Europe Plasmid DNA Manufacturing Market Size & Forecast:
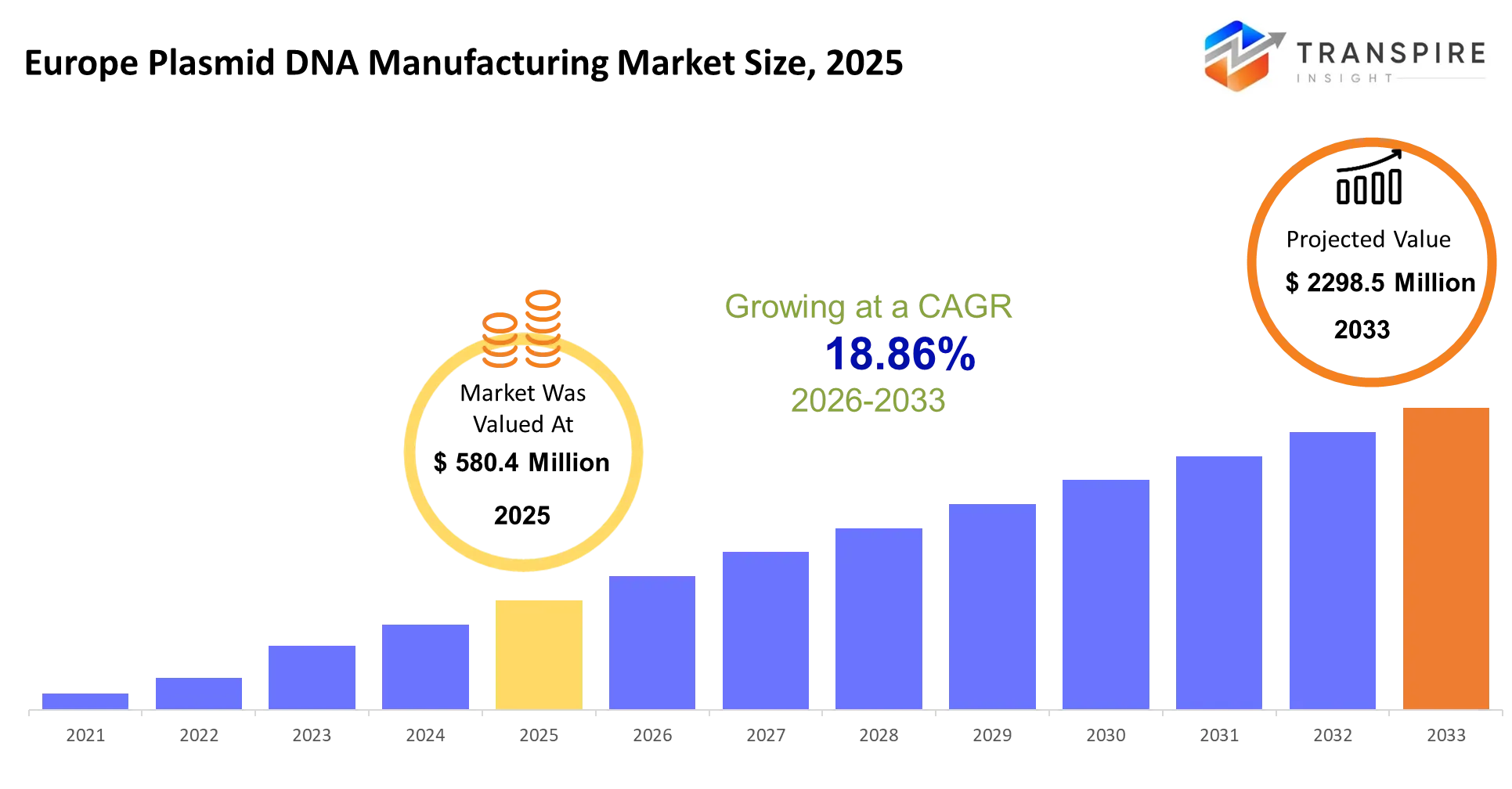
- Europe Plasmid DNA Manufacturing Market Size 2025: USD 580.4 Million
- Europe Plasmid DNA Manufacturing Market Size 2033: USD 2298.5 Million
- Europe Plasmid DNA Manufacturing Market CAGR: 18.86%
- Europe Plasmid DNA Manufacturing Market Segments: By Type (Plasmid DNA Production, Purification Services, CDMO Services, Others), By Application (Gene Therapy, Vaccine Development, Cell Therapy, Research, Others), By End-User (Biotech Firms, Pharma Companies, Research Institutes, CDMOs, Others), By Scale (Preclinical, Clinical, Commercial, Others).
To learn more about this report, Download Free Sample Report
Europe Plasmid DNA Manufacturing Market Summary:
The Europe Plasmid DNA Manufacturing Market size is estimated at USD 580.4 Million in 2025 and is anticipated to reach USD 2298.5 Million by 2033, growing at a CAGR of 18.86% from 2026 to 2033. The Europe plasmid DNA manufacturing market provides essential genetic material which enables the creation of gene therapies and mRNA vaccines and advanced biologics development. The system enables biotech companies to produce their required plasmid DNA products through its system which delivers high-quality plasmid DNA essential for their clinical and commercial operations.
The market has developed from producing small research batches to manufacturing large-scale products which meet GMP standards during the last 3 to 5 years. The development of cell and gene therapy pipelines has accelerated this industry evolution toward which this transition leads. The COVID-19 pandemic forced governments and biotech companies to develop local manufacturing facilities because the outbreak revealed existing capacity limitations.
Companies establish permanent supply agreements while they develop their internal production capabilities. The industry experiences revenue growth because European biopharma companies now recognize plasmid DNA as an essential production material instead of a specialized product.
Key Market Insights
- Germany controls more than 25% of the European Plasmid DNA Manufacturing Market because its strong biopharmaceutical infrastructure and research and development funding support the industry.
- The UK will become the fastest developing region until 2030 because it invests in advanced therapies and benefits from regulatory changes which occurred after Brexit.
- The market for GMP-grade plasmid DNA production achieves its leading position in 2024 by capturing approximately 60% of total market share which results from growing demand for commercial therapeutic products.
- Research-grade plasmid DNA holds the second-largest share due to ongoing preclinical and academic research activities.
- Custom plasmid DNA services emerge as the most rapidly expanding business sector because biotech companies choose to outsource their complex vector development needs until 2030.
- The gene therapy application field holds more than 45% market share in 2024 because of its late-stage clinical pipeline products and ongoing development of advanced treatment options.
- The market for mRNA vaccine development has experienced its fastest growth because the technology moved beyond COVID-19 to create applications in oncology and infectious diseases.
- The biopharmaceutical industry controls almost 55% of the market in 2024 because companies use plasmid DNA to produce therapies and develop their product pipelines.
- The growth rate of Contract Research and Manufacturing Organizations (CROs/CMOs) outpaces all other end-user sectors because companies expand their outsourcing efforts to overcome their production capacity challenges.
What are the Key Drivers, Restraints, and Opportunities in the Europe Plasmid DNA Manufacturing Market?
The Europe Plasmid DNA Manufacturing Market experiences its primary growth driving force because of the fast development of both gene therapy and mRNA-based treatment systems. The pandemic established mRNA platforms through its actual deployment, which provided proof for advanced therapy systems to receive official approval. The developers need to obtain all necessary materials for their upcoming trials because their research projects now handle extended testing periods. The need for plasmid DNA results in higher contract manufacturing income, which motivates biopharma companies to establish permanent supply contracts, thus driving facility development throughout all European nations.
The existing industrial systems face a fundamental obstacle because there is not enough GMP-certified manufacturing facilities which can handle production at industrial scale. The creation of these facilities needs extensive financial resources, specialized knowledge, and complete adherence to all relevant rules, which makes it difficult to expand their operations. The biotech industry experiences production delays and extended development periods which lead to increased operating expenses. The smaller companies in the market experience clinical development delays, which results in reduced market income while preventing the widespread acceptance of plasmid-based treatments.
The adoption of continuous and cell-free plasmid DNA manufacturing technologies creates a significant business opportunity. Countries such as Germany and the Netherlands are currently establishing synthetic biology platform investments which will accelerate their ability to produce products at industrial scales. Enzymatic DNA synthesis methods enable manufacturers to create their products within shorter timeframes, which allows them to satisfy rising clinical and commercial needs while boosting profit margins.
What Has the Impact of Artificial Intelligence Been on the Europe Plasmid DNA Manufacturing Market?
The use of artificial intelligence and advanced digital technologies has begun to change plasmid DNA production methods in European laboratories through the establishment of better process controls and improved stability of production batches. Bioreactors use AI-based control systems to monitor pH levels and dissolved oxygen levels and nutrient feed systems in real-time. The system adjusts these parameters to create the ideal conditions for maximum plasmid production. Operators use machine learning models to study past batch results which enables them to identify potential quality issues before they become major problems. The system achieves better production outcomes because it decreases batch rejection rates and enhances facility performance which leads to increased production capacity at GMP facilities.
Predictive capabilities extend to equipment performance as well. Maintenance requirements are predicted through algorithms that monitor fermentation and purification system performance which decreases unexpected equipment failures while extending system lifespan. The production line digital twins create virtual models of manufacturing processes which assist manufacturers in developing their production scaling solutions while decreasing their required validation time.
The implementation of these enhancements produces outcomes that deliver more dependable results while decreasing production expenses and enabling quicker delivery of clinical-grade materials. The implementation process faces challenges because essential integration expenses remain excessive and standardized data sets continue to provide insufficient coverage. The majority of facilities still depend on outdated systems which creates challenges for AI implementation and test facilities because they experience operational advantages from AI technologies.
Key Market Trends
- The biopharmaceutical industry adopted multi-year supply contracts which they established to secure plasmid DNA production capacity since 2020 because they wanted to prevent shortages during clinical and commercial development phases.
- The post-pandemic period saw both governments and private investors increase their financial support for regional GMP facilities which enabled European suppliers to reduce their reliance on US suppliers while building stronger supply chain systems.
- Between 2021 and 2024, CDMOs expanded aggressively and Lonza Group together with Cobra Biologics increased their plasmid production capacity to meet late-stage pipeline requirements.
- Manufacturers adopted high-density fermentation technologies which enabled them to produce more plasmids with lower production costs compared to traditional methods.
- The biotech industry started outsourcing its complex plasmid design and production work to external partners since 2022 because they wanted to decrease time-to-clinic and reduce their capital needs.
- The approval of advanced therapies led regulatory bodies to raise their GMP compliance standards which forced smaller companies to either upgrade their facilities or stop producing clinical-grade supplies.
- The introduction of mRNA platforms for COVID-19 treatment created new demand patterns which required increased plasmid DNA production for various therapeutic uses including oncology and rare diseases.
- Digitalization trends emerged after 2021 because manufacturers started using real-time monitoring and analytics systems to decrease batch failures and create more reliable processes.
- Plasmid producers and gene therapy developers established more strategic partnerships which allowed them to develop products together while synchronizing production schedules with clinical development timelines.
Europe Plasmid DNA Manufacturing Market Segmentation
By Type
The dominant method of producing plasmid DNA according to its type exists because it directly supports therapeutic production while maintaining necessary GMP-grade standards. The purification services process functions as an essential procedure that establishes both regulatory compliance and product quality standards which apply to clinical use. CDMO services have gained strong momentum as biotech firms shift toward outsourcing complex production processes to reduce capital burden and accelerate timelines.
The current demand patterns demonstrate that clients now select integrated service offerings instead of using fragmented supply models because they want complete solutions that cover vector design through to final delivery. Providers face two main factors which determine their ability to expand through capacity development and regulatory requirements. Manufacturers who establish production and purification processes within one system will achieve better contract results which help them build lasting partnerships with their customers.
By Application
The application of gene therapy has become the principal treatment method because research studies for advanced clinical trials need substantial amounts of top-grade plasmid DNA. Vaccine development remains a significant segment, especially with continued expansion of mRNA platforms into oncology and infectious diseases beyond pandemic use cases. The research field and cell therapy applications generate ongoing demand, which occurs at lower rates than commercial product development.
Production capabilities develop according to specific requirements of each application because gene therapy develops production capacity while vaccines create production capacity and operational flexibility. The production process needs manufacturers to adjust their operations according to different production quantities and the requirements of various regulatory bodies. The organization needs to achieve two goals for upcoming expansion which require them to develop multiple treatment methods while maintaining their operational efficiency and legal requirements.
By End-User
Biopharmaceutical companies maintain their leadership position through direct drug development and commercialization activities because these activities create ongoing need for both clinical and commercial-grade plasmid DNA. The fastest-growing industry sector includes CDMOs because companies choose to outsource their operations as a viable option which provides them operational flexibility and quicker project delivery.
Research institutes and smaller biotech firms contribute to early-stage demand but often lack the infrastructure for large-scale production. Companies have started to outsource their operations because this practice helps them achieve better capital distribution while they concentrate on their main business activities. Service providers who can develop scalable systems and provide regulatory knowledge will establish enduring business relationships as competition becomes more intense. The existing trend indicates that manufacturing partners will become less numerous while their capabilities will increase through supply base consolidation.
By Scale
The majority of pipelines remain in Phase I to Phase III stages which require ongoing production of clinical-scale manufacturing. Preclinical production supports early research but generates lower revenue due to smaller batch sizes and less stringent requirements. The current demand for validated production systems arises from the rapid growth of commercial-scale manufacturing which needs to produce high volumes of approved therapies.
The process of moving from clinical operations to commercial production creates difficulties with maintaining process consistency and managing expenses. The early investment in scalable platforms will provide manufacturers with a competitive edge during the process of bringing new therapies to market. The industry will experience a capacity planning transformation which will create a demand for standard production methods that use high-throughput processes.
What are the Key Use Cases Driving the Europe Plasmid DNA Manufacturing Market?
The primary application which drives plasmid DNA production throughout Europe remains gene therapy. The manufacturing process for vector production requires extensive quantities of GMP-grade plasmid DNA which supports late-stage clinical programs and approved advanced therapies. The highest demand for this use case arises because therapy batches require identical genetic templates which bind plasmid availability to both business revenue and patient treatment schedules.
The biopharmaceutical industry and CDMOs expand their operations to develop mRNA vaccines and cell therapy applications. Oncology mRNA pipelines and CAR-T therapies need flexible plasmid design which allows fast production of their required materials. The segments continue to grow as companies expand their product range from vaccines developed during the pandemic toward personalized and targeted treatments.
The emerging use cases enable development of DNA-based vaccines while advancing in vivo gene editing through CRISPR and other platforms. European early-stage trials demonstrate possibilities for scalable therapies which depend on next-generation plasmid constructs and advanced synthesis methods.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 580.4 Million |
|
Market size value in 2026 |
USD 685.8 Million |
|
Revenue forecast in 2033 |
USD 2298.5 Million |
|
Growth rate |
CAGR of 18.86% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Lonza, Thermo Fisher, Aldevron, Catalent, WuXi AppTec, GenScript, BioNTech, Merck, Takara Bio, Cytiva, Cobra Biologics, Vigene Biosciences, AGC Biologics, Oxford Biomedica, Samsung Biologics |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Plasmid DNA Production, Purification Services, CDMO Services, Others), By Application (Gene Therapy, Vaccine Development, Cell Therapy, Research, Others), By End-User (Biotech Firms, Pharma Companies, Research Institutes, CDMOs, Others), By Scale (Preclinical, Clinical, Commercial, Others) |
Which Regions are Driving the Europe Plasmid DNA Manufacturing Market Growth?
The market is dominated by Western Europe which shows strong biopharma research and GMP production capabilities centered in Germany and the United Kingdom and Switzerland. The European Medicines Agency employs strict regulatory standards which enable faster approval processes for advanced therapies that need plasmid DNA. The region possesses established CDMO networks and trained workers and its location near major biotech centers. The integrated ecosystem enables companies to move from clinical trials to full-scale production while they maintain their local supply chain operations which strengthens the area's economic dominance.
Northern European countries maintain constant economic contributions through their focus on process improvements and operational efficiency according to Denmark Sweden and the Netherlands. The area concentrates on producing high-quality specialized products through efficient manufacturing processes that include automated systems which Western Europe uses for its industrial output. Research institutes and new biotech companies create steady demand through government-supported life sciences initiatives and ongoing funding programs. Northern Europe provides a stable platform for conducting both long-term contracts and targeted plasmid development work.
The fastest expansion in Europe is occurring in Southern and Eastern Europe because of fresh investments which have built biotech facilities and established affordable manufacturing capabilities. Spain and Poland have developed their GMP facilities while providing tax incentives to attract international CDMO companies. European Union funding has increased which has resulted in decentralized advanced therapy production through cooperation among member states. The region will experience growth which will create chances for new businesses that want to reduce their operational expenses while it becomes a vital expansion area until 2033.
Who are the Key Players in the Europe Plasmid DNA Manufacturing Market and How Do They Compete?
The Europe Plasmid DNA Manufacturing Market shows moderate competition because a few major CDMOs maintain most of the valuable GMP production capacity while niche markets and early-stage segments create opportunities for smaller specialized companies. Incumbents maintain market share because their business size and regulatory history and their extended agreements protect them from new market entrants who bring production methods based on adaptable technological solutions. The primary basis of competition has shifted from cost to integrated service capability, where clients prefer partners that can handle design, production, and purification under one platform. Companies establish their geographical presence near biotech hubs because they want to decrease logistical risks and achieve faster project completion.
Lonza Group operates its business through large-scale production of GMP-standard manufacturing while using its expertise to support both late-stage and commercial gene therapy development. The company invests continuously in its European facilities to enhance its operational capabilities and obtain contracts that last multiple years. Thermo Fisher Scientific uses its complete service model to connect comprehensive plasmid manufacturing operations with additional biologics and analytical functions, which helps clients who handle complex product development processes.
Merck KGaA focuses its research efforts on developing new technologies which enhance its capacity to produce and purify high-yield plasmid systems through faster production times and reliable product quality. Cobra Biologics uses its specialized knowledge of viral vector and plasmid development to create customized solutions which attract early-stage biotech companies. Aldevron builds its business through partnerships with gene therapy developers which enable the company to match its production capabilities with clinical needs while obtaining predictable customer demand for an extended period.
Company List
- Lonza
- Thermo Fisher
- Aldevron
- Catalent
- WuXi AppTec
- GenScript
- BioNTech
- Merck
- Takara Bio
- Cytiva
- Cobra Biologics
- Vigene Biosciences
- AGC Biologics
- Oxford Biomedica
- Samsung Biologics
Recent Development News
In May 2026, NunaBio secures £6.5M to scale DNA manufacturing platform: Newcastle University spinout NunaBio secured £6.5 million funding to scale its next-generation DNA manufacturing platform. The investment focuses on improving rapid, high-quality synthetic DNA production for applications including gene therapy and vaccine development, addressing key manufacturing bottlenecks in Europe’s biotech ecosystem.
Source: https://www.sunderland.ac.uk
In April 2026, 4basebio announces major expansion to support the growing demand of RUO and HQ synthetic DNA with lease of an innovation hub and manufacturing facility in Cambridge, UK : 4basebio announced a major expansion in Cambridge with a new innovation hub and GMP-focused manufacturing facility. The site will increase capacity for high-quality synthetic DNA used in gene and cell therapies, mRNA vaccines, and genome editing programs, strengthening Europe’s advanced nucleic acid production base.
Source: https://pharmapress.co.uk
What Strategic Insights Define the Future of the Europe Plasmid DNA Manufacturing Market?
The Europe Plasmid DNA Manufacturing Market is moving toward industrial-scale, platform-driven production over the next 5–7 years because gene and mRNA therapies have moved from clinical testing to reliable commercial production. Systematic plasmid backbone standardization together with increased adoption of modular production systems will achieve two main results. The manufacturing sector will benefit from this development because it enables companies to run extensive automated operations which maintain regulatory standards across different regions.
The emerging synthesis and cell-free DNA technologies present a hidden threat which will decrease reliance on established fermentation processes. The upcoming technologies will disrupt conventional plasmid production methods when they achieve their required production capacity and economic feasibility before the expected timeline.
The development of enzymatic DNA synthesis platforms presents a major business prospect for innovation centers located in Germany and the Netherlands because these two countries provide financial support together with advanced technical knowledge. Companies need to invest in hybrid manufacturing systems which merge traditional DNA production methods with upcoming DNA production technologies to maintain their market position while safeguarding themselves from technological advancements.
Europe Plasmid DNA Manufacturing Market Report Segmentation
By Type
- Plasmid DNA Production
- Purification Services
- CDMO Services
- Others
By Application
- Gene Therapy
- Vaccine Development
- Cell Therapy
- Research
- Others
By End-User
- Biotech Firms
- Pharma Companies
- Research Institutes
- CDMOs
- Others
By Scale
- Preclinical
- Clinical
- Commercial
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Plasmid DNA Manufacturing Market size for the Market will be USD 2298.5 Million in 2033.
The key Segments of the Europe Plasmid DNA Manufacturing Market are By Type (Plasmid DNA Production, Purification Services, CDMO Services, Others), By Application (Gene Therapy, Vaccine Development, Cell Therapy, Research, Others), By End-User (Biotech Firms, Pharma Companies, Research Institutes, CDMOs, Others), By Scale (Preclinical, Clinical, Commercial, Others).
Major Players in the Europe Plasmid DNA Manufacturing Market are Lonza, Thermo Fisher, Aldevron, Catalent, WuXi AppTec, GenScript, BioNTech, Merck, Takara Bio, Cytiva, Cobra Biologics, Vigene Biosciences, AGC Biologics, Oxford Biomedica, Samsung Biologics.
The Current Market size of the Europe Plasmid DNA Manufacturing Market is USD 580.4 Million in 2025.
The Europe Plasmid DNA Manufacturing Market CAGR is 18.86%.
- Lonza
- Thermo Fisher
- Aldevron
- Catalent
- WuXi AppTec
- GenScript
- BioNTech
- Merck
- Takara Bio
- Cytiva
- Cobra Biologics
- Vigene Biosciences
- AGC Biologics
- Oxford Biomedica
- Samsung Biologics
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


