Europe Clinical Trial Supply and Logistics Market Size & Forecast:
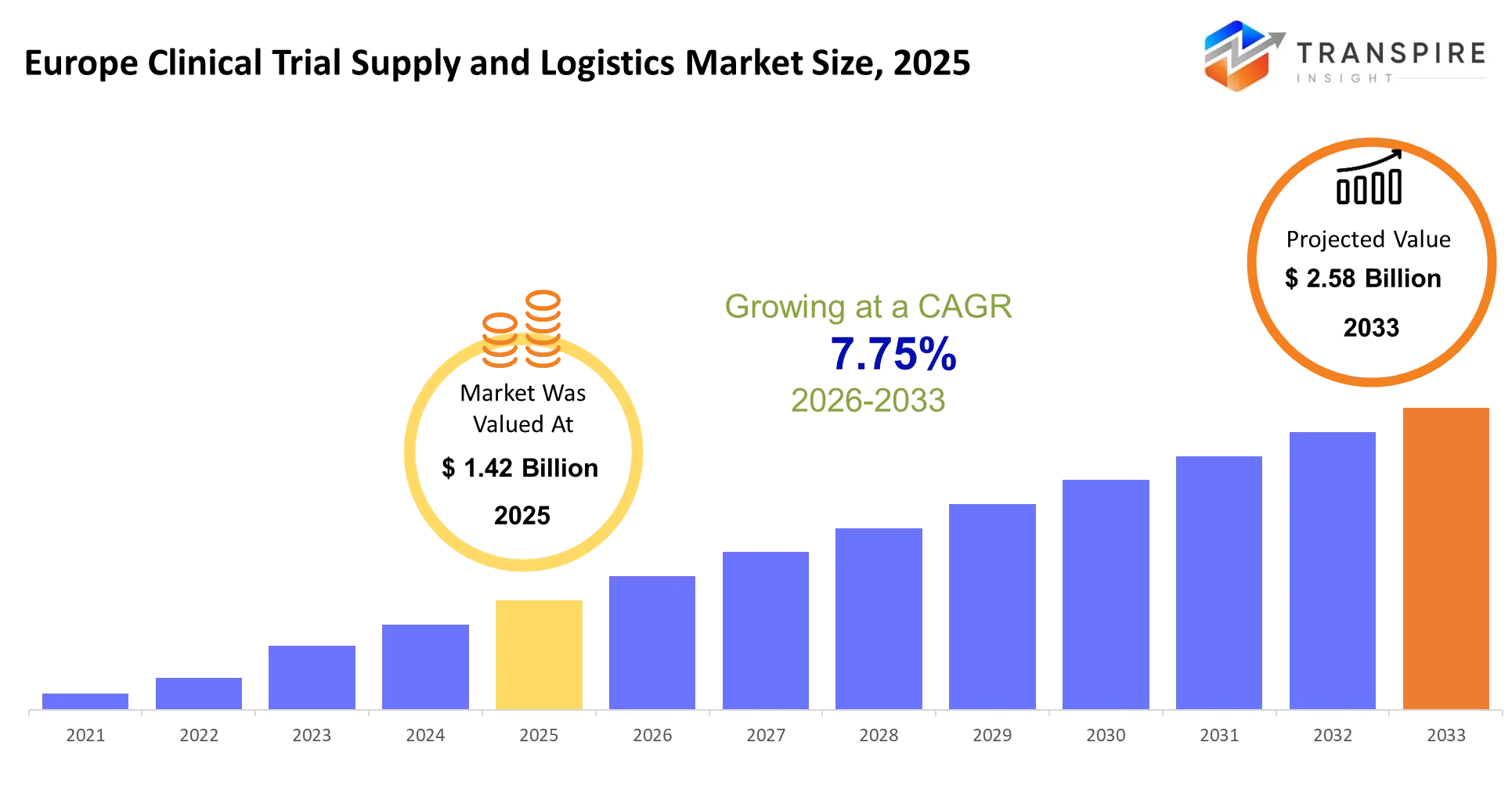
- Europe Clinical Trial Supply and Logistics Market Size 2025: USD 1.42 Billion
- Europe Clinical Trial Supply and Logistics Market Size 2033: USD 2.58 Billion
- Europe Clinical Trial Supply and Logistics Market CAGR: 7.75%
- Europe Clinical Trial Supply and Logistics Market Segments: By Type (Packaging, Distribution, Storage, Others), By Application (Phase I, Phase II, Phase III, Phase IV, Others), By End-User (Pharma Companies, CROs, Research Institutes, Others), By Service (Cold Chain, Non-cold Chain, Others).
To learn more about this report, Download Free Sample Report
Europe Clinical Trial Supply and Logistics Market Summary:
The Europe Clinical Trial Supply and Logistics Market size is estimated at USD 1.42 Billion in 2025 and is anticipated to reach USD 2.58 Billion by 2033, growing at a CAGR of 7.75% from 2026 to 2033. The Europe Clinical Trial Supply and Logistics Market operates to ensure that investigational drugs and biological samples and trial materials arrive at their designated locations according to scheduled times while maintaining required temperature controls and regulatory standards. The system operates to resolve coordination problems between sponsors and contract research organizations and trial sites because any delays and errors during the process will endanger both patient safety and data integrity.
The market needs direct-to-patient delivery systems together with flexible distribution networks because decentralized and hybrid trials have now become the standard operation since three to five years ago. The COVID-19 pandemic caused site access restrictions which created a need for organizations to implement remote trial logistics solutions.
Organizations make investments in cold chain infrastructure together with digital tracking systems and demand forecasting tools to support their operations. The increasing complexity of trials together with their expanded geographic reach requires logistics providers to implement advanced technology services which enable them to build strategic capabilities while generating new revenue opportunities.
Key Market Insights
- The Clinical Trial Supply and Logistics Market in Europe will be led by Western Europe which holds more than 45% of the market share in 2024 because of its strong pharmaceutical research and development facilities.
- The fastest growing area until 2030 will be Eastern Europe which provides affordable trial sites and develops its clinical research facilities.
- The distribution services and logistics operations together exceed 40% market share in 2024 because of the complicated global requirements needed for clinical trials.
- The storage and cold chain management system stands as the second most important part of the business because it enables the storage of biologics and temperature-sensitive medications.
- The packaging and labeling services operate as the fastest expanding business unit because the needs of personalized medicine and small-scale clinical trials force them to grow.
- The Phase III clinical trials account for 50% of clinical trial operations in 2024 because they involve multiple sites testing their medication on large groups of patients.
- The application of direct patient testing systems through decentralized and hybrid trials will experience the most rapid growth until 2030.
- Pharmaceutical and biotechnology companies lead with over 60% share in 2024 because their drug development activities encompass multiple medication research projects.
- The end-user segment that shows the most growth potential operates through Contract Research Organizations which develop their trial logistics outsourcing practices.
What are the Key Drivers, Restraints, and Opportunities in the Europe Clinical Trial Supply and Logistics Market?
The primary force accelerating the Europe Clinical Trial Supply and Logistics Market is the shift toward decentralized and hybrid clinical trials. The operational disruptions which happened during the COVID-19 pandemic caused this transition because those disruptions restricted patient access to trial sites and required sponsors to change their trial delivery methods. The companies in the industry now use direct-to-patient drug distribution together with real-time tracking and localized storage networks. The shift creates more complex logistics requirements because it demands additional services which results in higher contract values and continuous revenue growth for specialized logistics companies.
The fragmented regulatory system which exists across European countries establishes a significant structural obstacle. The different requirements which each nation has for drug labeling and importation and temperature compliance create obstacles for cross-border trial logistics. The national healthcare systems together with their legal frameworks create a long-term connection which prevents the system from being fixed immediately. The logistics providers need to preserve duplicative procedures together with their compliance departments, which results in higher expenses and extended timeframes for trial initiation. The small service providers experience market growth delays because their operational processes create inefficiencies which decrease profit margins.
The implementation of advanced digital supply chain platforms which use IoT and predictive analytics technology presents a significant opportunity for business growth. The distribution of biologics benefits from real-time temperature monitoring and AI-based demand forecasting because these technologies enable organizations to manage risks before they occur. Eastern European countries attract businesses because their infrastructure improvements and lower operational expenses create an ideal environment for technology-based logistics operations which can expand.
What Has the Impact of Artificial Intelligence Been on the Europe Clinical Trial Supply and Logistics Market?
Clinical trial supply chains experience transformation through artificial intelligence and advanced digital technologies which automate the multiple tasks of study coordination and monitoring and compliance monitoring across their complex international research operations. The AI-driven platforms of the system implement automated functions for inventory distribution and temperature alert notifications and shipment routing while changing traditional methods through their capacity to manage operations in real time. The machine learning models examine site enrollment data and historical demand information to determine optimal depot stocking levels which decrease excess production of investigational drugs.
The development of predictive capabilities has resulted in enhanced reliability and improved control over project expenses. The system uses algorithms to predict supply disruptions by combining weather information with customs clearance schedules and carrier performance data. The system enables logistics teams to identify optimal shipment routes while ensuring trial operations continue without interruptions. The combination of real-time sensor data with AI analytics systems enables cold chain management systems to detect temperature changes which leads to better product protection and decreased expensive product losses. The systems have produced tangible results that include shorter delivery times and better adherence to European Union regulatory requirements.
The process of implementation faces obstacles because organizations need to pay high expenses for system integration while they manage their disordered information systems. The clinical trial industry faces challenges because many sponsors and logistics companies rely on outdated systems that do not support data sharing between different systems which reduces the effectiveness of AI-driven insights that protect the entire clinical trial process.
Key Market Trends
- The period since 2020 has seen sponsors move away from centralized depot systems towards direct-to-patient distribution which has decreased site need while creating greater need for last-mile delivery capabilities.
- The biologics pipelines between 2021 and 2024 experienced major growth which compelled logistics companies to enhance ultra-cold storage facilities and purchase approved temperature-controlled shipping equipment.
- The EU Clinical Trials Regulation from 2022 states that regulatory bodies must achieve two objectives which include establishing unified rules for cross-border test approvals and executing compliance procedures that require operators to maintain documentation.
- The period from 2020 to present shows that companies such as DHL Group and FedEx Corporation have developed their clinical trial divisions because they want to provide specialized medical logistics solutions which generate higher profits.
- After the COVID-19 pandemic hybrid trial systems became popular which created ongoing needs for home healthcare services and patient-focused supply chain systems.
- AI demand forecasting tools which used artificial intelligence between 2022 and 2025 reduced drug waste from clinical trials through better supply planning that used enrollment data for multinational studies.
- The period after 2021 saw Eastern Europe become a popular trial location because sponsors shifted their research activities to areas with lower operational costs which created demand for distribution centers and transportation networks throughout various regions.
- The period starting in 2023 saw organizations implement blockchain and serialization systems through pilot projects to improve product traceability because new regulations against counterfeiting and auditing requirements mandated these systems.
- The operational model of Contract Research Organizations changed after 2020 because they now partner with logistics vendors while using integrated providers for supply chain operations to enhance efficiency and speed up trial processes.
Europe Clinical Trial Supply and Logistics Market Segmentation
By Type
The distribution and logistics sector maintains its dominant market share because it needs to manage operations which connect multiple countries through their shipping activities and customs operations and emergency delivery services. The storage industry competes with storage systems which need specialized facilities to handle the rising demand for storage of biologics that need controlled condition storage. The primary focus on packaging and labeling operations continues to maintain their importance because businesses need to follow different regulations which exist in various countries.
The current demand patterns show that trial complexity is increasing because researchers need to use smaller batch sizes and adaptive protocols which need flexible packaging and quick product redistribution. The existence of multiple regulations across different regions creates obstacles which require organizations to conduct their operations in all required areas of their business. Service providers will develop integrated service models which combine their distribution services with storage facilities and packaging functions into one platform. The shift will lead vendors to merge their operations while they allocate resources towards developing digital tracking systems and creating uniform packaging standards.
By Application
The Phase III trials require multiple sites to handle their large patient groups because their testing needs extensive logistical support and higher quantities of materials. The demand for Phase II trials remains constant because proof-of-concept studies increase their usage in biologics and specialty therapies. Early-stage trials such as Phase I testing require precise results but maintain a small testing capacity.
Phase IV studies use post-marketing surveillance to gather data about products but their operational requirements remain below standard testing operations. The growth patterns demonstrate that all trial phases now use both decentralized and hybrid testing methods which results in increased shipments directly to patients. The new system requires organizations to establish flexible logistical systems together with systems for tracking operations in actual time which leads to new business possibilities for organizations that specialize in handling intricate trial protocols.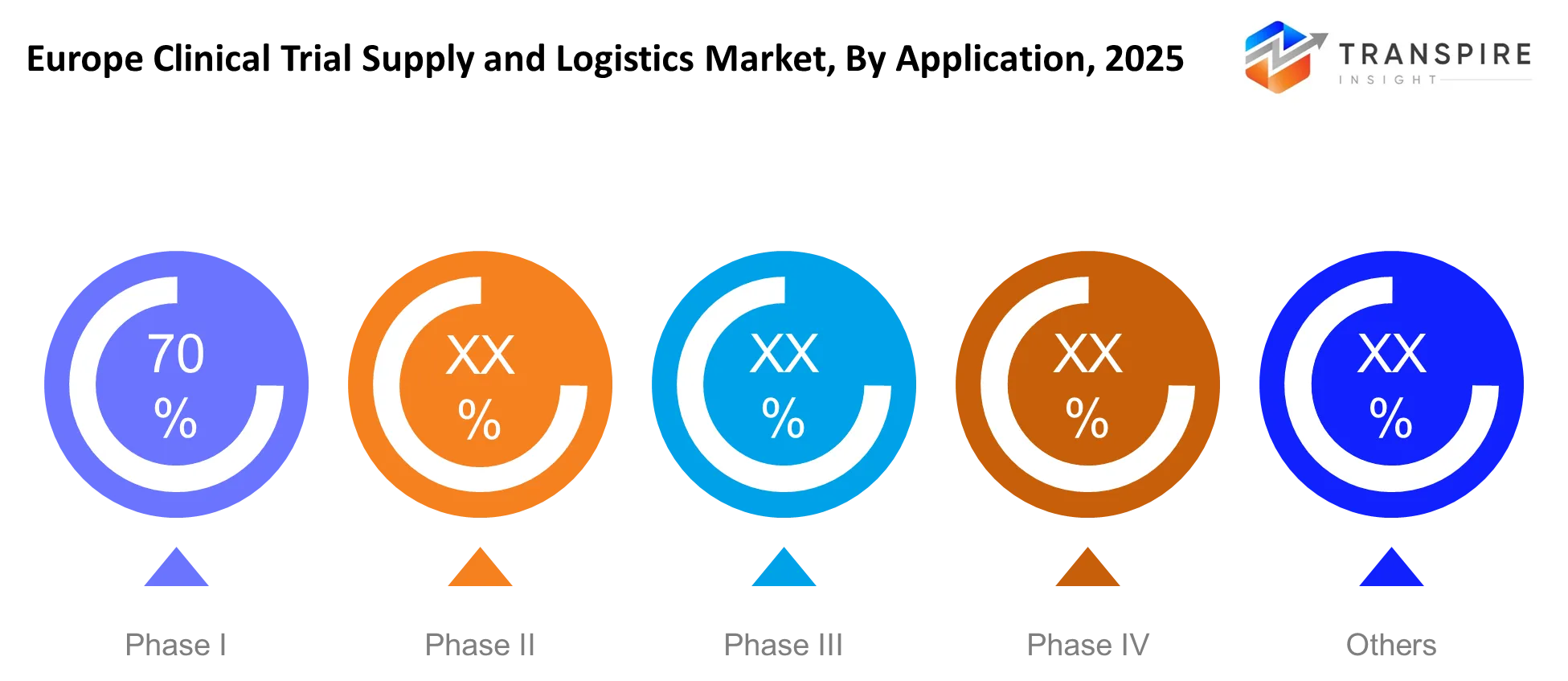
To learn more about this report, Download Free Sample Report
By End-User
The drug development pipelines of pharmaceutical and biotechnology companies demonstrate their market leadership because their specialized logistics requirements force them to spend more on outsourcing services. The share of contract research organizations has increased because sponsors prefer to outsource trial management for two reasons which include reduced operational costs and faster project completion. Research institutes and academic centers contribute smaller volumes but remain important for early-stage and niche studies.
Three main factors drive demand which include cost control and speed to market and the need for businesses to satisfy multiple regulatory requirements. The rising trend of outsourcing requires logistics providers to create complete solution packages instead of delivering standalone services. The upcoming trend will develop ongoing service provider relationships which will establish predictable income streams for businesses.
By Service
The service sector of cold chain logistics shows the highest market share because biologics and vaccines and cell therapies and gene therapies require strict temperature control for their transportation. Non-cold chain services maintain their importance because they still support traditional small molecule drugs although their expansion rate remains limited. The rising demand for advanced therapies drives the need for ultra-low temperature storage and real-time monitoring and validated transport systems.
The two main obstacles for smaller providers are expensive infrastructure requirements and mandatory regulatory validation procedures which restrict their market entry. The future development of the industry will focus on cold chain networks that use technology for IoT-based tracking and predictive analytics. The new development will create product differentiation through three factors: reliability, compliance performance, and product loss control during transit.
What are the Key Use Cases Driving the Europe Clinical Trial Supply and Logistics Market?
The primary purpose of this study involves studying large-scale clinical trials which investigate new drugs that pharmaceutical sponsors need to distribute throughout various European nations according to strict regulations. Phase III studies generate the highest demand because they involve large patient cohorts which require multiple resupply operations to meet their strict deadlines. The logistics providers maintain operations through their responsibility to manage cross-border deliveries and temperature management and site-level inventory management to prevent deviations from established protocols.
Pharmaceutical companies and contract research organizations mainly drive the growth of decentralized trials and home-based treatment models which show increasing use across different fields of application. The operational requirements of these models include direct-to-patient drug delivery and reverse logistics for sample collection and the establishment of connections to home healthcare services which depend on specialized last-mile distribution networks.
The emerging use cases of cell and gene therapy trials require ultra-low temperature storage and speedy logistics operations. Digital trial platforms enable remote monitoring which creates the need for real-time system tracking and adaptive supply chain management solutions.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1.42 Billion |
|
Market size value in 2026 |
USD 1.53 Billion |
|
Revenue forecast in 2033 |
USD 2.58 Billion |
|
Growth rate |
CAGR of 7.75% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Parexel, IQVIA, Catalent, Thermo Fisher, Marken, Almac Group, PCI Pharma Services, DHL, FedEx, UPS Healthcare, Movianto, Sharp Services, World Courier, KLIFO, Biocair |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Packaging, Distribution, Storage, Others), By Application (Phase I, Phase II, Phase III, Phase IV, Others), By End-User (Pharma Companies, CROs, Research Institutes, Others), By Service (Cold Chain, Non-cold Chain, Others) |
Which Regions are Driving the Europe Clinical Trial Supply and Logistics Market Growth?
The market in Western Europe maintains its leading position because of strong regulatory systems and extensive clinical research centers. The EU Clinical Trials Regulation has established unified rules which make it easier to conduct international research studies through better cross-border research study management. The advanced medical facilities of Germany France and the Netherlands enable researchers to conduct numerous clinical trials while they handle complicated biological materials. The existing network of pharmaceutical firms contract research organizations and dedicated logistics companies maintains this market leadership.
Northern Europe operates as a stable market segment which depends on ongoing funding for medical research and technological development in healthcare and digital systems. The Swedish and Danish healthcare systems enhance their trial processes through their commitment to patient data protection and their ability to create patient databases. The region focuses on developing specialized research facilities instead of expanding its operations to Western Europe which results in its ability to attract specialized research projects and preliminary research studies. The strong public healthcare systems and regular enforcement of regulations create stable logistics service requirements in the area.
The cost benefits which Eastern Europe offers together with its expanding clinical trial facilities make it the fastest growing region of the world. The market entry process has become easier because of recent infrastructure investments and enhanced compliance with EU regulatory standards. The multinational trial market of Poland and Hungary attracts researchers who want to study various patient groups while minimizing their study costs. The period between 2026 and 2033 will create significant growth possibilities for logistics companies and investors who want to expand their operations.
Who are the Key Players in the Europe Clinical Trial Supply and Logistics Market and How Do They Compete?
The Europe Clinical Trial Supply and Logistics Market remains moderately fragmented because global logistics companies compete with specialized clinical supply organizations. Incumbents maintain their market share through integrated service delivery systems while niche companies build their market presence through specialized solutions which include biologics and cell therapy treatment methods. The primary basis of competition has shifted from cost efficiency to technology-enabled visibility regulatory compliance and the ability to manage decentralized trials. Buyers now prioritize partners that can combine distribution storage packaging and real-time tracking within a single platform which creates higher challenges for smaller companies that lack digital capabilities.
DHL Group establishes its unique position in the market through its specialized division which provides Life Sciences and Healthcare services that deliver clinical trial logistics through their network of temperature-controlled transportation systems and GDP-certified storage locations. The company expands through its regional infrastructure development projects and its strategic partnerships with pharmaceutical companies which enable the organization to conduct multi-country clinical trials. Thermo Fisher Scientific provides complete clinical supply solutions by delivering manufacturing and packaging and distribution services which lead to improved trial efficiency through reduced transfer processes. Companies achieve better inventory control and forecasting accuracy through ongoing investments in digital supply chain solutions.
FedEx Corporation uses its air cargo network to enable urgent deliveries and patient direct delivery systems through its worldwide operational network which delivers fast service. The company enhances its capabilities through technological advancements that improve both shipment tracking and temperature monitoring systems. Parexel uses service integration through its logistics system which operates as an essential component of its complete clinical trial management system to establish a unified responsibility structure for sponsors. Catalent
Company List
- Parexel
- IQVIA
- Catalent
- Thermo Fisher
- Marken
- Almac Group
- PCI Pharma Services
- DHL
- FedEx
- UPS Healthcare
- Movianto
- Sharp Services
- World Courier
- KLIFO
- Biocair
Recent Development News
In April 2026, Clinical Trials Arena Highlights New Outsourcing and Logistics Partnerships at OCT Europe 2026: Industry leaders gathered in Barcelona to address growing operational challenges in clinical trial supply chains, including decentralized trials, cross-border distribution, and cold-chain management. Several European service providers showcased new partnership-driven logistics models designed to improve trial execution speed and resilience.
Source: https://www.clinicaltrialsarena.com
In February 2026, Thermo Fisher Patheon Promotes Advanced Clinical Supply Technologies at CTS Europe 2026: At Clinical Trial Supply Europe 2026 in Barcelona, Thermo Fisher’s Patheon division emphasized innovations in digital supply visibility, adaptive trial logistics, and temperature-controlled distribution. The company positioned advanced supply-chain integration as critical for handling increasingly complex European clinical studies.
Source: https://www.patheon.com
What Strategic Insights Define the Future of the Europe Clinical Trial Supply and Logistics Market?
The Europe Clinical Trial Supply and Logistics Market is moving toward fully integrated, digitally orchestrated supply networks that support decentralized and adaptive trial designs. The industry moves in this direction because biologics pipelines have become more complex and require real-time multi-country system coordination. The next five to seven years will show logistics providers evolving from execution partners into strategic enablers who will incorporate predictive analytics and direct-to-patient distribution and end-to-end visibility into their fundamental service offerings.
A less visible risk exists because rising market concentration enables large technology-enabled providers to consolidate their capabilities through acquisition. The result would be reduced sponsor control over their decisions because they would have to work with integrated vendors who control everything which would lead to increased expenses and decreased power during price discussions.
Eastern European countries that are developing their infrastructure and regulatory framework now provide market participants with an opportunity to establish special logistics systems for cell and gene therapy products. Market participants should invest early in modular cold chain systems and interoperable digital platforms to capture this high-value, next-generation segment.
Europe Clinical Trial Supply and Logistics Market Report Segmentation
By Type
- Packaging
- Distribution
- Storage
- Others
By Application
- Phase I
- Phase II
- Phase III
- Phase IV
- Others
By End-User
- Pharma Companies
- CROs
- Research Institutes
- Others
By Service
- Cold Chain
- Non-cold Chain
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Clinical Trial Supply and Logistics Market size for the Market will be USD 2.58 Billion in 2033.
The key Segments of the Europe Clinical Trial Supply and Logistics Market are By Type (Packaging, Distribution, Storage, Others), By Application (Phase I, Phase II, Phase III, Phase IV, Others), By End-User (Pharma Companies, CROs, Research Institutes, Others), By Service (Cold Chain, Non-cold Chain, Others).
Major Players in the Europe Clinical Trial Supply and Logistics Market are Parexel, IQVIA, Catalent, Thermo Fisher, Marken, Almac Group, PCI Pharma Services, DHL, FedEx, UPS Healthcare, Movianto, Sharp Services, World Courier, KLIFO, Biocair.
The Current Market size of the Europe Clinical Trial Supply and Logistics Market is USD 1.42 Billion in 2025.
The Europe Clinical Trial Supply and Logistics Market CAGR is 7.75%.
- Parexel
- IQVIA
- Catalent
- Thermo Fisher
- Marken
- Almac Group
- PCI Pharma Services
- DHL
- FedEx
- UPS Healthcare
- Movianto
- Sharp Services
- World Courier
- KLIFO
- Biocair
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


