel, ICON plc, Charles River, Medpace, PRA Health Sciences, Covance, Eurofins, WuXi AppTec, SGS, PPD, Clinipace, Veristat |
|
Customization scope |
Free report customization (regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Clinical Research, Preclinical Research, Laboratory Services, Others); By Application (Drug Development, Clinical Trials, Toxicology Studies, Data Management, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Phase (Phase I, Phase II, Phase III, Phase IV, Others) |
What is Driving Growth in the France Pharmaceutical CRO Market?
France offers CROs three structural advantages that other European markets cannot replicate at equivalent scale. First, the country operates a centralized healthcare system with comprehensive electronic health records covering over 65 million citizens, providing CROs with patient identification capabilities and real-world evidence access that fragmented healthcare systems cannot match. Second, France maintains dense concentrations of specialized investigator sites in Paris, Lyon, and Marseille where academic medical centers, teaching hospitals, and research institutes cluster within geographic proximity, which simplifies trial logistics and reduces monitoring costs compared to geographically dispersed site networks. Third, ANSM has established predictable regulatory review timelines and clear guidance on emerging trial methodologies like decentralized protocols and real-world evidence, which reduces uncertainty for sponsors planning multi-year development programs.
The competitive dynamic in France differs from other European markets because language and regulatory requirements create natural barriers to entry for non-EU CROs. Trial protocols, informed consent documents, and regulatory submissions must be in French, which requires dedicated local staff rather than centralized European teams operating in English. Additionally, French data protection regulations impose specific requirements on health data handling that differ from general GDPR provisions, requiring CROs to maintain France-specific compliance programs. These barriers protect established players like IQVIA, Labcorp, and Syneos Health who have operated in France for decades and invested in local infrastructure, while creating higher entry costs for new competitors attempting to establish French operations.
The fastest-growing demand segment is rare disease trials where France's national health database enables rapid patient identification for ultra-rare indications. In conditions affecting fewer than 1,000 French patients, traditional site-based recruitment can take years because investigators must manually screen medical records to identify eligible candidates. CROs that have established data access agreements with French health authorities can screen the entire national patient population electronically, identifying eligible patients in weeks rather than months. This capability has made France a preferred location for European rare disease trials, with sponsors increasingly choosing French sites as lead countries for multinational studies in ultra-rare indications where patient identification speed determines overall program timelines.
Who are the Key Players in the France Pharmaceutical CRO Market and How Do They Compete?
The French CRO market operates as a two-tier structure. IQVIA, Labcorp, Syneos Health, Parexel, and ICON collectively hold approximately 55-60% market share and compete as full-service providers offering integrated capabilities from preclinical through Phase IV. These companies compete on therapeutic area expertise, investigator site networks, and technology platforms that provide sponsors with real-time trial visibility. Mid-tier and smaller CROs occupy specialized niches based on therapeutic focus, service type, or regional presence, typically partnering with larger CROs on complex programs rather than competing directly for full-service contracts.
IQVIA competes through its integrated data and clinical services platform that combines trial management with real-world evidence capabilities. The company operates France's largest commercial health database covering prescription data, hospital discharge records, and claims information, which it uses for patient identification, site selection, and feasibility assessments. IQVIA expanded its French clinical operations by 25% between 2023 and 2025, adding therapeutic area specialists in oncology, rare diseases, and immunology to support strategic partnership contracts with large pharma sponsors. The company differentiates on analytics capabilities, providing sponsors with predictive enrollment models and comparative effectiveness analyses that smaller CROs cannot replicate without equivalent data assets.
Labcorp competes through vertical integration of laboratory services and clinical trial management, allowing the company to offer sponsors unified programs that combine trial execution with central laboratory testing, biomarker analysis, and companion diagnostic development. This integration matters in oncology and rare disease trials where biomarker-driven patient selection and pharmacodynamic assessments require coordination between clinical sites and specialized laboratories. Labcorp operates GLP-certified laboratories in France that serve both clinical trial sponsors and routine diagnostic markets, creating economies of scale that pure-play CROs cannot match. The company targets biotech sponsors developing precision medicine candidates where integrated laboratory and clinical services reduce coordination overhead and accelerate development timelines compared to engaging separate vendors for trial management and laboratory testing.
Recent Developments
In March 2026, IQVIA announced a strategic partnership with a French rare disease patient registry to provide genetic testing and natural history data for ultra-rare neurological conditions. The collaboration aims to accelerate patient identification and trial feasibility assessments for sponsors developing therapies for conditions affecting fewer than 500 French patients. https://www.iqvia.com (IQVIA)
In February 2026, Labcorp expanded its French central laboratory operations with new biomarker testing capabilities for cell and gene therapy trials. The facility processes pharmacodynamic samples and companion diagnostic assays for advanced therapy sponsors conducting French clinical studies. https://www.labcorp.com (Labcorp)
What Strategic Insights Define the Future of the France Pharmaceutical CRO Market?
Over the next five to seven years, competitive advantage in the French CRO market will shift from operational execution capabilities to data and technology platforms that enable new trial methodologies. The current market rewards CROs that can enroll patients quickly and deliver clean data on schedule, but these capabilities are becoming commoditized as electronic data capture and remote monitoring tools standardize across the industry. The next competitive frontier is real-world evidence integration, decentralized trial execution, and AI-powered patient identification, all of which require substantial technology investments and data access agreements that create barriers to entry for competitors lacking scale.
The risk that market projections may underestimate is regulatory acceptance timelines for novel trial methodologies. ANSM has published guidance on decentralized trials and real-world evidence, but actual regulatory review of submissions using these approaches remains limited. If regulatory authorities prove more conservative in practice than guidance documents suggest, sponsors may delay adoption of these methodologies, which would reduce demand for CRO capabilities in these areas and favor traditional site-based trial execution where regulatory precedent is well established. This would benefit incumbent CROs with strong site networks while disadvantaging newer entrants that invested heavily in decentralized trial platforms.
The opportunity that remains underpriced is post-approval real-world evidence services that combine Phase IV trial management with observational studies using French health databases. Payers increasingly require real-world effectiveness data to support reimbursement decisions, and sponsors need CROs that can design integrated programs satisfying both regulatory post-marketing requirements and payer evidence needs. The segment is currently small but offers higher margins than traditional Phase IV work because it requires specialized capabilities in health economics, epidemiology, and database analytics that most CROs do not maintain. Companies that build these capabilities will capture growing sponsor demand for evidence generation programs that serve multiple stakeholders rather than just satisfying regulatory obligations.
Frequently Asked Questions
Find quick answers to common questions.
The France Pharmaceutical CRO Market is Expected to reach USD 2,358.7 Million by 2033.
Key Segments for the France Pharmaceutical CRO Market are By Type (Clinical Research, Preclinical Research, Laboratory Services, Others); By Application (Drug Development, Clinical Trials, Toxicology Studies, Data Management, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Phase (Phase I, Phase II, Phase III, Phase IV, Others).
Major France Pharmaceutical CRO Market Players are IQVIA, Labcorp, Syneos Health, Parexel, ICON plc, Charles River, Medpace, PRA Health Sciences, Covance, Eurofins, WuXi AppTec, SGS, PPD, Clinipace, Veristat.
The Current France Pharmaceutical CRO Market size is USD 1,295.8 Million in 2025.
The France Pharmaceutical CRO Market CAGR is 7.82% from 2026 to 2033.
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


