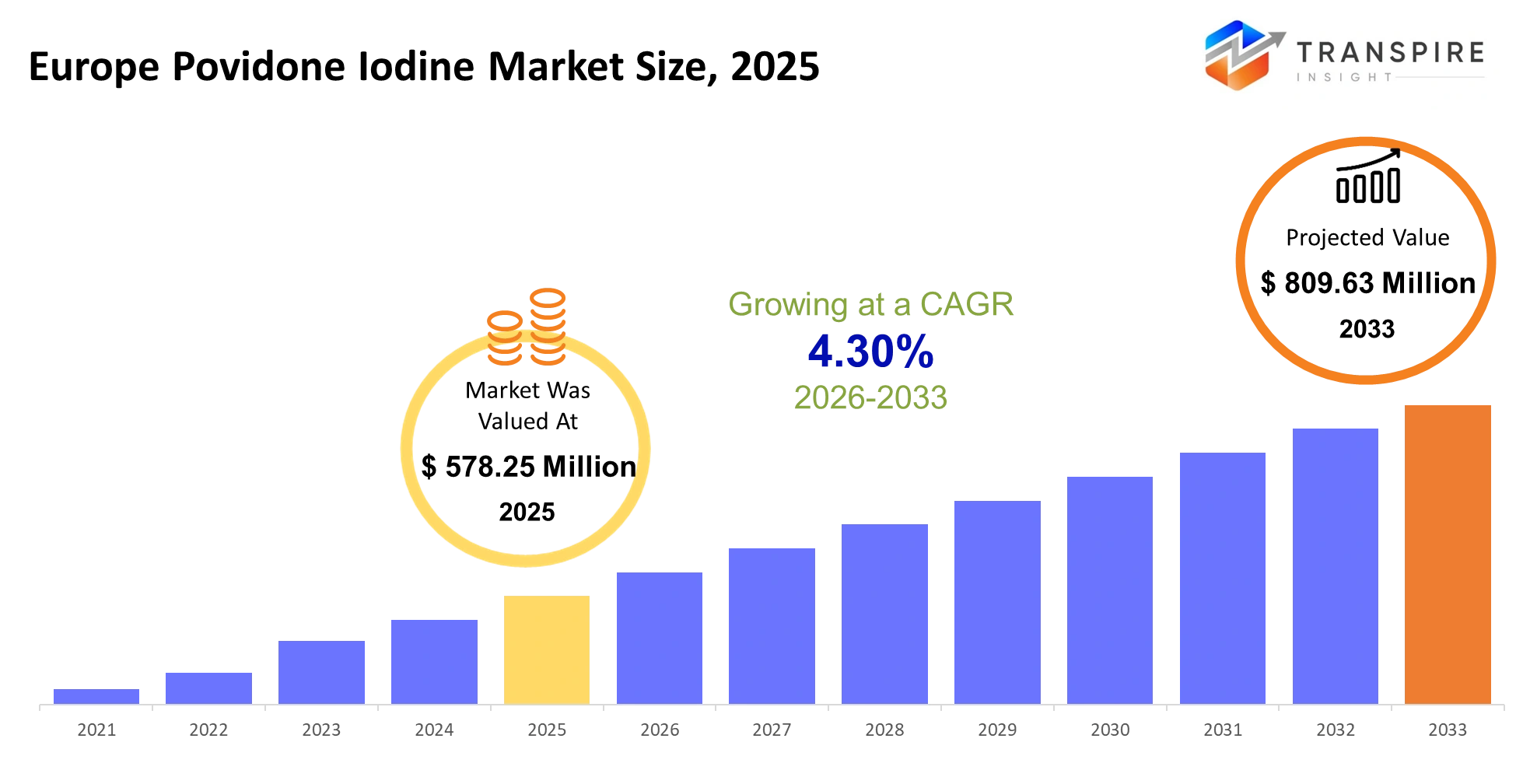
Europe Povidone Iodine Market Size & Forecast:
- Europe Povidone Iodine Market Size 2025: USD 578.25 Million
- Europe Povidone Iodine Market Size 2033: USD 809.63 Million
- Europe Povidone Iodine Market CAGR: 4.30%
- Europe Povidone Iodine Market Segments: By Type (Solution, Ointment, Scrub, Powder, Others), By Application (Wound Care, Surgical Antiseptic, Infection Prevention, Skin Disinfection, Others), By End-User (Hospitals, Clinics, Homecare, Laboratories, Others), By Form (Liquid, Gel, Spray, Powder, Others).
To learn more about this report, Download Free Sample Report
Europe Povidone Iodine Market Summary:
The Europe Povidone Iodine Market size is estimated at USD 578.25 Million in 2025 and is anticipated to reach USD 809.63 Million by 2033, growing at a CAGR of 4.30% from 2026 to 2033. The Europe Povidone Iodine Market operates as an essential element for infection control across hospitals, outpatient clinics, pharmaceutical manufacturing, and veterinary facilities because its contamination control methods directly determine patient safety and operational effectiveness. The healthcare system utilizes povidone iodine products which established themselves as essential components for surgical preparation, wound treatment, and medical equipment sterilization processes that hospitals must use because they cannot find cheaper substitutes.
During the past three to five years, the market transformed from basic antiseptic products toward clinically distinct antiseptic formulations which include low-irritation solutions and ready-to-use applicators and extended-release products designed for high-throughput healthcare environments. The COVID-19 pandemic served as a significant driver which revealed weaknesses in infection control systems while hospitals were forced to implement wider antiseptic product selection procedures.
Europe's increasing elderly population together with its growing number of surgical procedures has generated higher demand for medical procedures which establishes a connection between healthcare usage and ongoing antiseptic usage. The new process enables manufacturers to maintain pricing stability when they fulfill increased regulatory demands for product traceability and hospital purchasing standards.
Key Market Insights
- The Germany market for Povidone Iodine products leads all European countries through its 2025 market share which reaches 24% because of its developed healthcare system and high number of surgical procedures.
- The United Kingdom and France maintain their large market share because hospitals modernize and pharmaceutical companies increase their production capabilities.
- The Italian market exhibits the highest growth rate among all regional markets until 2030 because of its aging population and increasing outpatient surgical operations.
- Eastern European countries display rising healthcare demand as they increase spending and develop infection control measures following their infrastructure improvements from the pandemic.
- The Europe Povidone Iodine Market features solutions and liquid antiseptics as its leading products which account for more than 48% of market share because hospitals need quick medical preparation solutions for surgical procedures.
- The market for ointments and creams occupies the second biggest position because people use them widely to treat chronic wounds and skin conditions.
- Healthcare facilities choose ready-to-use applicators as their most efficient solution for improving work processes and decreasing contamination throughout the entire forecasting period.
- The European market for pharmaceutical-grade povidone iodine formulations expands because regulations require antiseptic products to have traceable high-purity components.
- The Europe Povidone Iodine Market generates 42% of its revenue through surgical site infection prevention which grows with the increase of orthopedic and cardiovascular surgical operations.
- European market shows the highest growth rate for wound care products because diabetic ulcers and chronic injuries create a growing need for treatment solutions.
- Medical device disinfection functions as a vital application area because hospitals and diagnostic laboratories now enforce stricter rules about their sterilization practices.
What are the Key Drivers, Restraints, and Opportunities in the Europe Povidone Iodine Market?
The COVID-19 pandemic has caused hospitals and outpatient surgical centers to implement stricter infection control measures which now serve as the primary driver for growth in the European Povidone Iodine Market. The European healthcare systems implemented mandatory sterilization and pre-operative antiseptic procedures to decrease hospital-acquired infections during high-risk surgical procedures and when treating elderly patients. Hospitals now choose products that demonstrate effective results and fast use and accepted by regulatory bodies which brought about increased buying of broad-spectrum antiseptics including povidone iodine. Manufacturers who provide surgical scrubs and wound care products and sterilization equipment achieve more dependable revenue streams through their permanent contracts with healthcare facilities.
The market's primary obstacle exists because regulatory and formulation-related challenges create difficulties for iodine sensitivity testing and product stability assessments and clinical approval processes. European medical safety standards require extensive testing, traceability documentation, and manufacturing compliance before new formulations can enter hospital procurement systems. Smaller manufacturers face financial difficulties when they try to pay for certification processes, which causes their product development to take more time and decreases their ability to introduce new products to the market. The advanced delivery system implementation process faces revenue growth restrictions because market entry remains restricted to established suppliers.
Southern and Eastern Europe present a significant future prospect through their advanced wound management systems and their home healthcare services. The demand for simple antiseptic products which people can use beyond hospital environments is growing because of the increasing number of diabetic ulcers and the aging population. The market will grow in this next phase for manufacturers who invest in single-use applicators and telehealth-compatible wound care kits and pharmacy distribution partnerships.
What Has the Impact of Artificial Intelligence Been on the Europe Povidone Iodine Market?
The European Povidone Iodine Market experiences a transformation through artificial intelligence and digital technologies which enhance manufacturing accuracy as well as inventory control and hospital supply chain operations. Pharmaceutical companies utilize AI-powered quality control systems to continuously track iodine concentration and sterility levels and batch quality throughout the production process. The automatic inspection systems that employ machine vision technology can detect formulation changes at a faster rate than traditional manual testing methods which enables manufacturers to decrease production waste while preventing expensive compliance violations.
Machine learning models enhance demand forecasting accuracy for antiseptic products used in hospitals and outpatient care facilities. Suppliers who analyze surgical volumes and seasonal infection patterns and procurement cycles can achieve better inventory distribution while decreasing stock shortages. European healthcare distributors now implement predictive analytics to optimize their logistics processes which results in faster delivery times and reduced storage expenses. The predictive maintenance systems operating in manufacturing plants oversee filling machines and sterilization systems to minimize unexpected downtime and boost operational efficiency by specific percentages.
Digital systems enable businesses to maintain regulatory compliance because they automatically document raw material origins and track product distribution throughout the supply chain. The implementation process faces significant challenges because mid-sized manufacturers lack the necessary funding to establish required AI systems while their existing systems depend on outdated production methods.
Key Market Trends
- The European hospitals have established permanent contracts for antiseptic supplies since 2021 to combat supply shortages which the pandemic revealed as a critical problem.
- After the European surgical facilities experienced an increase in operations during 2022 3M and B. Braun developed new ready-to-use applicator products for their existing product lines.
- The automated systems which handle pharmaceutical filling and maintain sterility monitoring achieved a 10 to 15 percent reduction in batch rejection rates from 2020 until 2024.
- Outpatient surgical centers started using more povidone iodine because patients began choosing minimally invasive procedures which no longer required them to stay in hospitals.
- The European regulators established stricter requirements for traceability and sterilization compliance which obligated smaller manufacturers to spend more money on developing digital quality-control systems from 2022 onward.
- The market share for chronic wound care products increased because diabetes-related ulcer treatment demands rose among Germany and Italy's elderly population.
- Pharmaceutical buyers increasingly favored low-irritation and extended-release iodine formulations after hospitals reported improved patient tolerance during post-surgical recovery programs.
- The Eastern European healthcare systems started investing more in antiseptic infrastructure after 2021 which created new opportunities for multinational infection-control distributors.
- The European healthcare distribution networks adopted AI-powered inventory forecasting systems which enabled suppliers to decrease stock shortages while enhancing their delivery planning capabilities.
- The competitive landscape between 2022 and 2025 adopted a new direction because hospitals began forming partnerships which offered bundled infection-prevention solutions instead of purchasing standalone antiseptic products.
Europe Povidone Iodine Market Segmentation
By Type
Solution-based products hold the leading position within the type segment because hospitals and surgical centers depend on rapid-acting antiseptic formulations for pre-operative preparation and infection control. The commercial value of scrubs remains strong because hospital staff uses them frequently to prevent contamination in both operating rooms and intensive care settings. Ointments continue gaining traction in chronic wound management because longer skin contact improves treatment efficiency for burns, ulcers, and post-surgical recovery.
The adoption rate of powder formulations remains limited because institutional healthcare environments need multiple application methods. Procurement teams select ready-to-use and low-irritation formulations because they enhance workflow efficiency while maintaining patient safety standards. Manufacturers are investing in formulation stability, extended shelf life, and controlled iodine release technologies to strengthen differentiation. The type segment will experience future competition from clinically specialized products that deliver improved treatment outcomes while fulfilling European regulatory and traceability requirements.
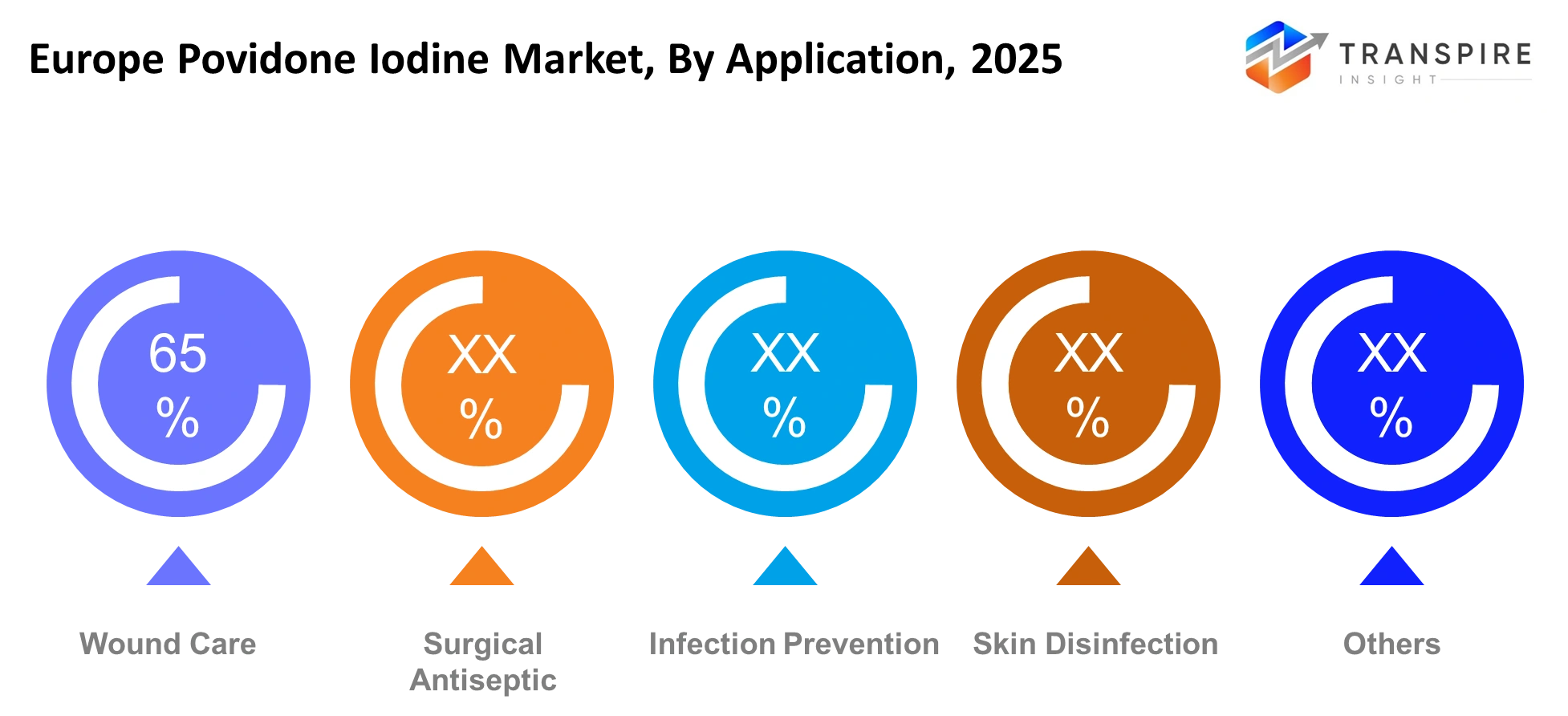
By Application
The healthcare system maintains its primary use of surgical antiseptics because European medical facilities have increased their capacity to perform orthopedic and cardiovascular and outpatient surgical procedures. Hospitals generate their main revenue through infection prevention which they accomplish by enhancing their sterilization methods that they adopted after the healthcare industry underwent changes following the pandemic. The field of wound care research progresses forward because there exists a rising need for antiseptic solutions to treat diabetic ulcers and traumatic injuries and age-related diseases which require continuous treatment. The demand for skin disinfection products remains constant in both emergency medical situations and normal hospital operations while specialized products maintain their presence in veterinary treatment facilities and research laboratories.
The current market trends display a movement toward businesses developing comprehensive infection control systems which customers purchase as complete solutions instead of individual disinfectant products. Healthcare organizations now prefer products that provide fast bacterial elimination together with minimal skin discomfort and easy use in medical situations. The upcoming funding possibilities will focus on advanced technologies for wound care and home-health solutions because healthcare systems need to decrease their hospital expenses and minimize patient time in medical facilities.
To learn more about this report, Download Free Sample Report
By End-User
The primary end-user group for antiseptic products remains hospitals because these institutions require large quantities of antiseptic products to support their surgical procedures and wound treatment needs and emergency medical services and sterilization practices. Clinics represent the second-largest segment as outpatient procedures continue shifting away from traditional inpatient settings across several European healthcare systems. Homecare applications are expanding rapidly because aging populations and increasing chronic wound cases and growing acceptance of self-managed recovery programs backed by pharmacies and telehealth services.
Laboratories need infection-control products which they require to handle samples and produce pharmaceuticals and conduct sterile testing. End users prefer suppliers who can provide them with unfailing product supply together with all necessary compliance documents and complete distribution services. Public healthcare organizations use their budget constraints to create supplier relationships which result in predictable purchasing patterns. The end-user segment will experience growth in the future which will benefit manufacturers whose product development matches the demand for outpatient services and decentralized health systems.
By Form
The form segment of medical products is primarily filled through liquid formulations because hospitals and surgical facilities need antiseptic solutions which they can use immediately during their extensive medical operations. Wound management and dermatological treatment now use gel formulations because their thicker consistency enables better targeted treatment while preventing excessive product waste. Spray formats are gaining attention in emergency response, outpatient care, and home treatment settings where quick and contact-minimized application supports operational convenience.
Powder forms continue to exist for specialized applications but their acceptance rate remains low because they lack the flexible uses of liquid and gel products. The market now prefers packaging solutions that include single-use dispensers, sterile applicators, and controlled-dose delivery systems which minimize contamination risks and make handling easier. Manufacturers are directing investments toward user-friendly formats that improve storage efficiency and support portable healthcare delivery. Future product development will focus on creating convenient formulations which medical centers, home healthcare services, and digital wound monitoring systems can use.
What are the Key Use Cases Driving the Europe Povidone Iodine Market?
The main reason European hospitals and ambulatory surgical centers use surgical antiseptic preparation products is because it functions as their primary use case. The orthopedic and cardiovascular and trauma surgery fields maintain their need for surgical procedures because hospitals must follow infection control regulations before performing any invasive medical procedure.
Clinics and homecare services and long-term care facilities for elderly patients show increasing interest in using wound care products and outpatient skin disinfection solutions. The rising cases of diabetic ulcers and post-surgery home recovery programs are causing more people to use gel-based and ready-to-apply antiseptic products in non-hospital settings.
Antiseptics now find new uses through their application in telehealth-based wound management kits and veterinary infection control systems that commercial farms use. Pharmaceutical companies now investigate new delivery systems for povidone iodine which will provide extended antibacterial protection while reducing skin discomfort during long-term usage.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 578.25 Million |
|
Market size value in 2026 |
USD 603.09 Million |
|
Revenue forecast in 2033 |
USD 809.63 Million |
|
Growth rate |
CAGR of 4.30% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
3M, BASF, Ashland, Glide Chem, Adani Pharma, Boai NKY, JH Nanhang Life Sciences, Changzhou Yinsheng, Huaan Chemical, Nice Chemicals, Lasa Supergenerics, Samrat Pharmachem, Quat Chem, Radiant Pharma, Avrio Health. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Solution, Ointment, Scrub, Powder, Others), By Application (Wound Care, Surgical Antiseptic, Infection Prevention, Skin Disinfection, Others), By End-User (Hospitals, Clinics, Homecare, Laboratories, Others), By Form (Liquid, Gel, Spray, Powder, Others). |
Which Regions are Driving the Europe Povidone Iodine Market Growth?
The market extends its leadership to Western Europe because Germany, France, and the United Kingdom operate healthcare systems which enable their medical facilities to perform numerous surgeries while adhering to stringent protocols for preventing infections. The government enforces regulations which require hospitals to disinfect their facilities and pharmaceutical companies to produce safe drugs and protect patient welfare. The major manufacturers establish predictable long-term supply agreements through their partnerships with large hospital groups and centralized purchasing systems. The region maintains its dominant market position because pharmaceutical companies produce drugs at high capacity and distribute products through established networks while investing in outpatient medical facilities.
Northern Europe represents a more stability-driven market shaped by healthcare efficiency and long-term clinical planning rather than sheer procedural scale. The countries of Sweden and Denmark and the Netherlands develop standardized infection prevention protocols which all their hospitals and elderly care facilities and community healthcare systems must follow. The combination of stable government healthcare funding and the early implementation of digital procurement systems allows suppliers to maintain consistent product replacement cycles which generate dependable revenue streams. The region shows moderate growth compared to Western Europe yet serves as a vital source for continuous market demand because of its reduced procurement fluctuations and strong compliance with regulations.
The healthcare modernization programs in Southern and Eastern Europe create rapid growth in Italy and Spain and Poland and multiple Balkan nations. The pandemic resulted in increased hospital infrastructure funding which enhanced surgical capacity and created higher demand for antiseptic and wound-care products needed in both urban and regional healthcare facilities. The increasing diabetes rates and the growing elderly population require more chronic wound care in outpatient and homecare facilities because healthcare systems need to implement cost-effective patient recovery methods. The period from 2026 to 2033 will present substantial business chances for companies that produce and distribute and for investors who concentrate on cost-effective instant use products and local distribution agreements.
Who are the Key Players in the Europe Povidone Iodine Market and How Do They Compete?
The Europe Povidone Iodine Market maintains a moderately consolidated market structure because multinational healthcare companies and infection-control firms control most institutional procurement pipelines. The competitive environment now requires companies to demonstrate superior formulation quality and regulatory adherence and sterile packaging capabilities and hospital delivery systems instead of relying on price competition. Established manufacturers use their long-term healthcare agreements and their comprehensive infection-prevention product lines to protect their market position while smaller companies seek to enter through specialized wound-care and dermatology markets. The combination of high regulatory approval expenses and mandatory product traceability requirements creates obstacles for new market entrants because it benefits companies that possess modern manufacturing facilities and established medical reputation.
3M competes through efficiency improvements and surgical process systemization. The company uses preoperative skin preparation systems to establish a unique antiseptic product line which reduces contamination risks in operating rooms through its fast skin application method. The company establishes repeat sales through its strong partnerships with Western European hospital purchasing networks. B. Braun develops clinical-grade infection prevention products which work together with its complete surgical and infusion care product range. The company helps European healthcare providers through its focus on sterilization compatibility and regulatory compliance which enables hospital service support that leads to long-term procurement agreements.
Ecolab increases its business operations through its hygiene management services and its infection-control consulting services which include more than just antiseptic products. The company establishes its unique market position through its combination of digital monitoring technology and hospital sanitation systems which enhance both compliance assessment and operational performance. Mundipharma focuses its business operations on wound care solutions and specialized antiseptic products which serve hospitals and outpatient treatment facilities. The company expands its operations into home healthcare distribution networks while establishing partnerships with pharmacies to enhance patient reach throughout its service areas. Avrio Health boosts its market presence through its consumer-oriented antiseptic products and its partnerships with local retailers to distribute products specifically for self-care and non-hospital medical treatments.
Company List
- 3M
- BASF
- Ashland
- Glide Chem
- Adani Pharma
- Boai NKY
- JH Nanhang Life Sciences
- Changzhou Yinsheng
- Huaan Chemical
- Nice Chemicals
- Lasa Supergenerics
- Samrat Pharmachem
- Quat Chem
- Radiant Pharma
- Avrio Health
Recent Development News
In April 2026, Iofina Raises Guidance After Record Iodine Production and Advances Permian Expansion: UK-based iodine producer Iofina plc reported a strong operational jump in iodine output in Q1 2026, driven by expanded production capacity and improved brine conditions in its US operations. While not a finished povidone iodine product, iodine is a key raw material for antiseptic formulations widely used in Europe’s healthcare supply chain.
Source: https://uk.advfn.com
In March 2026, CORONA Remedies enters povidone iodine market via Wokadine acquisition: Corona Remedies acquired the antiseptic brand Wokadine from Dr Reddy’s Laboratories, strengthening its position in the povidone iodine segment. Although India-based, the deal impacts global supply chains, including European antiseptic procurement networks due to increased export availability.
Source: https://medicaldialogues.in
What Strategic Insights Define the Future of the Europe Povidone Iodine Market?
The Europe Povidone Iodine Market is developing toward a medical field that utilizes highly effective infection control solutions for outpatient treatment and wound care and decentralized patient treatment. The financial requirements of European healthcare systems need to decrease inpatient expenses while they continue to implement strict infection control measures for their elderly patients and increased surgical procedures. The market will favor manufacturers who develop new products through their combination of product development expertise and their ability to track products and extend their digital inventory systems and create packaging that suits home healthcare use.
The industry does not sufficiently acknowledge the danger which arises from manufacturers who depend on specific raw materials to acquire their pharmaceutical-grade iodine resources. Manufacturing companies face cost increases because their iodine supply interruptions force them to comply with new chemical handling requirements which particularly impact small regional businesses that operate with limited purchasing capacities.
Telehealth-enabled wound care programs present a major business opportunity which currently exists in Southern and Eastern European countries that develop their remote patient monitoring and pharmacy-based recovery systems. Market participants should prioritize partnerships with outpatient networks, homecare distributors, and digital healthcare providers rather than relying solely on traditional hospital procurement channels.
Europe Povidone Iodine Market Report Segmentation
By Type
- Solution
- Ointment
- Scrub
- Powder
- Others
By Application
- Wound Care
- Surgical Antiseptic
- Infection Prevention
- Skin Disinfection
- Others
By End-User
- Hospitals
- Clinics
- Homecare
- Laboratories
- Others
By Form
- Liquid
- Gel
- Spray
- Powder
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Povidone Iodine Market size for the market will be USD 809.63 Million in 2033.
The key segments of the Europe Povidone Iodine Market are By Type (Solution, Ointment, Scrub, Powder, Others), By Application (Wound Care, Surgical Antiseptic, Infection Prevention, Skin Disinfection, Others), By End-User (Hospitals, Clinics, Homecare, Laboratories, Others), By Form (Liquid, Gel, Spray, Powder, Others).
Major players in the Europe Povidone Iodine Market are 3M, BASF, Ashland, Glide Chem, Adani Pharma, Boai NKY, JH Nanhang Life Sciences, Changzhou Yinsheng, Huaan Chemical, Nice Chemicals, Lasa Supergenerics, Samrat Pharmachem, Quat Chem, Radiant Pharma, Avrio Health.
The current market size of the Europe Povidone Iodine Market is USD 578.25 Million in 2025.
The Europe Povidone Iodine Market CAGR is 4.30%.
- 3M
- BASF
- Ashland
- Glide Chem
- Adani Pharma
- Boai NKY
- JH Nanhang Life Sciences
- Changzhou Yinsheng
- Huaan Chemical
- Nice Chemicals
- Lasa Supergenerics
- Samrat Pharmachem
- Quat Chem
- Radiant Pharma
- Avrio Health
Recently Published Reports
-
Apr 2026
Healthcare Polymer Packaging Market
Healthcare Polymer Packaging Market Size, Share & Analysis Report By Packaging Type (Syringes, IV Bottles and Pouches, Clamshells, Blisters, Bottles & Jars, Containers, Tubes, IV Parental Packaging, Others), By Type (Regulated, Non-regulated), By Polymer Type (LDPE (Low-Density Polyethylene), HDPE (High-Density Polyethylene), Homo-polymer (Homo), Random Copolymer (Random), Block Copolymer (Block), PET, Polystyrene, Polyvinyl Chloride, Polyamide/EVOH, Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Hydrophilic Tape (Waterstop) Market
Hydrophilic Tape (Waterstop) Market Size, Share & Analysis Report By Type (Bentonite-Based Hydrophilic Tape, Rubber-Based Hydrophilic Tape), By Application (Residential Buildings, Commercial Buildings, Infrastructure Projects), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Metalens Market
Metalens Market Size, Share & Analysis Report By Type (Visible Light Metalens, and Infrared Metalens), By Application (Consumer Electronics, Automotive Electronics, Industrial, Medical, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
PBT Resin Market
PBT Resin Market Size, Share & Analysis Report By Type (Reinforced PBT Resin, Unreinforced PBT Resin), By Processing Method (Injection Molding, Extrusion, Blow Molding, Others), By End-User (Automotive, Electrical & Electronics, Consumer Appliances, Industrial Machinery, Medical Devices, Packaging, Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


