Europe Plug-and-Play CDMO Market Size & Forecast:
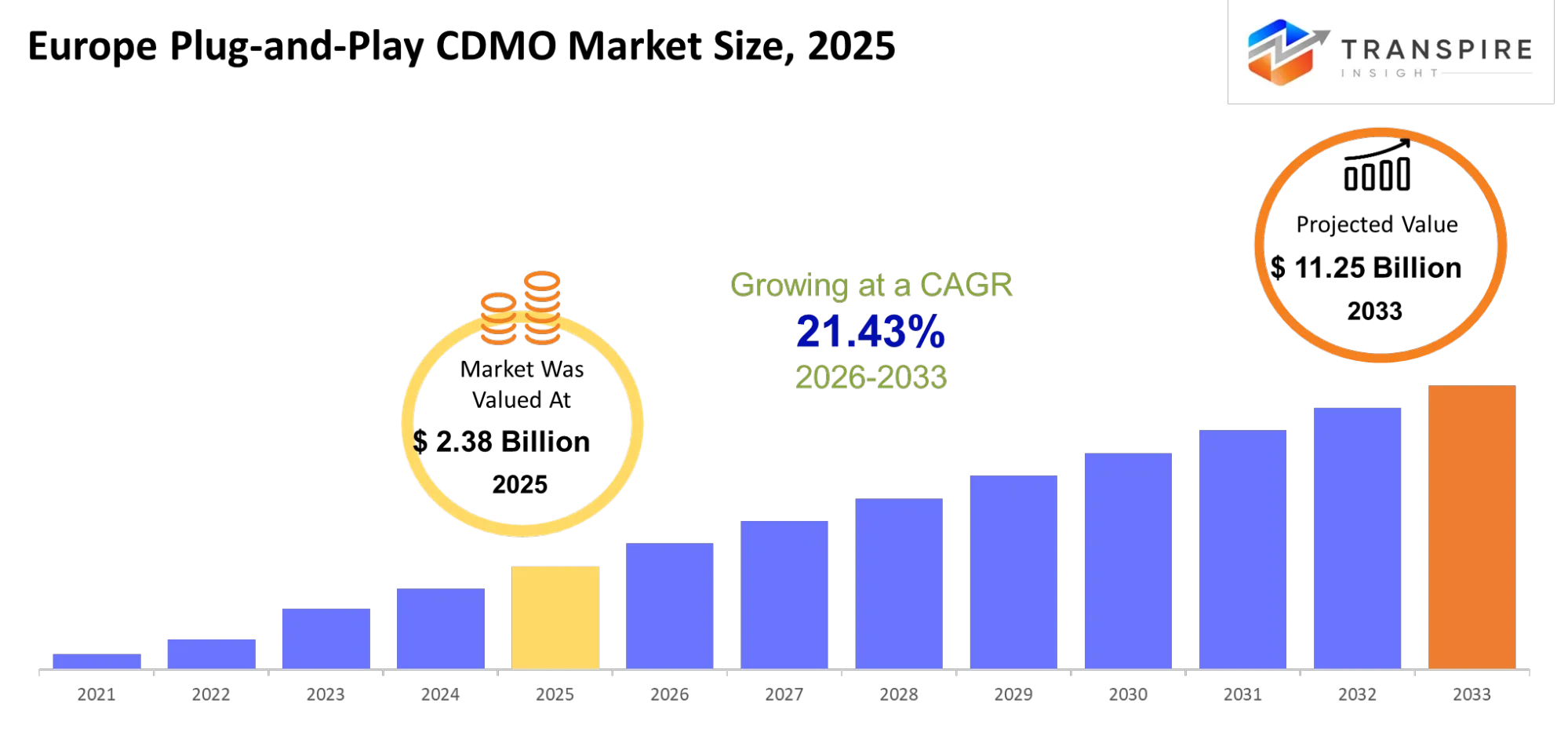
- Europe Plug-and-Play CDMO Market Size 2025: USD 2.38 Billion
- Europe Plug-and-Play CDMO Market Size 2033: USD 11.25 Billion
- Europe Plug-and-Play CDMO Market CAGR: 21.43%
- Europe Plug-and-Play CDMO Market Segments: By Type (Biologics CDMO, Small Molecule CDMO, Cell & Gene Therapy CDMO, Others); By Application (Drug Development, Manufacturing, Clinical Trials, Commercial Production, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Startups, Others); By Service (Process Development, Analytical Testing, Packaging, Regulatory Support, Others)
To learn more about this report, Download Free Sample Report
Europe Plug-and-Play CDMO Market Summary:
The Europe Plug-and-Play CDMO Market size is estimated at USD 2.38 Billion in 2025 and is anticipated to reach USD 11.25 Billion by 2033, growing at a CAGR of 21.43% from 2026 to 2033.
The European plug-and-play contract development and manufacturing organization market exists within the pharmaceutical services sector and will transform how pharmaceutical companies establish their manufacturing partnerships. The industry will develop through modular facilities that provide full operational capabilities, enabling biotech companies to move from research into production without substantial financial investment. The market demand will require quicker production times together with the ability to produce customized treatment solutions that match specific patient needs. The combination of single-use technology advancements and digital process monitoring systems will enable businesses to enhance their operational efficiency while maintaining better product quality. The European regulatory framework will establish stricter quality and traceability requirements which will force service providers to implement more visible operational systems that will determine how customers choose and maintain their manufacturing relationships.
What Has the Impact of Artificial Intelligence Been on the Europe Plug-and-Play CDMO Market?
Artificial intelligence is rapidly changing the decision-making process and value generation method of the europe plug-and-play cdmo market because it provides better operational accuracy and deeper business understanding. The European plug-and-play CDMO market uses AI to enhance market research capabilities through its ability to analyze large datasets which reveal unknown customer demand patterns and provide instant customer behavior updates. The European plug-and-play CDMO market uses predictive analytics and machine learning to allow businesses to better understand customer requirements while improving their demand estimation abilities and quick response capabilities to new treatment developments. The data-driven method will help organizations make better capacity decisions by decreasing unknown factors which hinder their planning process.
Artificial intelligence creates smart automation systems for modular production facilities in European plug-and-play CDMO market environments, which enhances operational efficiency while preserving high-quality product standards. The AI-powered monitoring systems will identify performance inconsistencies at an early stage, which enables them to enhance batch results while decreasing operational downtime and decreasing total expenses. Machine learning models will improve supply chain optimization by enhancing inventory control and optimizing logistical operations throughout multiple locations.
Key Market Trends & Insights:
- The European plug-and-play CDMO market sees Western Europe as its leading region which holds 55 percent market share in 2025 because of its established pharmaceutical system and its compliance with regulations.
- Central and Eastern Europe will become the most rapidly expanding area between 2026 and 2030 because of its cost benefits and growing international investment.
- The market for integrated plug-and-play manufacturing services will reach 48 percent in 2025 because customers need complete CDMO solutions.
- The modular facility leasing business will become the second biggest market segment because it helps startups which want to spend less money and expand quickly.
- The single-use technology services market will experience the highest growth rate until 2030 because these services provide operational efficiency and lower contamination risks and enable fast service delivery.
- The Europe plug-and-play CDMO market sees biologics manufacturing as its leading segment which accounts for 52 percent of the market because the demand for monoclonal antibody and cell therapy production increases.
- The demand for precise medical treatments drives personalized medicine and gene therapy applications which will grow at double-digit rates during the next decade.
- Biopharmaceutical companies lead the market in 2025 with more than 60 percent market share because they require flexible manufacturing solutions which can grow with their business.
- The company achieves better operational performance through its work with smart automation technologies and AI-based analytics and single-use system implementation.
- Product innovation and flexible service offerings are helping companies gain significant market share and maintain leadership in evolving CDMO market trends.
Europe Plug-and-Play CDMO Market Segmentation
By Type:
The Biologics CDMO services market will expand because more businesses develop complex therapies that require antibody manufacturing. The demand for small molecule CDMO services will remain stable because pharmaceutical companies have established drug development processes. The market for cell and gene therapy CDMO services will experience fast growth because medical providers now focus on personalized treatment methods. The other segments will encompass specialized manufacturing services which combine different production methods to meet specific requirements.
To learn more about this report, Download Free Sample Report
By Application:
Drug development will depend on its need for initial assistance combined with its need for methods that enable expansion to improve development speed. The main focus of manufacturing operations will continue to be manufacturing while modular systems provide operators with the ability to create different product types. Clinical trials will experience improved results because they can produce small quantities of products with high production efficiency. The commercial production capacity will grow because of the need for fast product development while the rest of the applications will tackle specialized production needs that emerge during operations.
By End-User:
Pharmaceutical companies will drive market demand because they need to outsource their operations while managing their expenses. Biotech companies will require adaptable production methods to support their research and development of new products. Research institutes will require smaller-scale production support. CROs and startups will increasingly depend on plug-and-play models to reduce infrastructure costs and accelerate entry into competitive markets.
By Service:
The process development team will work on enhancing production efficiency through their work on production scalability. The analytical testing process will verify that products meet established quality standards. Packaging services will create solutions that match different product specifications and shipping requirements. The European regulatory framework will guide our regulatory support services while our other services will provide customized solutions for specialized operational needs.
What are the Main Challenges for the Europe Plug-and-Play CDMO Market Growth?
The European plug-and-play contract development and manufacturing organization market expansion faces operational and technical limitations which create ongoing challenges. The application of modular systems provides users with flexible solutions but different platforms struggle to work together because of their different operational needs. Handling and storage requirements for biologics and cell-based therapies will create ongoing challenges because product stability requires specialized techniques. Supply chain interruptions for essential raw materials and single-use products will restrict production capacity because they create operational delays which disrupt manufacturing processes throughout all production sites.
The development of the European plug-and-play contract development and manufacturing organization market faces multiple challenges which include manufacturing and commercialization obstacles. Service providers will experience increased operational requirements with higher expenses because they must follow all Good Manufacturing Practice regulations. The process to authenticate modular facilities and maintain consistent product standards between different locations will require major financial resources. The high costs associated with advanced technologies along with delays in regulatory approval will prevent companies from bringing their products to market, which will impact smaller businesses that need to expand their production capacity.
The European plug-and-play contract development and manufacturing organization market faces challenges in adoption because of inconsistent infrastructure development and the lack of skilled workers. Emerging regions will experience slow progress toward adopting plug-and-play systems because they lack essential technical expertise and digital resources and financial backing. Many companies will remain cautious in shifting from traditional manufacturing due to perceived risks and integration costs. The evolution of outsourcing models together with existing contract development and manufacturing organizations will create more market restrictions, which will increase competitive pressure and pricing limits.
Regional Insights
The europe plug-and-play cdmo market will show varied performance across regions because different areas have different infrastructure systems and they follow different regulatory rules and they receive different levels of funding. Western Europe will lead with advanced facilities and strong pharmaceutical presence which enables other areas to develop at a constant speed. Market conditions will reflect demand for flexible manufacturing and faster development timelines which research environments need.
The biopharma clusters in western Europe and the availability of skilled workers and the existence of strong compliance systems create a foundation that leads to western Europe's dominance in advanced production models. Northern Europe will focus on innovation and sustainable manufacturing practices. Southern and Eastern Europe will experience gradual growth because of decreasing costs and infrastructure development and rising interest from global companies who want to expand their operations.
Recent Development News
In January 2026, Lonza reported slower projected growth for its core CDMO business in 2026, forecasting 11–12% sales growth compared to over 21% in 2025, reflecting changing demand dynamics and market pressures. https://www.reuters.com
In April 2026, Catalent Inc. announced expansion of its commercial license agreement with Cartherics Pty Ltd. The partnership will strengthen cell therapy manufacturing capabilities and accelerate commercialization of immune cell therapies in Europe. https://pharmaceuticalmanufacturer.media
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 2.38 Billion |
|
Market size value in 2026 |
USD 2.89 Billion |
|
Revenue forecast in 2033 |
USD 11.25 Billion |
|
Growth rate |
CAGR of 21.43% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Lonza, Catalent, Thermo Fisher, WuXi AppTec, Samsung Biologics, Recipharm, Siegfried, Cambrex, Evotec, Charles River, Boehringer Ingelheim, Fujifilm Diosynth, Piramal Pharma, Alcami, Jubilant Biosys |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Biologics CDMO, Small Molecule CDMO, Cell & Gene Therapy CDMO, Others); By Application (Drug Development, Manufacturing, Clinical Trials, Commercial Production, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Startups, Others); By Service (Process Development, Analytical Testing, Packaging, Regulatory Support, Others) |
How Can New Companies Establish a Strong Foothold in the Europe Plug-and-Play CDMO Market?
New entrants aiming to succeed in the europe plug-and-play cdmo market will need a focused strategy built on specialization and speed. The company will achieve better results through niche manufacturing of cell and gene therapy products and small-batch biologics, which face increasing demand while lacking sufficient production capacity. New players will establish themselves as solutions specialists by matching their services to existing industry trends that include personalized medicine and fast clinical development.
The europe plug-and-play cdmo market will use technology differentiation as its primary method for acquiring market traction. The implementation of smart automation and single-use systems and digital monitoring tools will enhance operational efficiency while decreasing the possibility of operational failures. The use of predictive analytics in process development will enable companies to deliver dependable outcomes which attract clients who value consistency. The implementation of innovative strategies which decrease turnaround time while increasing production capacity will resolve essential challenges that manufacturing facilities face in outsourced production.
The europe plug-and-play cdmo market entry will receive additional support through strategic partnership development. The company will establish its market position through partnerships with biotech startups and research institutes and established pharmaceutical firms, which will deliver credibility and consistent project flow. The emerging companies ExcellGene and NorthX Biologics have grown their capabilities through strategic partnerships and modular facility enhancements. The new companies will use these methods to expand their operations, take advantage of market opportunities, and create a strong competitive position within an industry that demands strict regulations.
Key Europe Plug-and-Play CDMO Market Company Insights
The Europe Plug-and-Play CDMO Market maintains its competitive strength because businesses now use modular manufacturing systems and their requirement for flexible manufacturing solutions. Market participants will focus on efficiency, rapid deployment, and compliance standards. Competitive intensity will rise as new entrants and established firms invest in advanced technologies and scalable solutions.
The main companies will use technology upgrades and strategic partnerships and European market entry to strengthen their market position. Automation and digital monitoring systems will boost operational performance while attracting new clients. Smaller firms will compete by offering specialized services and flexible pricing, while partnerships will support faster market entry and long-term growth stability.
Company List
- Lonza
- Catalent
- Thermo Fisher
- WuXi AppTec
- Samsung Biologics
- Recipharm
- Siegfried
- Cambrex
- Evotec
- Charles River
- Boehringer Ingelheim
- Fujifilm Diosynth
- Piramal Pharma
- Alcami
- Jubilant Biosys
What are the Key Use-Cases Driving the Growth of the Europe Plug-and-Play CDMO Market?
The europe plug-and-play cdmo market is gaining momentum through targeted use-cases that address speed, flexibility, and cost efficiency in drug development. The manufacturing of biologics serves as the main market driver since modular facilities enable fast production increases for both monoclonal antibodies and recombinant proteins. The method decreases time required for setup while enabling quicker clinical trials which results in both increased market development and higher customer acceptance.
Cell and gene therapy production represents a major use-case which drives up demand for european plug-and-play cdmo services. The therapies need plug-and-play systems because they require highly controlled environments to produce small-batch streams. The companies utilize these systems to manage their complex operations while following all necessary regulations which makes the systems suitable for developing personalized treatments. The upcoming trend will maintain its current path until advanced therapies achieve their market launch.
The european plug-and-play cdmo market receives substantial support from both clinical trial supply operations and small-scale production facilities. Biotech firms depend on flexible production units which enable them to adjust their production according to varying trial requirements without needing to invest heavily in new equipment. The contract manufacturing sector for niche and orphan drugs is experiencing growth because flexible systems can manage low-volume production needs.
The introduction of modular production methods will create new innovation possibilities beyond healthcare because they can be applied to adjacent sectors such as specialty chemicals and advanced materials. The expansion of the market will receive assistance from improved scalability and technological progress because these use-case applications create new revenue possibilities for service providers.
Europe Plug-and-Play CDMO Market Report Segmentation
By Type
- Biologics CDMO
- Small Molecule CDMO
- Cell & Gene Therapy CDMO
- Others
By Application
- Drug Development
- Manufacturing
- Clinical Trials
- Commercial Production
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- CROs
- Startups
- Others
By Service
- Process Development
- Analytical Testing
- Packaging
- Regulatory Support
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Plug-and-Play CDMO Market size for the market will be USD 11.25 Billion in 2033.
Key segments for the Europe Plug-and-Play CDMO Market are By Type (Biologics CDMO, Small Molecule CDMO, Cell & Gene Therapy CDMO, Others); By Application (Drug Development, Manufacturing, Clinical Trials, Commercial Production, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Startups, Others); By Service (Process Development, Analytical Testing, Packaging, Regulatory Support, Others).
Major Europe Plug-and-Play CDMO Market players are Lonza, Catalent, Thermo Fisher, WuXi AppTec, Samsung Biologics, Recipharm, Siegfried, Cambrex, Evotec, Charles River, Boehringer Ingelheim, Fujifilm Diosynth, Piramal Pharma, Alcami, Jubilant Biosys.
The Europe Plug-and-Play CDMO Market size is USD 2.38 Billion in 2025.
The Europe Plug-and-Play CDMO Market CAGR is 21.43%.
- Lonza
- Catalent
- Thermo Fisher
- WuXi AppTec
- Samsung Biologics
- Recipharm
- Siegfried
- Cambrex
- Evotec
- Charles River
- Boehringer Ingelheim
- Fujifilm Diosynth
- Piramal Pharma
- Alcami
- Jubilant Biosys
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)