Europe Life Science Logistics Market Size & Forecast:
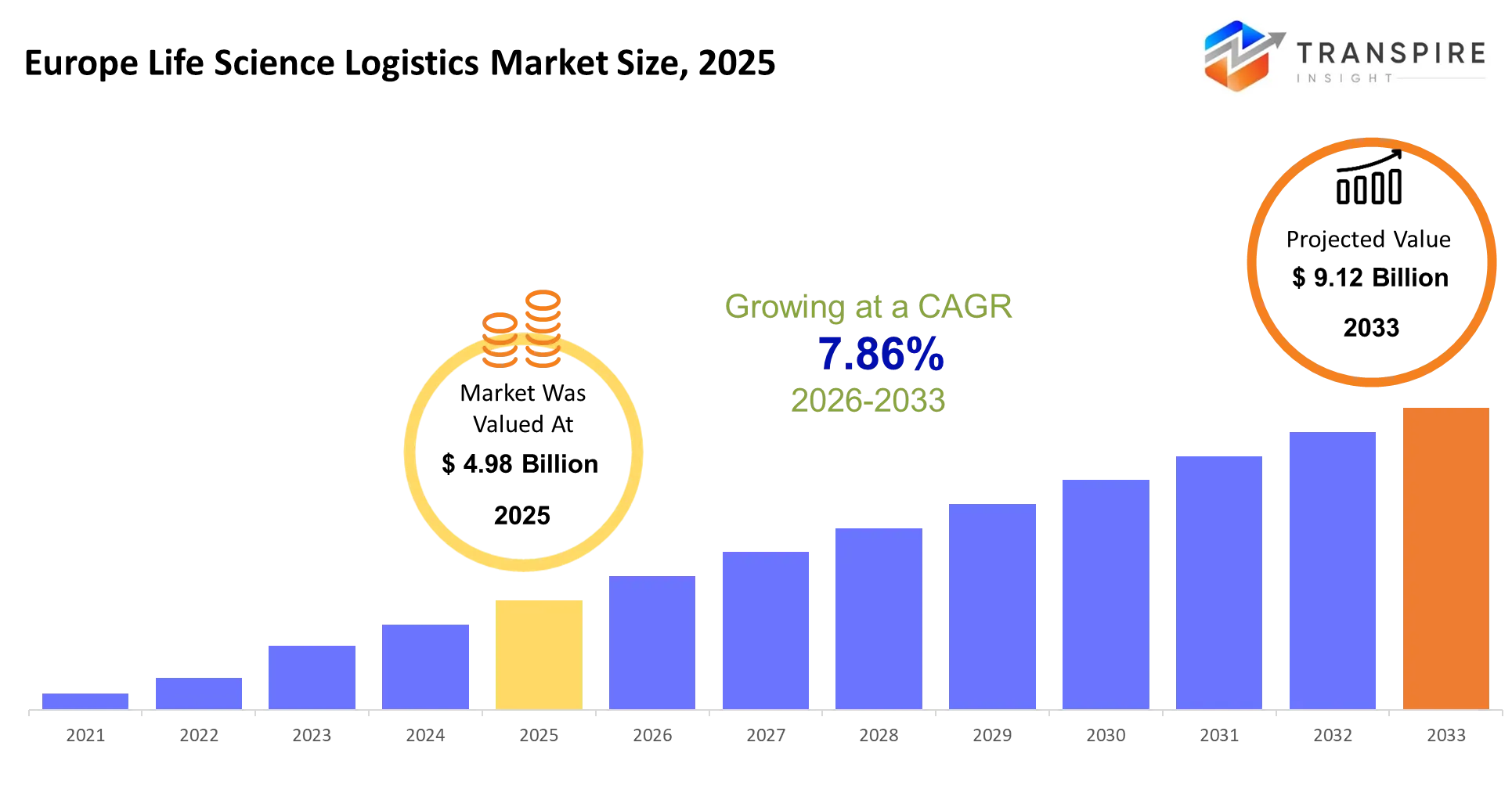
- Europe Life Science Logistics Market Size 2025: USD 4.98 Billion
- Europe Life Science Logistics Market Size 2033: USD 9.12 Billion
- Europe Life Science Logistics Market CAGR: 7.86%
- Europe Life Science Logistics Market Segments: By Type (Cold Chain Logistics, Non-cold Chain, Others); By Application (Pharmaceuticals, Biologics, Clinical Trials, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Mode (Air, Sea, Road, Others)
To learn more about this report, Download Free Sample Report
Europe Life Science Logistics Market Summary
The Europe Life Science Logistics Market was valued at USD 4.98 Billion in 2025. It is forecast to reach USD 9.12 Billion by 2033. That is a CAGR of 7.86% over the period.
The Europe Life Science Logistics Market operates medical supply routes which deliver temperature-controlled medicines and biologics and vaccines and clinical trial equipment through secure supply chains that protect product safety and traceability and operational efficacy from manufacturing facilities to medical centers and research facilities.The sector has progressed through five years toward implementing digital cold chain systems while EU Good Distribution Practice now requires more stringent compliance because biologics and personalized therapies demand ultra-low temperature shipping with live monitoring.
The COVID-19 pandemic created a trigger which demonstrated weaknesses in international vaccine distribution systems and needed the quick expansion of approved cold storage facilities while later European geopolitical conflicts interrupted international transport routes and required companies to establish inventory collection points nearer to their main customer base.Pharmaceutical companies need to outsource more transportation work to logistics experts because operational challenges and regulatory requirements have increased their business expenses which will result in higher costs for temperature-sensitive products.
Key Market Insights
- Western Europe maintains a 48% market share of the European Life Science Logistics Market because of its robust pharmaceutical production capacity and its sophisticated cold chain distribution systems.
- The distribution network of Germany, France, and the Netherlands functions as their main operational center which supports their intensive clinical research and biotechnology activities.
- Eastern Europe between 2025 and 2032 will become its most rapidly developing area because of its affordable logistics outsourcing solutions and its growing healthcare accessibility.
- Temperature-controlled transportation accounts for 55 of the market share because vaccines and biologics and cell and gene therapy shipments need this service.
- The second largest market segment consists of warehousing services which enable pharmaceutical companies to handle their inventory needs for large-scale operations.
- The adoption of IoT-based real-time tracking systems has made cold chain monitoring solutions into the most rapidly expanding market segment.
- The pharmaceutical industry accounts for approximately 60 of all application requirements in the Europe Life Science Logistics Market because of its substantial drug transportation needs.
- The application segment of clinical trials logistics experiences the most significant growth because of both decentralized trials and international research activities.
- Pharmaceutical companies remain the leading end users with approximately 50 market share because they depend on third-party logistics companies for most of their distribution needs.
- Biotechnology companies represent the most rapidly expanding end-user segment because they are developing numerous new advanced therapies through their research pipelines.
What are the Key Drivers, Restraints, and Opportunities in the Europe Life Science Logistics Market?
The rapid expansion of biologics vaccines and cell and gene therapies requires the most intense logistics operations because these products need to be transported under strict temperature control. The introduction of advanced therapies through commercial distribution established the current system but the distribution of COVID-19 vaccines demonstrated the urgent need for dependable cold chain solutions. Pharmaceutical companies currently drive their business growth by increasing their spending on GDP-certified cold storage facilities and real-time tracking technologies and dedicated transport vehicles which deliver value through both contract agreements and ongoing business partnerships
The European regulatory system operates as a major structural barrier because different countries maintain distinct customs codes and documentation rules which create delays for pharmaceutical products moving between European nations. The healthcare logistics market cannot reach a resolution because EU countries and non-EU neighboring markets enforce different regulations about their logistics operations. The result leads to increased administrative expenses and extended delivery times which restrict the growth of European logistics systems while operational efficiency improvements fail to match the demand that keeps increasing.
The combination of AI-powered predictive cold chain optimization and regional manufacturing decentralization creates a significant future market opportunity. Pharmaceutical companies establish micro-distribution centers in Central and Eastern Europe to decrease their reliance on long-distance shipping. The combination of IoT-based temperature monitoring with predictive analytics enables organizations to establish distribution systems that operate safely at higher speeds while simultaneously decreasing their risk of product loss throughout the European Life Science Logistics Market distribution network.
What Has the Impact of Artificial Intelligence Been on the Europe Life Science Logistics Market?
The life science logistics system of Europe undergoes operational process changes because advanced digital systems and artificial intelligence technology continuously reshape its work procedures which especially applies to its temperature-controlled transportation methods that require exact delivery tracking.
AI-based control systems now serve as monitoring tools for cold chain equipment because they perform real-time temperature validation of temperature excursions while producing compliance reports that meet EU GDP requirements. Fleet management systems use AI technology to assess how well vehicles travel their assigned routes while tracking driver performance and fleet compliance with regulations which decreases the need for manual checks during international pharmaceutical shipments.
Predictive capabilities have become a primary factor that distinguishes one organization from another. Machine learning models use historical shipment data with sensor inputs to predict when refrigeration units will fail and to determine optimal energy use in warehouses while forecasting delays that result from customs and weather issues. The systems demonstrated their capability to increase equipment uptime from 10 to 20 percent and decrease unplanned maintenance expenses while enhancing on-time delivery rates for high-value biologics.
Logistics providers achieve demonstrable improvements in fuel efficiency through their ability to select the best delivery routes which results in fewer instances of temperature-sensitive products spoiling and creates better service results while decreasing total shipping expenses.
European logistics providers face obstacles to adoption because their existing systems use outdated IT infrastructure that requires expensive integration work. The deployment of predictive analytics in cross-border life science supply chains faces delays because operators deal with limited compatibility between their IoT devices and cloud systems.
Key Market Trends
- Logistics providers have achieved better shipment visibility throughout Europe because they replaced manual tracking methods with IoT systems for cold chain monitoring since 2020.
- The EU GDP compliance audits which started in 2021 required carriers to spend money on certified temperature-controlled systems until the end of 2025.
- Pharmaceutical companies now outsource more than 30% of their work since 2019 which has changed competitive relationships between businesses within the European Life Science Logistics Market.
- The adoption of AI-based predictive maintenance tools by major companies such as DHL and Kuehne+Nagel resulted in decreased cold chain failures.
- The expansion of clinical trial logistics networks after 2022 occurred because decentralized trial models needed cross-border sample transport coordination.
- The vaccine distribution infrastructure which medical organizations developed during the COVID-19 pandemic now handles biologics and mRNA therapies which results in better long-term asset utilization.
- Since 2023, digital twin technology adoption has increased which allows organizations to model temperature-sensitive shipment routes before actual execution.
- Eastern Europe logistics hubs became more popular because companies started to move their operations to this region after 2022 because they wanted to avoid Western European traffic and price problems.
- The implementation of real-time emissions monitoring systems increased after 2021 because logistics operations needed to comply with EU sustainability reporting requirements.
- Firms that adopted end-to-end visibility platforms experienced competitive pressure because these systems enabled them to decrease delays while enhancing their service-level agreements throughout Europe.
Europe Life Science Logistics Market Segmentation
By Type:
The distribution of life science products in Europe relies on cold chain logistics which controls more than 50 percent of all handling operations because of the temperature requirements needed for biologics and vaccines and advanced therapies. Businesses need to keep their products safe throughout the extended distance between borders because even tiny temperature changes will result in product damages and fines from authorities. Non-cold chain logistics maintains its existing market share, which supports packaging materials and non-sensitive pharmaceutical products.
The mRNA-based therapies and specialty biologics together with their requirements for ultra-low temperature storage and continuous monitoring have produced stronger growth in cold chain logistics. The development of non-cold chain services progresses at a slower rate because product volume increases which result in products moving into temperature-controlled categories. The European Union Good Distribution Practice regulations have increased the necessity for cold chain procedures which now exist in all important pharmaceutical distribution routes.
The need for precise therapies will drive cold chain logistics to achieve greater market share during the projected period. Investors and logistics providers are investing resources into refrigerated systems and monitoring technologies which operate in real time while the non-cold chain sectors experience market standardization and decreasing price levels.
To learn more about this report, Download Free Sample Report
By Application:
The largest application segment of the market is pharmaceuticals which develops through the ongoing movement of high-volume drugs between manufacturing sites and distribution centers and European hospitals. The established prescription drug supply chains maintain their market dominance because they need ongoing restocking and require strict compliance with their operational procedures.
The market for biologics products is currently experiencing rapid growth because advanced therapies are gaining regulatory approval and their distribution needs special handling requirements.
The application of clinical trial logistics has developed into its most rapidly expanding sector because decentralized trial models and international research activities have increased. Highly controlled logistics operations are required to move delicate research materials and testing medicines from one research facility to another. Healthcare businesses depend on niche applications to operate their essential operations which include diagnostic materials.
The future business growth will be supported by biologics and clinical trials because pharmaceutical companies are now developing customized medical solutions. The logistics sector will benefit from cold chain systems and quick delivery systems because they create new competitive advantages for businesses that invest in these technologies. The traditional pharmaceutical logistics business will produce steady income but it will grow at a lower rate than the adaptive cold chain system business.
By End-User:
Pharmaceutical companies dominate end-user demand in Europe’s life science logistics ecosystem, controlling roughly half of total outsourced logistics spending. The company maintains its leadership position through its extensive drug production capabilities and international distribution networks that need transportation systems to follow compliance regulations. The cell and gene therapy market is growing because biotech companies are emerging as a smaller but rapidly expanding business sector.
Research institutes maintain a continuous need for resources because they handle clinical trial materials and conduct academic collaborative research. The company requires special logistics solutions to handle its medical operations which operate on smaller volume than pharmaceutical companies do. Diagnostic laboratories and other end users help to create specialized medical supply chains which function in niche areas of the market.
Future growth will shift toward biotech firms as pipeline complexity increases and outsourcing becomes more common. Investors are focusing on flexible logistics providers who can handle small-batch shipments of high-value items while pharmaceutical companies work to improve efficiency through long-term agreements and combined supply chain solutions.
By Mode:
Air transport dominates high-value pharmaceutical logistics because it provides quick delivery and maintains strict temperature control for biologics and emergency medical shipments. This mode is preferred for long-distance, time-sensitive deliveries, especially for clinical trial materials and vaccines. Road transport remains essential for intra-European distribution, supporting last-mile connectivity between manufacturing hubs and healthcare facilities.
Sea freight operates as a limited transportation method which provides cost-effective bulk pharmaceutical ingredient shipping for secure transit of non-urgent shipments. Its slower transit time restricts its use in temperature-sensitive applications, but it remains relevant for cost optimization in upstream supply chains. Companies increasingly select rail and other transportation methods to develop their environmentally friendly logistics solutions.
Over the forecast period, air and road transport will continue to dominate, while sustainability pressures encourage modal shifts toward lower-emission options. Logistics providers who establish hybrid routing systems and operate low-carbon vehicle fleets will achieve competitive benefits because European emissions regulations will strengthen.
What are the Key Use Cases Driving the Europe Life Science Logistics Market?
The Europe Life Science Logistics Market exists because pharmaceutical distribution needs to maintain complete cold chain operations for temperature-sensitive medications which must be transported from manufacturing facilities to hospitals and pharmacies. The transportation industry needs to preserve approved storage facilities and tracking systems and shipping methods because European Union Good Distribution Practice standards and increasing demand for biological products create essential requirements for transportation services.
The European market for clinical trial logistics and biotechnology shipment services experiences rapid growth because research activities and decentralized trial models become more widespread. Biotech companies choose to work with specialized courier services for shipping biological samples which have high market value and need immediate international delivery with exact temperature maintenance. The hospital supply system for advanced therapy medications continues to develop into a more vital component which now requires special attention in this area.
New use cases have emerged through personalized medicine delivery systems and cell and gene therapy logistics which involve transporting materials at ultra-low temperatures while providing specific delivery methods to patients. Special logistics companies are establishing new delivery systems through their development of direct-to-patient networks which will operate under fresh regulatory requirements for home treatment.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 4.98 Billion |
|
Market size value in 2026 |
USD 5.37 Billion |
|
Revenue forecast in 2033 |
USD 9.12 Billion |
|
Growth rate |
CAGR of 7.86% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
DHL, FedEx, UPS Healthcare, Kuehne+Nagel, DB Schenker, CEVA Logistics, AmerisourceBergen, Cardinal Health, Marken, World Courier, Agility Logistics, Nippon Express, Bollore Logistics, Expeditors, Sinotrans |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Cold Chain Logistics, Non-cold Chain, Others); By Application (Pharmaceuticals, Biologics, Clinical Trials, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Mode (Air, Sea, Road, Others) |
Which Regions are Driving the Europe Life Science Logistics Market Growth?
The life science logistics market in Europe has its main center in Western Europe which is operated by Germany, France, and the Netherlands. The region maintains its dominant position because it has strong pharmaceutical manufacturing facilities and extensive hospital networks and it enforces EU GDP standards with strict compliance. The advanced warehousing and multimodal connectivity at Rotterdam and Frankfurt major logistics centers enable them to handle large quantities of temperature-sensitive materials between different modes of transport. The business operations of the organization run efficiently because of the established network of contract logistics vendors and their connected IT systems and official cold storage facilities.
Southern Europe contributes a stable and steadily expanding share, driven by Spain and Italy. The region depends on its distribution of imported goods and its healthcare supply chains which serve local markets. The city of Barcelona and the city of Genoa both provide strong port infrastructure which supports the companys growth while the company works toward achieving EU pharmaceutical logistics compliance. The region develops into a reliable secondary cross-border pharmaceutical transport route because ongoing investments into road and coastal shipping networks create dependable transport connections.
Eastern Europe becomes the fastest developing area because Western Europe pharmaceutical companies increase their outsourcing operations while the region builds new infrastructure facilities. The countries of Poland Hungary and the Czech Republic have built new GDP-compliant warehouses which they use to establish cold chain corridors since 2022. EU funding for transport modernization and lower operational costs have attracted logistics relocation from congested Western hubs.This shift creates strong entry opportunities for logistics providers and investors targeting scalable warehousing, regional distribution centers, and cost-efficient clinical trial logistics over the 2026–2033 period.
Who are the Key Players in the Europe Life Science Logistics Market and How Do They Compete?
European Life Science Logistics Market competition displays moderate consolidation, as a few global integrators control valuable cold chain and pharmaceutical distribution operations, while local companies serve specific market segments. The incumbents maintain their market share through combined service offerings and digital tracking systems, which they use to compete against their rivals. The primary basis of competition has shifted toward temperature precision, regulatory compliance under EU GDP standards, and end-to-end visibility across cross-border pharmaceutical shipments. Existing providers maintain their market dominance because new companies face two obstacles, which include certification requirements and expensive infrastructure needs.
DHL Group uses technology-based logistics solutions to create IoT-enabled cold chain systems that enable real-time tracking of shipments containing biological and clinical trial materials. The company produces competitive advantages through its extensive European distribution center network and its early investment in facilities that meet GDP temperature control requirements. The company continues expanding through partnerships with pharmaceutical manufacturers and targeted acquisitions in life science logistics specialists to strengthen end-to-end control.
Kuehne+Nagel presents its unique value proposition through its temperature-controlled transportation solutions, which enable precise execution of air and road delivery operations for sensitive medical products. The KN PharmaChain service helps European transportation routes by improving compliance tracking and decreasing product waste. The company plans to grow through digital freight solutions and warehouse modernization projects, which will be implemented in locations that serve major pharmaceutical markets across Germany and Switzerland.
"The competition between FedEx Corporation and United Parcel Service (UPS) exists because both companies operate their global air networks and provide standard cold chain express services." The two companies make substantial investments in developing advanced tracking technologies while they build new healthcare logistics facilities throughout Europe to meet the increasing needs of biotech companies and clinical research organizations.
Company List
- DHL
- FedEx
- UPS Healthcare
- Kuehne+Nagel
- DB Schenker
- CEVA Logistics
- AmerisourceBergen
- Cardinal Health
- Marken
- World Courier
- Agility Logistics
- Nippon Express
- Bollore Logistics
- Expeditors
- Sinotrans
Recent Development News
In March 2025, DHL Group announced acquisition of CRYOPDP from Cryoport to strengthen its life sciences logistics division. The deal expands DHL’s specialty courier network for clinical trials, biopharma, and cell and gene therapy shipments, enhancing its cold chain and white-glove delivery capabilities across Europe and global markets.https://group.dhl.com
In April 2026, Kuehne+Nagel launched a new temperature-controlled airfreight cross-dock facility in Hyderabad to support pharmaceutical and healthcare logistics flows between Europe and Asia. The expansion enhances its global cold chain network and improves transit reliability for sensitive life science shipments.https://m.economictimes.com
What Strategic Insights Define the Future of the Europe Life Science Logistics Market?
The European Life Science Logistics Market is developing into cold chain systems which operate through complete digital systems that handle all aspects of their operations. The period between 5 to 7 upcoming years will generate its most valuable results through companies which deliver both temperature control and immediate data processing capabilities for their operations. The pharmaceutical supply chain is transitioning from its current system of separate transport operations toward complete network management.
The dependency on certified infrastructure operators presents a hidden danger because it creates price concentration risk for hubs which include Germany and the Netherlands when these hubs experience capacity limitations or operational system failures. The system faces disruptions to its cross-border operations which will occur during high-demand periods and times of intensified regulatory enforcement.
Eastern and Southern Europe present a new opportunity for businesses through their development of decentralized micro-fulfillment cold hubs, which receive support from the EU healthcare infrastructure projects that fund local biologics production facilities. The nodes operate by decreasing the chances of transit delays while achieving faster delivery times for high-value therapies.
The market participants need to focus on building modular cold chain systems which work with digital systems that allow them to expand their operations throughout different European regulatory markets while minimizing their risk of centralized operational delays.
Europe Life Science Logistics Market Report Segmentation
By Type
- Cold Chain Logistics
- Non-cold Chain
- Others
By Application
- Pharmaceuticals
- Biologics
- Clinical Trials
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Others
By Mode
- Air
- Sea
- Road
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Europe Life Science Logistics Market size is USD 9.12 Billion in 2033.
Key segments for the Europe Life Science Logistics Market are By Type (Cold Chain Logistics, Non-cold Chain, Others); By Application (Pharmaceuticals, Biologics, Clinical Trials, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Mode (Air, Sea, Road, Others).
Major Europe Life Science Logistics Market Players are DHL, FedEx, UPS Healthcare, Kuehne+Nagel, DB Schenker, CEVA Logistics, AmerisourceBergen, Cardinal Health, Marken, World Courier, Agility Logistics, Nippon Express, Bollore Logistics, Expeditors, Sinotrans.
The Current Europe Life Science Logistics Market size is USD 4.98 Billion in 2025.
The Europe Life Science Logistics Market CAGR is 7.86% from 2026 to 2033.
- DHL
- FedEx
- UPS Healthcare
- Kuehne+Nagel
- DB Schenker
- CEVA Logistics
- AmerisourceBergen
- Cardinal Health
- Marken
- World Courier
- Agility Logistics
- Nippon Express
- Bollore Logistics
- Expeditors
- Sinotrans
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


