Europe Healthcare Contract Research Organization Market Size & Forecast:
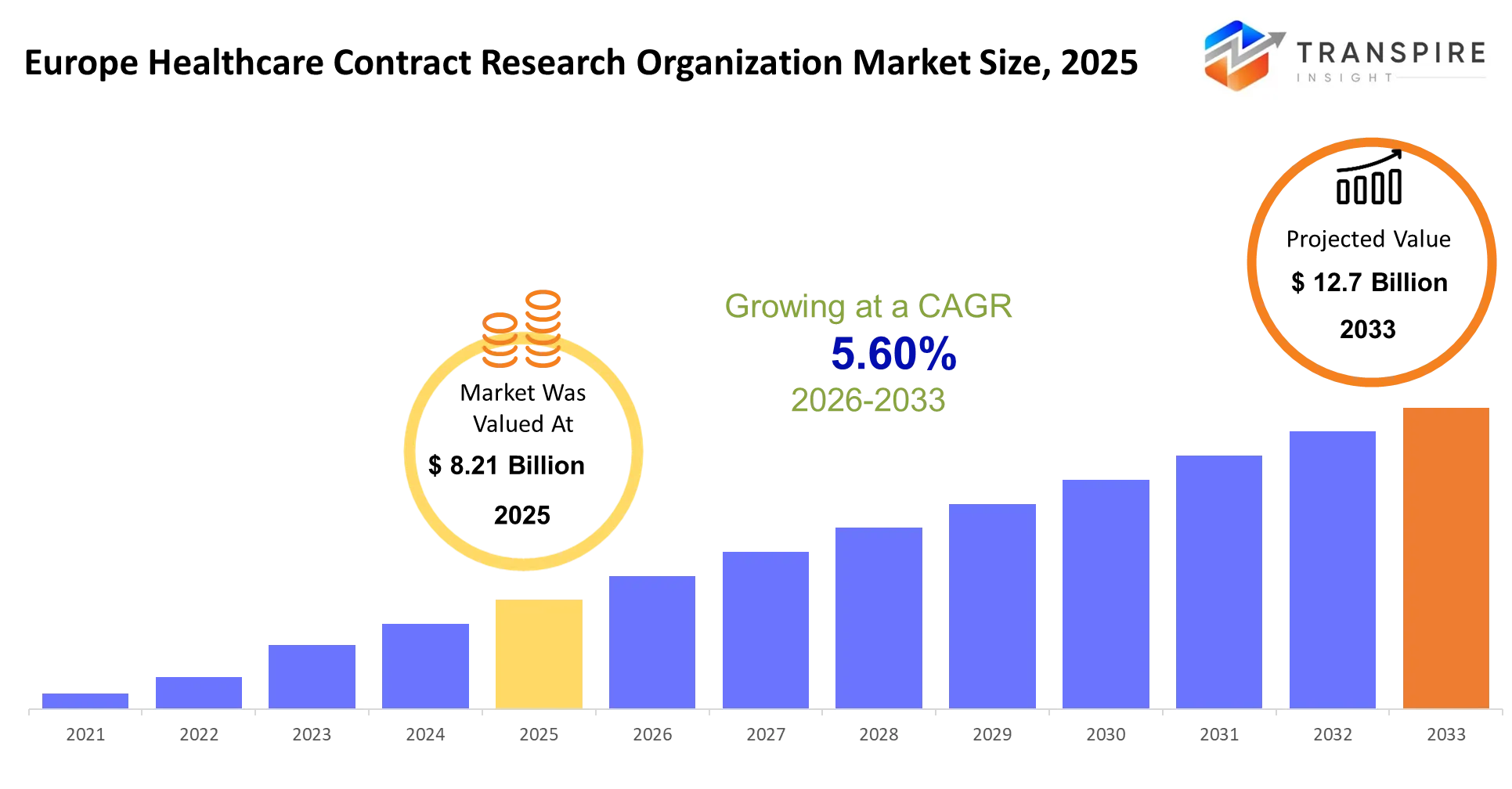
- Europe Healthcare Contract Research Organization Market Size 2025: USD 8.21 Billion
- Europe Healthcare Contract Research Organization Market Size 2033: USD 12.7 Billion
- Europe Healthcare Contract Research Organization Market CAGR: 5.60%
- Europe Healthcare Contract Research Organization Market Segments: By Type (Clinical Research Services, Preclinical Services, Laboratory Services, Others), By Application (Drug Development, Clinical Trials, Medical Device Testing, Regulatory Services, Others), By End-User (Pharma Companies, Biotech Firms, Medical Device Companies, Research Institutes, Others), By Phase (Phase I, Phase II, Phase III, Phase IV, Others)
To learn more about this report, Download Free Sample Report
Europe Healthcare Contract Research Organization Market Summary:
The Europe Healthcare Contract Research Organization Market size is estimated at USD 8.21 Billion in 2025 and is anticipated to reach USD 12.7 Billion by 2033, growing at a CAGR of 5.60% from 2026 to 2033. European healthcare contract research organizations function as outsourced drug development laboratories which handle all aspects of clinical trial execution for pharmaceutical and biotech clients. The organizations enable sponsors to conduct therapy development from laboratory testing to market release by providing essential operational resources and specialized expertise which they lack.
Decentralized and hybrid trial models which use digital monitoring tools and remote patient engagement methods have become the primary research approach during the past three to five years. The shift enables research teams to work with more patients because it reduces their need to use specific research facilities. The COVID-19 pandemic served as the main catalyst because it required trials to continue despite restrictions on movement and it resulted in faster acceptance of virtual methods by regulatory bodies.
The increasing requirement for fast and flexible solutions has emerged as the main driver behind sponsors who now use outsourcing services at higher rates. The combination of data analytics and real-world evidence and adaptive trial capabilities has enabled CROs to create scalable revenue streams from their operational complexities.
Key Market Insights
- Western European countries control the European Healthcare Contract Research Organization Market because they hold more than 65% market share which Germany, the United Kingdom, and France lead in 2024.
- Eastern Europe stands as the most rapidly expanding area until 2030 because of its capacity to conduct cost-effective trials and recruit participants quickly and its progress toward matching European Union regulatory requirements.
- The clinical trial services sector leads the market with a 70% share because European countries choose to outsource their Phase II and III studies.
- The second-largest market share in the industry belongs to preclinical services which receive support from biotech companies developing new products and their expanding research and development activities.
- The segment of data management and real-world evidence services experiences rapid growth because digital trial implementation drives expansion from 2025 to 2030.
- The oncology field holds the largest application market share which reached 30% in 2024 because medical researchers conduct complex trial studies and cancer researchers develop numerous cancer medications.
- The development of personalized medicine together with regulatory support for rare diseases creates the most rapidly developing medical treatment areas which develop targeted therapies.
- Pharmaceutical companies control about 60% of the market because they partner with CROs to decrease their operational expenses while they expedite their drug development process.
- Small and mid-sized biotech firms represent the most rapidly developing user segment which now depends on complete service outsourcing solutions to support their growth needs.
What are the Key Drivers, Restraints, and Opportunities in the Europe Healthcare Contract Research Organization Market?
The main element that drives growth in the Europe Healthcare Contract Research Organization market results from increased outsourcing of complicated clinical development work which arose from rising research and development expenses that now exceed USD 2 billion per approved drug. Pharmaceutical companies need to reduce their product development timeframes while dealing with complicated regulatory requirements that exist throughout various European countries. Organizations now practice higher outsourcing rates because their late-stage clinical trials contain the most difficult execution work and patient enrollment difficulties. Sponsors who spend more money on external trial expenses create more valuable contracts which lead to extended service relationships with CROs.
The European regulatory system remains divided between different national regulations despite the existence of EMA regulations which control research activities. Every nation operates its own separate systems for obtaining ethics approvals and managing patient data and enforcing compliance at research facilities. The existing operational model for clinical research organizations (CROs) faces challenges because they need to maintain standardized operations which results in longer trial preparation times and increased administrative expenses. The process leads to study delays which make certain markets less appealing thereby limiting revenue opportunities and restricting the growth of European-wide research models.
The growth potential for decentralized clinical trials shows significant promise because digital health systems become more advanced in countries such as Germany and the Nordic region. Remote monitoring and eConsent and wearable data capture technologies make it possible for more patients to participate in studies which leads to quicker study enrollment.
What Has the Impact of Artificial Intelligence Been on the Europe Healthcare Contract Research Organization Market?
European contract research organizations now utilize artificial intelligence together with advanced digital tools to transform their clinical trial design process through automated execution of essential operational tasks. AI-powered systems now use natural language processing together with robotic process automation to execute the patient identification process and site feasibility assessments and data cleaning tasks. The system establishes a trial setup and monitoring process which requires less manual work to achieve between 20% and 20% time savings while producing more accurate data results through multi-country studies. Digital control layers enable organizations to monitor compliance because they continuously verify trial data against changing regulatory standards.
Machine learning models add predictive capabilities that directly impact trial performance. Sponsors and CROs use algorithms to forecast patient recruitment rates, predict dropout risks, and optimize site selection based on historical and real-world datasets. The process results in better enrollment efficiency which decreases expensive delays, with some studies reporting 15–25% faster recruitment cycles. AI supports adaptive trial designs by simulating protocol changes before implementation, which enhances decision-making under uncertain conditions.
The technologies enable organizations to achieve better operational results through three main benefits which include higher trial success rates, fewer protocol amendments, and reduced total development expenses. The process faces implementation challenges because data ecosystems remain fragmented and integration costs remain high, especially for unifying traditional clinical systems across various European regulatory frameworks.
Key Market Trends
- The field of decentralized and hybrid trials experienced rapid growth since 2020 because more than 40 percent of European research studies adopted remote testing methods to continue their operations during travel limitations.
- The pharmaceutical industry has raised its outsourcing activities from 45 percent in 2018 to more than 55 percent in 2024 which indicates the industry now prefers to outsource its clinical research work to external partners.
- Eastern Europe became the most popular location for clinical trials after 2021 because patient recruitment processes in this region required 15 to 20 percent less time than in Western European markets.
- The period from 2019 to 2024 brought increasing complexity to oncology trials which resulted in clinical research organizations needing complete service solutions for their biomarker testing and flexible trial development needs.
- Biotechnology companies which operate at mid-sized scale began establishing strategic partnerships with CROs after 2020 because this approach enabled them to decrease their need for internal operational capacity while speeding up their first-in-human trials.
- The COVID-19 pandemic created new regulatory rules which permitted agencies to use remote monitoring and eConsent, thus changing compliance standards throughout Europe.
- Real-world evidence integration gained speed after 2022 as sponsors utilized outside data to back their regulatory applications and post-market research work.
- CROs transformed their business model from being service providers to becoming strategic development partners by investing in digital health technologies and site network capabilities.
Europe Healthcare Contract Research Organization Market Segmentation
By Type
The primary clinical research services category maintains market leadership because companies outsource their complex trial operations which require specialized expertise particularly during late-stage trials that involve maximum expenditure and regulatory scrutiny. Preclinical services maintain a stable share because European biotech companies expand their early-stage research activities while laboratory services experience continuous growth due to increasing requirements for biomarker tests and centralized diagnostic services. Sponsors now prefer complete service solutions because they view integrated service models as more effective than using multiple separate vendor partnerships.
CROs that offer clinical services combined with advanced data analytics and real-world evidence capabilities currently experience the highest demand according to market trends. The upcoming growth period will require providers to develop digital tool packages which they can combine with their existing services to create more valuable contracts while maintaining operational efficiency throughout various phases of clinical trials.
To learn more about this report, Download Free Sample Report
By Application
Clinical trials make up the largest portion of research through their ongoing funding which supports drug development pipelines and Europe-based multinational research studies that need complex execution. The drug development services sector maintains its position because biotech clusters experience increased funding from venture capital that supports their early-stage research work. The EU Medical Device Regulation now requires more compliance work and testing procedures which has led to increased demand for medical device testing services.
Sponsors now seek regulatory services because they need help to navigate multiple approval processes which exist in their business operations. The field of research shows increasing focus on precision medicine and rare disease studies which demand distinct clinical trial methods and specific techniques for selecting research participants. Future developments will lead to CROs and sponsors working together more closely through adaptive management of regulatory requirements and trial testing which uses data-driven techniques for faster trial approval processes.
By End-User
The end-user market supports pharmaceutical companies because their extensive clinical research programs and ongoing needs to minimize operational expenses. Biotech firms represent the fastest-growing group, which depends on outsourcing because they lack sufficient internal capabilities and need adaptable development processes. Medical device companies maintain ongoing demand because of stricter compliance requirements, while research institutions advance knowledge through their academic and government-funded research projects.
The different groups exhibit unique patterns of involvement because pharmaceutical companies prefer to establish permanent partnerships while biotechnology companies need to develop operations that can expand rapidly. The future research direction will establish stronger connections between CRO capabilities and sponsor organization size because customized service packages and risk-sharing agreements will determine market competition and customer retention methods.
By Phase
The three trial phases establish Phase III as the main revenue source for CROs because of its extensive size and expensive operations and its vital function in obtaining regulatory approval. The increasing need for proof-of-concept studies and dose optimization research enables Phase II studies to maintain their substantial market share. The number of Phase I trials keeps increasing because more biotech companies create new products and conduct their initial human tests. The demand for Phase IV services increases because companies need to conduct post-marketing surveillance under new safety monitoring regulations.
The demand patterns show that later phase testing needs more complex services which leads to companies outsourcing their work and seeking specialized expertise. The future will bring new adaptive trial designs together with decentralized methods which will change how clinical trials distribute their phases while they search for faster ways to complete all testing phases and enhance their operational performance.
What are the Key Use Cases Driving the Europe Healthcare Contract Research Organization Market?
The primary application of clinical trials centers on their execution because large pharmaceutical companies operate multi-country Phase II and III studies according to European regulatory requirements. The most demanding requirement arises from this use case because it involves operational activities between multiple jurisdictions which include patient recruitment and site management and compliance needs.
The company has developed its capabilities to support medical device testing according to EU Medical Device Regulation and provide regulatory consulting services for biotech companies entering first-in-human trials. The small and mid-sized biotech companies now turn to CROs for two purposes, which include finding their approval pathways and achieving efficient growth during their early-stage development process.
The emerging use cases of decentralized trials involve the application of remote monitoring tools, while real-world evidence studies function as methods for post-market surveillance. The models have become more popular because regulators have started to accept digital endpoints, while sponsors now pursue quicker access to substantial data through advanced clinical research methods.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 8.21 Billion |
|
Market size value in 2026 |
USD 8.67 Billion |
|
Revenue forecast in 2033 |
USD 12.7 Billion |
|
Growth rate |
CAGR of 5.60% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
IQVIA, LabCorp, Parexel, ICON, Syneos Health, Charles River, Medpace, PPD, Covance, WuXi AppTec, PRA Health Sciences, SGS, Eurofins, Envigo, Clinipace |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Clinical Research Services, Preclinical Services, Laboratory Services, Others), By Application (Drug Development, Clinical Trials, Medical Device Testing, Regulatory Services, Others), By End-User (Pharma Companies, Biotech Firms, Medical Device Companies, Research Institutes, Others), By Phase (Phase I, Phase II, Phase III, Phase IV, Others)) |
Which Regions are Driving the Europe Healthcare Contract Research Organization Market Growth?
Western Europe controls the Europe Healthcare Contract Research Organization Market because its regulatory system operates through European Medicines Agency and its clinical research facilities function at advanced levels. The countries of Germany France and the United Kingdom maintain extensive networks of trial sites and academic hospitals together with qualified investigators. The established system which includes pharmaceutical headquarters and biotech clusters as well as digital health platforms maintains continuous demand for outsourcing. The combination of robust data protection systems and advanced healthcare networks enables organizations to conduct complex international clinical trials which require numerous countries to participate.
Northern Europe operates as a dependable region which Sweden Denmark and Finland use to prioritize patient registry integration and data quality control. This region differs from Western Europe through its focus on smaller but highly efficient trials supported by national health databases and unified digital systems. CROs experience stable operational conditions because of regular regulatory compliance and government-sponsored research development initiatives. The system maintains growth through transparent patient data access and high public healthcare involvement which enables development without the fundamental operational requirements that other systems need.
Eastern Europe demonstrates its fastest development rate through its recent investments in clinical infrastructure together with its compliance to European regulatory standards. The countries of Poland Hungary and Romania have improved site capabilities and reduced approval timelines since 2020. Global sponsors have chosen to work in trial execution because the combination of lower operational costs and faster patient recruitment brings them efficiency gains. The current momentum creates multiple entry points for CROs and investors who want to build capacity while they capture new market demand between 2026 and 2033.
Who are the Key Players in the Europe Healthcare Contract Research Organization Market and How Do They Compete?
The Europe Healthcare Contract Research Organization market experiences competition which exists between two opposing market trends of consolidation and specialization. Global CROs maintain their market leadership with their vast operations and complete service offerings and their established relationships with pharmaceutical companies while mid-sized and niche companies use their specific therapeutic knowledge and regional expertise to compete. Companies now compete through technology-based services which deliver better results than their previous cost-based methods because data integration and decentralized trial systems and rapid execution methods determine their ability to win contracts. The digital platform and complete service expansion maintain market share for established companies, while newcomers disrupt markets that focus on rare diseases and early-stage trials through their flexible operations and specialized knowledge.
The advanced analytics and real-world data platforms developed by IQVIA provide sponsors with the tools they need to create adaptive predictive trials. Organizations that invest in AI-powered insights and develop partnerships with healthcare organizations enhance their ability to access data, which leads to better trial results. ICON plc provides complete service solutions which connect clinical development to healthcare intelligence, through its multiple acquisitions, to gain better control over project schedules and operational expenses. Parexel helps sponsors to navigate European regulatory requirements through its specialized consulting services and execution assistance.
The Labcorp Drug Development organization uses its laboratory facilities to link diagnostic testing with clinical trial operations, which enables better design of biomarker-based studies and patient grouping. Syneos Health provides a comprehensive business model which connects its services for clinical development with market access, through a single system, that provides seamless trial execution and market entry solutions.
Company List
- IQVIA
- LabCorp
- Parexel
- ICON
- Syneos Health
- Charles River
- Medpace
- PPD
- Covance
- WuXi AppTec
- PRA Health Sciences
- SGS
- Eurofins
- Envigo
- Clinipace
Recent Development News
In October 2025, Thermo Fisher Expands Clinical Trial Services via Clario Acquisition Integration: Thermo Fisher is actively progressing its acquisition of Clario Holdings, strengthening its European clinical trial data and analytics services footprint. Clario specializes in clinical trial data capture and management—core infrastructure used by CROs across Europe for late-stage trials. The deal is expected to close in early 2026.
Source: https://www.reuters.com
In April 2026, Thermo Fisher to Divest Microbiology Business to Astorg: Thermo Fisher Scientific has agreed to sell its microbiology business to European private equity firm Astorg in a ~$1.075 billion deal. The unit supports clinical and pharmaceutical testing workflows, which are closely linked to CRO-enabled laboratory services in Europe. The move reflects continued consolidation of outsourced clinical support capabilities in the region.
Source: https://www.wsj.com
What Strategic Insights Define the Future of the Europe Healthcare Contract Research Organization Market?
The Europe Healthcare Contract Research Organization market is following a structural shift toward platform-based systems which use digital infrastructure and real-world data and decentralized trial execution to create competitive advantages. The fundamental driver behind this movement requires organizations to shorten their development times while they develop complex therapies which require large amounts of data to create treatments such as biologics and personalized medicine. The market will move toward strategic collaboration as organizations develop their capabilities to serve as complete data and execution partners which will help them obtain more funding from sponsors.
The hidden danger of data fragmentation together with different regulations about patient data usage creates a danger that AI-driven trial models will struggle to operate across various countries. This limitation will create obstacles for international standardization efforts which will decrease the efficiency benefits that organizations expect to achieve through their digital transformation initiatives.
Eastern Europe presents an emerging opportunity for hybrid trial expansion because its developing infrastructure and regulatory framework enable easier patient recruitment which can be scaled up. Market participants need to develop digital platforms which enable system interoperability together with establishing regional site networks to achieve their goals of maintaining data access while ensuring regulatory compliance and operational flexibility.
Europe Healthcare Contract Research Organization Market Report Segmentation
By Type
- Clinical Research Services
- Preclinical Services
- Laboratory Services
- Others
By Application
- Drug Development
- Clinical Trials
- Medical Device Testing
- Regulatory Services
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Medical Device Companies
- Research Institutes
- Others
By Phase
- Phase I
- Phase II
- Phase III
- Phase IV
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Healthcare Contract Research Organization Market size for the Market will be USD 12.7 Billion in 2033.
The key Segments of the Europe Healthcare Contract Research Organization Market are By Type (Clinical Research Services, Preclinical Services, Laboratory Services, Others), By Application (Drug Development, Clinical Trials, Medical Device Testing, Regulatory Services, Others), By End-User (Pharma Companies, Biotech Firms, Medical Device Companies, Research Institutes, Others), By Phase (Phase I, Phase II, Phase III, Phase IV, Others).
Major Players in the Europe Healthcare Contract Research Organization Market are IQVIA, LabCorp, Parexel, ICON, Syneos Health, Charles River, Medpace, PPD, Covance, WuXi AppTec, PRA Health Sciences, SGS, Eurofins, Envigo, Clinipace.
The Current Market size of the Europe Healthcare Contract Research Organization Market is USD 8.21 Billion in 2025.
The Europe Healthcare Contract Research Organization Market CAGR is 5.60%.
- IQVIA
- LabCorp
- Parexel
- ICON
- Syneos Health
- Charles River
- Medpace
- PPD
- Covance
- WuXi AppTec
- PRA Health Sciences
- SGS
- Eurofins
- Envigo
- Clinipace
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


