France Pharmaceutical Contract Development And Manufacturing Organization Market Size & Forecast:
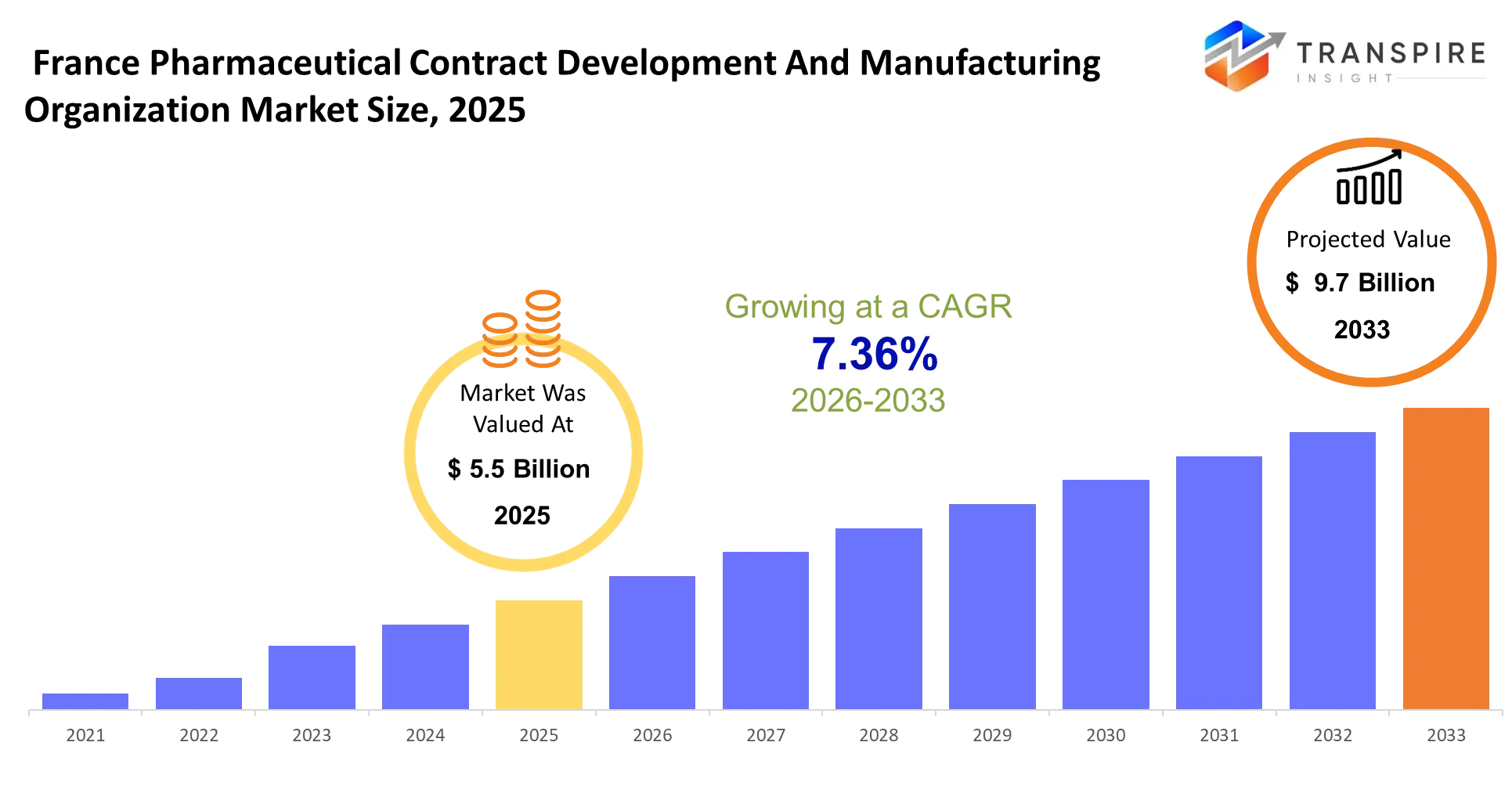
- France Pharmaceutical Contract Development And Manufacturing Organization Market Size 2025: USD 5.5 Billion
- France Pharmaceutical Contract Development And Manufacturing Organization Market Size 2033: USD 9.7 Billion
- France Pharmaceutical Contract Development And Manufacturing Organization Market CAGR: 7.36%
- France Pharmaceutical Contract Development And Manufacturing Organization Market Segments: By Type (API Manufacturing, Finished Dosage Manufacturing, Packaging Services, Others); By Application (Drug Development, Clinical Trials, Commercial Production, Biologics Manufacturing, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Scale (Preclinical, Clinical, Commercial, Others)
To learn more about this report, Download Free Sample Report
France Pharmaceutical Contract Development And Manufacturing Organization Market Summary
The France Pharmaceutical Contract Development And Manufacturing Organization Market was valued at USD 5.5 Billion in 2025. It is forecast to reach USD 9.7 Billion by 2033. That is a CAGR of 7.36% over the period.
The France Pharmaceutical Contract Development and Manufacturing Organization (CDMO) market operates as its main operational system because drug companies need to outsource their entire medicine development process which includes all aspects of manufacturing. The platform enables pharmaceutical and biotech companies to delegate their complex product development work which involves both clinical batch testing and large-scale production to external partners while maintaining regulatory standards to meet market entry schedules and financial obligations.
The market has undergone structural changes during the last three to five years because manufacturers now produce biologics and high-potency drugs which result from their need to develop increasingly complicated treatment solutions. The COVID-19 pandemic showed the world that supply chains could break down while European regulators wanted to create production systems which would protect their countries from external threats. France increased its dependence on local CDMO partners to guarantee supply protection and to speed up clinical research processes.
The industry has shifted from using external services mainly to cut expenses toward creating partnerships which help them maintain operational flexibility through their special skills and compliance with regulations.
Key Market Insights
- The pharmaceutical manufacturing infrastructure of France enables the country to maintain a 18 to 22 percent market share of Western European CDMO operations in 2025.
- The Southern France region will develop into its fastest expansion period from 2025 until 2033 because of its biotech cluster growth and government-supported pharmaceutical development.
- The Finished Dosage Manufacturing sector controls approximately 40 percent of the French Pharmaceutical Contract Development And Manufacturing Organization market according to 2025 market data.
- The second largest API manufacturing sector develops through rising needs for complex molecule production.
- The segment of biologics manufacturing will experience its most rapid growth pattern between 2025 and 2033 because of the development of monoclonal antibody pipelines.
- The application field of commercial production occupies the largest share at about 45 percent for 2025 because of existing pharmaceutical outsourcing agreements.
- The segment of clinical trials manufacturing shows the fastest growth rate because France experiences increased funding for its early-stage biotech industry.
- Pharma companies control approximately 55 percent of the market because they implement extensive outsourcing practices.
- Biotech firms now represent the fastest-growing end-user group because their startup pipelines continue to develop throughout France.
- Companies are developing their capacity through the construction of biologics manufacturing facilities and high-potency active pharmaceutical ingredient production facilities.
- The demand for clinical manufacturing increases because companies form strategic partnerships with biotech startups.
- The adoption of digital manufacturing systems enhances both production traceability and compliance requirements.
- The French expansion effort concentrates on establishing booster facilities throughout its regional hubs which connect to the Lyon and Paris biotech clusters.
What are the Key Drivers, Restraints, and Opportunities in the France Pharmaceutical Contract Development And Manufacturing Organization Market?
European pharmaceutical companies experience their primary growth driver through their current transition toward biologics and personalized medicine development pipelines. The process of developing these therapies requires drug developers to maintain a specific level of facility operations because they need access to sterile fill-finish systems and high-containment production environments which drug developers usually lack. The growing complexity of EU GMP standards has made it essential for French companies to use CDMO services instead of treating it as a business expense. The primary manufacturing plants have experienced a rise in both long-term contracts and capacity reservation activities.
The availability of advanced manufacturing facilities for cell and gene therapies represents a major limitation on business operations. Companies must spend substantial money to create facilities that meet compliance requirements which creates extensive validation processes that delay their growth. The organization faces a structural hindrance because it prevents companies from growing their operations during times of increased demand which results in project delays and the need to use non-European services that harm local income preservation.
The market presents a significant growth opportunity through the development of modular and flexible manufacturing systems. French contract development and manufacturing organizations (CDMOs) that invest in single-use bioreactor systems and continuous manufacturing systems will benefit from the increasing demand for biotech products. The Lyon biotech clusters have become attractive to international partners who want to conduct early-phase clinical production because this helps them achieve quicker drug development processes while creating additional revenue opportunities throughout their operations.
What Has the Impact of Artificial Intelligence Been on the France Pharmaceutical Contract Development And Manufacturing Organization Market?
The France Pharmaceutical Contract Development and Manufacturing Organization system experiences operational efficiency improvements because artificial intelligence enhances production accuracy and decreases manufacturing line downtime. CDMOs are using AI-based process monitoring systems to achieve better batch consistency results in biologics manufacturing, which requires strict control over product characteristics. Facilities use machine learning models to conduct predictive maintenance, which enables them to forecast reactor and filtration units and fill-finish system equipment failures before actual breakdowns occur, resulting in decreased unplanned shutdowns and higher plant utilization rates.
AI supports production scheduling optimization through its analysis of demand forecasts together with raw material availability and regulatory timelines, which leads to better throughput efficiency and shorter manufacturing lead times. Computer vision systems in quality control detect defects in vials and packaging with greater accuracy than manual inspection, which helps organizations achieve EU GMP standards.
The manufacturing sites in France encounter adoption difficulties because they must handle expensive system integration expenses and their existing systems use outdated infrastructure. The production facilities experience restricted access to real-time data, which hampers their ability to implement full-scale operations because high-security biologics environments require strict data transfer limitations.
Key Market Trends
- The manufacturing capacity for biologics increased by approximately 25% in France during the period from 2023 to 2025 because of the growing number of monoclonal antibody development projects.
- The European Union directed contract development manufacturing organizations to implement more rigorous compliance measures and to upgrade their operational facilities because of the upcoming Good Manufacturing Practice standards that will take effect in 2024.
- The percentage of clinical manufacturing work that French pharmaceutical companies outsource to external service providers reached a level above 60% by the year 2025.
- The pharmaceutical industry adopted modular and single-use manufacturing systems, which led to a significant decrease in setup times for biologics production lines.
- Recipharm and Lonza enhanced their French business operations through facility improvements and capacity growth based on their strategic partnerships.
- The demand for clinical trial outsourcing experienced rapid growth after 2022 because of the increasing number of biotech startups establishing operations in the Paris and Lyon regions.
- Digital batch monitoring systems enabled CDMO plants to achieve better production quality control by providing real-time tracking, which resulted in decreased production errors.
- The demand for high-potency active pharmaceutical ingredients increased substantially because European pharmaceutical companies developed new oncology drug pipelines.
- French companies increased their usage of domestic contract development manufacturing organizations because of supply chain localization strategies that became stronger after the COVID pandemic.
- The implementation of automation technology in packaging and fill-finish processes led to better operational efficiency while minimizing mistakes from human operators.
France Pharmaceutical Contract Development And Manufacturing Organization Market Segmentation
By Type
API manufacturing maintains a strong market position in France Pharmaceutical Contract Development And Manufacturing Organization Market because it supplies essential active ingredients to both generic and innovative drug products. Pharmaceutical companies maintain high demand because they prefer to outsource their complicated synthesis needs which require unique chemical knowledge and complete compliance with legal standards. The existing CDMO networks in Europe create a stronger connection to this market segment because they maintain their current capacity through their established distribution networks. EU GMP standards require companies to meet their production needs through outsourcing instead of building internal manufacturing facilities.
The segment experiences growth because there is an increasing need for high-potency active pharmaceutical ingredients which pharmaceutical companies use in oncology treatments and advanced medical therapies. Drug developers use specialized chemical infrastructure as their primary resource, which prevents them from achieving fast growth and results in them depending on third-party services. The API manufacturing sector will develop automated production systems which will run continuous production operations during the entire time period covered by our forecast. Process intensification and green chemistry development work enables investors and manufacturers to secure better long-term contract terms.
By Application
Drug development holds a significant position due to its foundational role in transforming molecular research into viable therapeutic candidates. Pharmaceutical and biotech firms rely on external CDMOs for formulation design, stability testing, and scale-up support. The high failure rates during early development stages require companies to depend on experienced outsourcing partners because these partners help decrease cost and time inefficiencies. France attracts early-stage development activities because its regulatory expertise enables effective research monitoring.
The application grows because novel therapies now require more complex solutions which include gene-based treatments and precision medicines. Biotech startups increase their research and development outsourcing which creates higher demand for specialized development services. Drug development services will shift towards data-driven operations which will incorporate predictive modeling and digital simulation tools throughout the forecast period. CDMOs that possess sophisticated analytical tools will secure more valuable contracts while establishing lasting research relationships.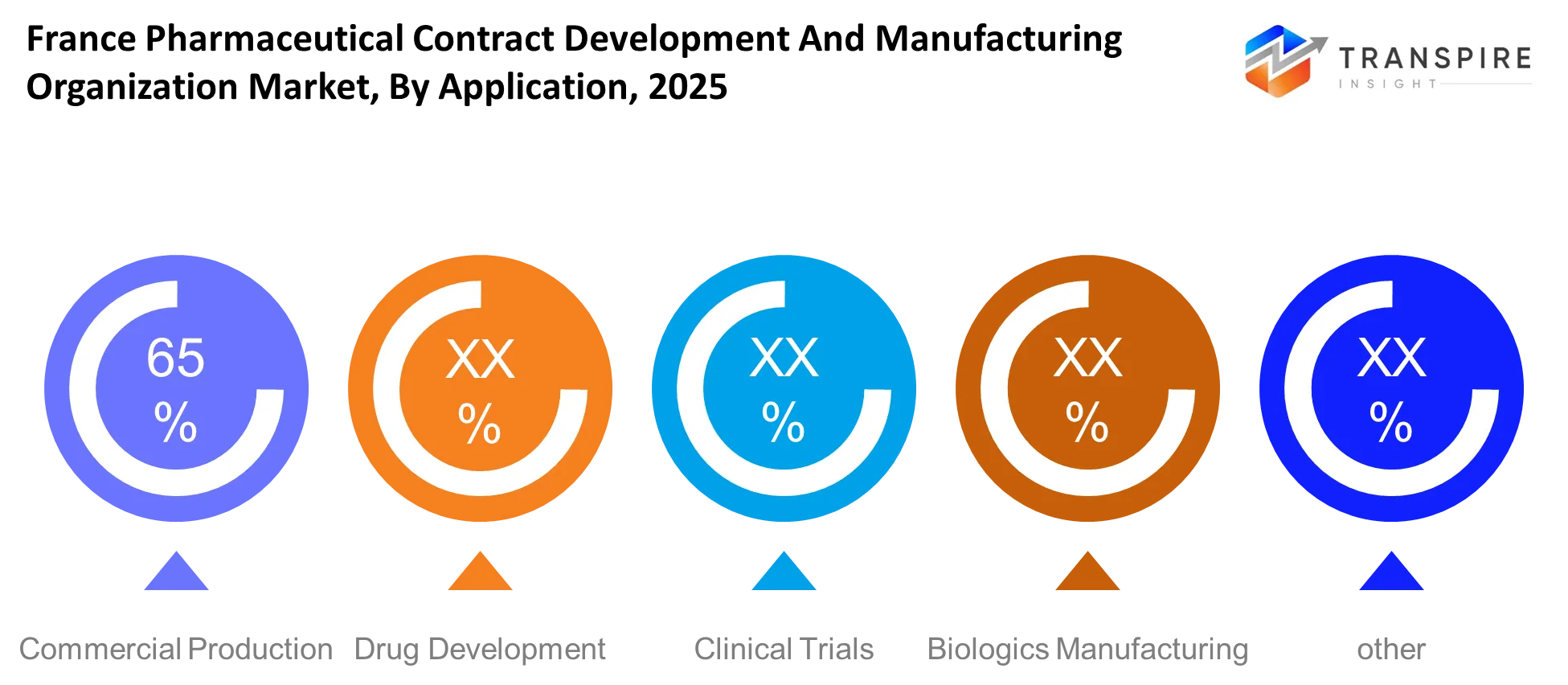
To learn more about this report, Download Free Sample Report
By End-User
Pharmaceutical companies control the end-user market because they contract most of their production and research work to outside partners. Established pharmaceutical firms rely on CDMOs to reduce operational costs and focus on core research activities. Companies depend on outsourcing because of the combination of strict regulatory rules and their complicated international supply networks. France serves as a strategic manufacturing base for many multinational pharmaceutical organizations.
The company experiences growth because it expands its portfolio through new chronic disease treatments and its existing biologics development pipelines. Companies face increasing pressure to decrease their internal production expenses which leads them to increase their outsourcing activities. Over the forecast period, pharma companies will deepen strategic partnerships with CDMOs for long-term capacity planning. The major pharmaceutical companies will start using integrated service models as their standard operating procedure.
By Scale
The preclinical scale serves as a crucial element that enables early drug candidate validation which occurs before human testing begins. The development pipeline needs risk reduction which pharmaceutical companies create through their product development requirements. CDMOs offer services which include testing formulations and conducting toxicity studies and optimizing production methods. The scientific infrastructure in France provides strong support for researchers who conduct their work during the initial stages of research.
The increasing number of biotech startups creates demand for their early-stage outsourcing needs. The rising design difficulty of molecular structures drives organizations to depend on particular preclinical testing solutions. The segment will use advanced predictive modeling tools during the upcoming forecast period. CDMOs that provide complete preclinical testing services will obtain competitive benefits.
The clinical scale sector shows rapid growth because more clinical trials begin worldwide. Pharmaceutical companies rely on contract development and manufacturing organizations to produce clinical trial materials while following regulatory requirements. The demand for European clinical trials receives support from decentralized clinical trial approaches which operate throughout the continent. France acts as a central location for both clinical manufacturing operations and supply chain activities.
What are the Key Use Cases Driving the France Pharmaceutical Contract Development And Manufacturing Organization Market?
The France Pharmaceutical Contract Development And Manufacturing Organization Market exists primarily because commercial production serves as its main operational purpose. Pharmaceutical companies need to outsource their manufacturing operations because it helps them save costs while ensuring they meet regulatory requirements and maintain product availability throughout Europe. The established CDMOs maintain long-term production contracts because there exists a strong demand for chronic disease medications and biologics.
The biotech sector and pharmaceutical companies drive growth in clinical trial manufacturing and biologics development because they need to speed up their pipeline development. Outsourcing enables the production of small batches needed for adaptive trials and oncology research and monoclonal antibody development. CDMOs provide research institutes and biotech firms with two essential resources: flexible capacity and expert sterile processing capabilities, which particularly benefit their early-stage and mid-stage development projects.
The upcoming applications will focus on developing personalized medicine products and manufacturing cell and gene therapies. These applications need manufacturing processes that create precise patient-specific batches and provide extensive regulatory tracking capabilities. The demand during the forecast period will increase because precision medicine becomes more widespread and CDMOs establish modular high-containment facilities throughout France’s biotech clusters.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 5.5 Billion |
|
Market size value in 2026 |
USD 5.9 Billion |
|
Revenue forecast in 2033 |
USD 9.7 Billion |
|
Growth rate |
CAGR of 7.36% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
United States; Canada; Mexico; United Kingdom; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; United Arab Emirates |
|
Key company profiled |
Lonza, Catalent, Thermo Fisher, Recipharm, WuXi AppTec, Samsung Biologics, Boehringer Ingelheim, AbbVie, Pfizer CentreOne, AGC Biologics, Siegfried, Fareva, Evonik, Piramal Pharma, Jubilant Pharmova |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (API Manufacturing, Finished Dosage Manufacturing, Packaging Services, Others); By Application (Drug Development, Clinical Trials, Commercial Production, Biologics Manufacturing, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Scale (Preclinical, Clinical, Commercial, Others) |
Which Regions are Driving the France Pharmaceutical Contract Development And Manufacturing Organization Market Growth?
The Île-de-France region dominates France's Pharmaceutical Contract Development And Manufacturing Organization Market because it contains a high number of pharmaceutical companies and regulatory bodies and advanced research facilities. Paris serves as the central location where global pharmaceutical companies develop their outsourcing plans together with their contract development and manufacturing organization partners. The region experiences continuous demand for outsourcing development and manufacturing services because major hospitals and research universities conduct extensive clinical trials. The area benefits from its close distance to regulatory agencies which helps speed up the approval process while companies make outsourcing decisions based on regulatory requirements.
Auvergne-Rhône-Alpes exists as a dependable industrial sector that maintains economic stability through its manufacturing facilities which function better than the capital region's operational structure. Lyon's existing pharmaceutical production system creates constant market need for active pharmaceutical ingredients and complete medication products. The region generates economic power through international pharmaceutical businesses which establish permanent operations and hire proficient workers for industrial roles. The region experiences growth through its manufacturing capacity which enables businesses to produce goods for export markets whereas Île-de-France depends on its policy framework to drive demand.
The fastest developing area in Provence-Alpes-Côte d'Azur results from recent establishment of biotech clusters together with government financial support for advancement in life sciences research. The establishment of new research parks and updated pharmaceutical manufacturing facilities has created an environment that attracts startups and mid-sized biotech companies. The region's demand patterns have changed since 2023 because of increased funding for biologics and clinical research facilities. The current business expansion creates significant market entry points for contract development and manufacturing organization companies which intend to develop flexible production systems and introductory development solutions throughout the southern United States.
Who are the Key Players in the France Pharmaceutical Contract Development And Manufacturing Organization Market and How Do They Compete?
The France Pharmaceutical Contract Development And Manufacturing Organization Market shows a moderately consolidated structure which enables global CDMOs to compete with pharmaceutical manufacturers that operate their own production facilities. The competition between companies is determined by their technological expertise and ability to meet regulations and their capacity to work with advanced biological medicines and high-potency active pharmaceutical ingredients. Established players defend market share through long-term contracts, while new market entrants concentrate their efforts on developing niche biologics through their quick manufacturing abilities and adaptable production methods.
The competition between Lonza and Samsung Biologics centers on their advanced biological manufacturing facilities which produce high-volume bioreactor systems needed for monoclonal antibody manufacturing. Lonza strengthens its position through integrated development-to-commercial manufacturing services, while Samsung Biologics expands through continuous facility scale-up in Europe-linked supply chains. Catalent strengthens its market position by providing flexible fill-finish services and expedited clinical trial supply solutions which help biotech companies that need quick turnaround times.
Recipharm and Fareva establish their European manufacturing operations through cost-effective production networks, with Recipharm using its acquisitions to build sterile manufacturing capabilities throughout various facilities. Fareva develops its competitive edge by offering diverse contract packaging options together with its ability to produce industrial-scale products. Thermo Fisher and AGC Biologics use their technology-driven platforms to compete against each other through their systems which combine advanced analytics and single-use equipment for enhanced production efficiency. These strategies collectively intensify competition through specialization rather than pure scale expansion.
Company List
- Lonza
- Catalent
- Thermo Fisher Scientific
- Recipharm
- WuXi AppTec
- Samsung Biologics
- Boehringer Ingelheim
- AbbVie
- Pfizer CentreOne
- AGC Biologics
- Siegfried
- Fareva
- Evonik Industries
- Piramal Pharma
- Jubilant Pharmova
Recent Development News
In March 2026, Lonza announced expansion of its biologics manufacturing facility network in Europe. The expansion increases large-scale monoclonal antibody production capacity and strengthens supply security for European pharmaceutical clients.
https://www.lonza.com
In January 2026, Samsung Biologics entered a strategic collaboration with a European biotech consortium. The partnership focuses on accelerating clinical-stage biologics manufacturing and improving cross-border supply chain integration.
https://www.samsungbiologics.com
What Strategic Insights Define the Future of the France Pharmaceutical Contract Development And Manufacturing Organization Market?
The France Pharmaceutical Contract Development And Manufacturing Organization Market is currently experiencing a structural transformation which results in biologics becoming the leading manufacturing technology and modular production systems and digital manufacturing networks becoming the essential components of its operations. The industry will grow through CDMOs which provide development and clinical supply and commercial manufacturing services from a single adaptable system. The production capacity will shift towards specialized facilities because the market will focus on treating high-value medical conditions.
The organization encounters an undisclosed danger because it relies on a few specialized biologics plants which will create operational delays during periods of intense clinical requirements. The market faces supply concentration risk which makes it vulnerable to interruptions that result from geopolitical events and regulatory changes affecting international raw material procurement.
AI-based manufacturing optimization together with continuous production systems create new business opportunities for companies operating in southern France's biotech clusters. CDMOs that establish their adaptive manufacturing platforms together with their data-driven compliance systems at an early stage will receive benefits through extended contract periods. The most powerful growth strategy for the period between 2026 and 2033 involves building modular biologics manufacturing facilities.
France Pharmaceutical Contract Development And Manufacturing Organization Market Report Segmentation
By Type
- API Manufacturing
- Finished Dosage Manufacturing
- Packaging Services
- Others
By Application
- Drug Development
- Clinical Trials
- Commercial Production
- Biologics Manufacturing
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Others
By Scale
- Preclinical
- Clinical
- Commercial
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Confirmed 2033 Market size figure is USD 9.7 Billion.
Key Segments for the France Pharmaceutical Contract Development And Manufacturing Organization Market are By Type (API Manufacturing, Finished Dosage Manufacturing, Packaging Services, Others); By Application (Drug Development, Clinical Trials, Commercial Production, Biologics Manufacturing, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Scale (Preclinical, Clinical, Commercial, Others).
Major France Pharmaceutical Contract Development And Manufacturing Organization Market players are Lonza, Catalent, Thermo Fisher, Recipharm, WuXi AppTec, Samsung Biologics, Boehringer Ingelheim, AbbVie, Pfizer CentreOne, AGC Biologics, Siegfried, Fareva, Evonik, Piramal Pharma, Jubilant Pharmova.
The Current France Pharmaceutical Contract Development And Manufacturing Organization Market size is USD 5.5 Billion in 2025.
The France Pharmaceutical Contract Development And Manufacturing Organization Market CAGR is 7.36% from 2026 to 2033.
- Lonza
- Catalent
- Thermo Fisher Scientific
- Recipharm
- WuXi AppTec
- Samsung Biologics
- Boehringer Ingelheim
- AbbVie
- Pfizer CentreOne
- AGC Biologics
- Siegfried
- Fareva
- Evonik Industries
- Piramal Pharma
- Jubilant Pharmova
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


