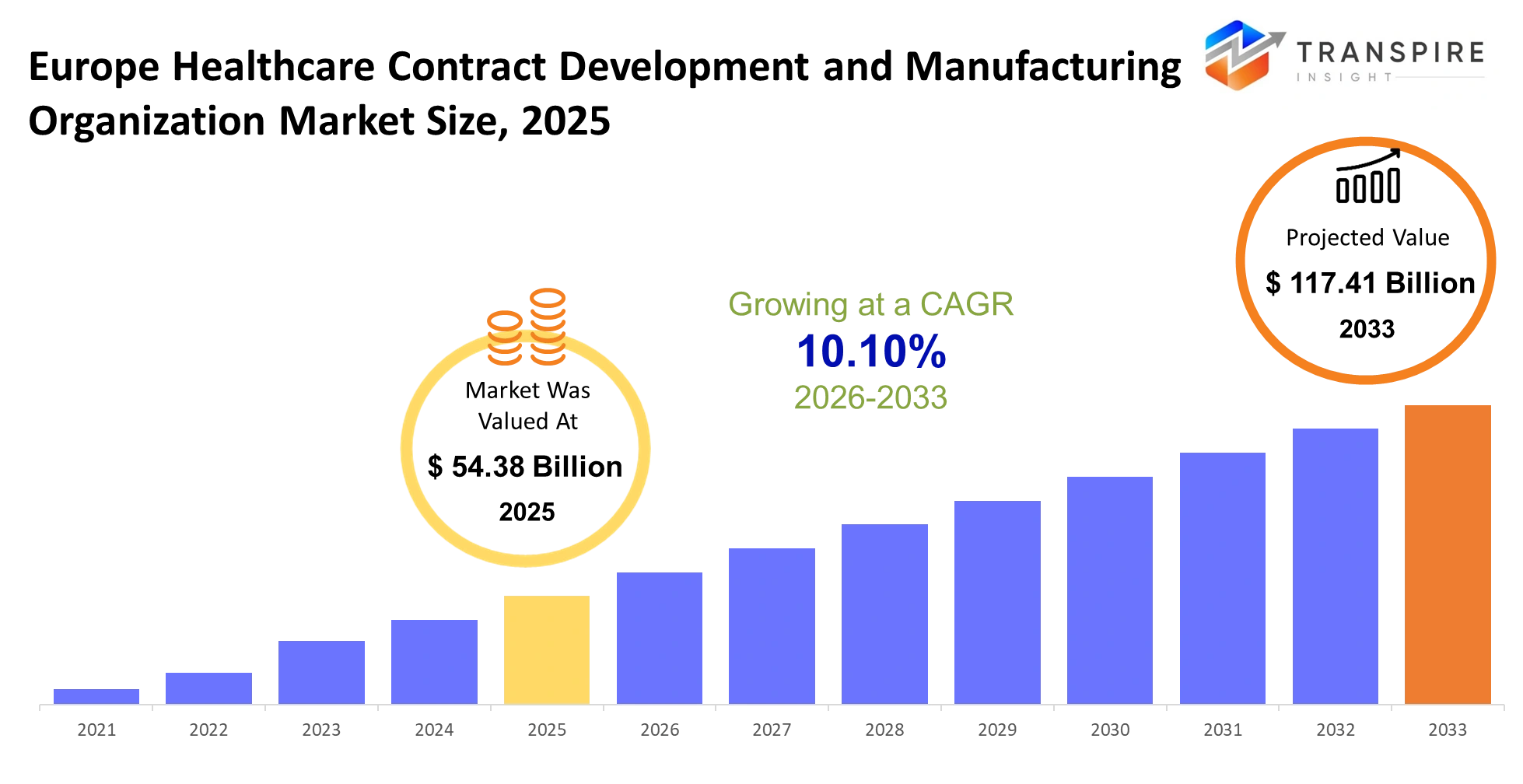
Europe Healthcare Contract Development and Manufacturing Organization Market Size & Forecast:
- Europe Healthcare Contract Development and Manufacturing Organization Market Size 2025: USD 54.38 Billion
- Europe Healthcare Contract Development and Manufacturing Organization Market Size 2033: USD 117.41 Billion
- Europe Healthcare Contract Development and Manufacturing Organization Market CAGR: 10.10%
- Europe Healthcare Contract Development and Manufacturing Organization Market Segments: By Type (Drug Development, Manufacturing, Packaging, Others), By Application (Oncology, Cardiology, Neurology, Infectious Diseases, Others), By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others), By Service (API, Finished Dosage, Biologics, Others).
To learn more about this report, Download Free Sample Report
Europe Healthcare Contract Development and Manufacturing Organization Market Summary:
The Europe Healthcare Contract Development and Manufacturing Organization Market size is estimated at USD 54.38 Billion in 2025 and is anticipated to reach USD 117.41 Billion by 2033, growing at a CAGR of 10.10% from 2026 to 2033. Europe's CDMO healthcare market supports drug development by transforming laboratory discoveries into products that meet regulatory requirements for patient accessibility. Pharmaceutical and biotech firms depend on their partners to handle intricate production processes while decreasing their equipment expenses and shortening their project completion times within an environment that requires regulatory compliance because any delay will impact both their financial success and their ability to provide treatment to patients.
The market underwent its first major transformation during the last three to five years when companies started developing biologics and advanced therapies which required facilities and expertise that most organizations could not create through internal development. The COVID-19 pandemic functioned as a real-life demonstration that showed global supply chains needed protection through flexible manufacturing systems built across different regions.
The current situation has created new outsourcing approaches for businesses to follow. Companies now prioritize long-term CDMO partnerships which result in increased contract values and greater operational capacity while CDMOs develop their high-margin business functions through cell and gene therapy production capabilities.
Key Market Insights
- Germany controls more than 25 percent of the European Healthcare Contract Development and Manufacturing Organization Market in 2024 because its biopharma infrastructure supports its market presence.
- The region of Ireland experiences its highest growth rate until 2030 because it offers attractive tax incentives and receives substantial foreign direct investment for its biologics manufacturing sector.
- Companies use outsourcing to their production capacity which creates manufacturing services as the market leader with 60 percent of total market share in 2024.
- The second-largest market share belongs to development services which receive support from European countries that are developing their drug pipelines.
- The fastest-growing segment of the company operations Biologics CDMO services will experience substantial growth from 2025 until 2030 because of the increasing need for complex therapy solutions.
- Oncology applications dominate with over 35% share in 2024 because of active clinical pipeline development and production of high-value biologic drugs.
- The application of cell and gene therapy development becomes the most rapidly growing field because precision medicine developments will enable its widespread implementation until 2030.
- Pharmaceutical companies lead with nearly 55% share in 2024, leveraging CDMOs for scalable and compliant production solutions.
- The end-user segment which includes small and mid-sized biotech firms now experiences its strongest growth because these companies lack sufficient production facilities.
What are the Key Drivers, Restraints, and Opportunities in the Europe Healthcare Contract Development and Manufacturing Organization Market?
The European Healthcare Contract Development and Manufacturing Organization market experiences its main growth driver from increasing demand for biologics and advanced therapies. Scientists achieved major advancements in monoclonal antibodies and cell therapies and mRNA platforms during the post-COVID-19 period which led to this development. Pharmaceutical companies need to maintain specialized manufacturing facilities because they lack capacity to produce these specialized treatments. The companies now prefer to partner with CDMOs who demonstrate both regulatory compliance and technical expertise. The growth of contract agreements between companies results in longer business relationships which provide CDMOs with more stable income streams because they enable drug development processes to proceed faster.
The healthcare industry faces its most important operational challenge because there are not enough advanced manufacturing facilities to support cell and gene therapy production. The process of establishing compliant facilities requires organizations to spend money on infrastructure development while facing lengthy requirements for facility validation and needing to find qualified personnel whose numbers cannot be increased at an instant. The existing limitations postpone the hiring process for upcoming projects and create production-based delays which prevent equipment from being used. As a result, biotech companies need to either wait for their clinical development schedules or pay extra for contracts which limits their access to the market and decreases their revenue growth speed.
Upcoming industrial growth opportunities arise from the development of decentralized systems together with modular manufacturing technologies. Organizations can establish their facilities with operational flexibility through the implementation of single-use bioreactors and continuous manufacturing systems. Ireland and Belgium use their policy incentives together with their developed infrastructure to attract investments which establish them as emerging centers for next-generation CDMO capacity.
What Has the Impact of Artificial Intelligence Been on the Europe Healthcare Contract Development and Manufacturing Organization Market?
European healthcare CDMO operations now experience operational efficiency improvements through artificial intelligence and advanced digital technologies yet research shows that companies only implement these technologies in specific areas rather than across their entire operations. The manufacturing sector uses AI-based automation to control processes which oversee the real-time operation of bioreactors and filtration systems and cleanroom environments. The systems enable automated operation which results in decreased operational needs and more consistent production runs that produce superior results which lead to lower operational costs and fewer production errors.
Machine learning models are increasingly used for predictive maintenance of critical equipment such as fermenters and filling lines. CDMOs use historical sensor data to predict equipment failures before they happen which results in better equipment availability and lower unexpected downtime between breaks. Predictive analytics also supports yield optimization which enables manufacturers to dynamically change settings that result in higher output efficiency for their most valuable biologics products.
These technologies help organizations achieve faster batch release times while their documentation process becomes automated to improve regulatory compliance and their operational expenses decrease throughout the day. The primary drawback of AI systems remains their need for specialized resources which use extensive financial resources while establishing operational connection between AI technology and existing systems. Many facilities operate with fragmented data systems, which limits model accuracy and slows large-scale deployment across manufacturing networks.
Key Market Trends
- Large pharmaceutical companies have transitioned to multi-year CDMO contracts because this business model provides better supply security and more consistent production capabilities than their previous method of using transactional outsourcing.
- The production of biologics has grown from its 2018 level of below 40% to its current share of more than 55% in 2024 because drug pipelines have become more complex.
- Companies in Europe started to manufacture products locally after the pandemic because supply chains were interrupted which led them to decrease their dependence on Asian API imports by almost 20% since 2021.
- The CDMOs Lonza Group and Recipharm AB have raised their investment in new facilities by more than 30% since 2022 to create new capacity for biologics production.
- The regulatory agencies established stricter GMP compliance requirements after 2021 which resulted in regulators conducting more frequent audits while companies needed to invest more in their digital quality management systems.
- Small biotech firms now account for over 45% of CDMO clients in 2024 because they require external manufacturing support after developing their internal production capabilities.
- The implementation of single-use technologies has reached double the 2019 level because this technology streamlines facility operations while protecting against contamination in environments that produce multiple products.
- Since 2021 the manufacturing demand for cell and gene therapies has increased rapidly which has resulted in European hubs reaching capacity utilization rates above 80%.
- Between 2021 and 2024 CDMOs formed partnerships with biotech innovators which recorded a 25% growth rate because these partnerships focused on joint development initiatives while sharing operational expenses.
Europe Healthcare Contract Development and Manufacturing Organization Market Segmentation
By Type
The manufacturing services which require high capital investment for their operations and which need to follow strict regulatory standards in drug production work as the most important service type within the industry. The drug development services sector follows this development because small and mid-sized biotech companies build their early-stage pipelines. The packaging services together with their supporting operations create a stable market presence which remains smaller than other market segments because European markets need serialization and compliance solutions.
The manufacturing sector shows growth because companies now outsource their complex production needs especially those related to biologics and high-potency substances. The trend exists because sponsors maintain limited operational abilities which they can perform within their organizations. The future path shows integrated service models which providers use to deliver development and manufacturing and packaging services through single contract agreements. The new approach leads to higher contract worth which helps businesses maintain client partnerships and drives market consolidation while companies invest more in their unique manufacturing capabilities.
By Application
The field of oncology remains the most important medical specialty because its research requirements create more clinical trials and its need for specialized drug development creates new manufacturing challenges. The pandemic caused a temporary rise in infectious disease research whereas neurology and cardiology fields experience growth because of demographic changes that accompany an aging population. Oncology maintains its top position because research funding and successful treatment outcomes present better financial results than other medical fields.
The studies about medical demand show that people increasingly prefer personalized medicine which creates difficulties for production and testing operations. The field of cell and gene therapy will experience expansion especially in the areas of oncology and rare disease treatment. Service providers need to develop advanced abilities because their operations need to create manufacturing systems that can handle changing medical requirements.
By End-User
The pharmaceutical industry maintains its top position because of its established pipeline systems which allow them to outsource their manufacturing needs. The fastest-growing group in the market occurs because biotech companies cannot develop their products through their existing facilities but they focus on creating new innovations. Research institutes and academic centers create partnerships to work on projects but they only maintain a small portion of the commercial production process.
Biotech companies need outside help because their venture capital funding supports initial growth stages without providing money for their manufacturing plants. The current business environment establishes ongoing needs for organizations that provide adaptable services which can grow according to demand. Biotech firms will establish deeper strategic alliances with contract development and manufacturing organizations according to future trends because both parties will share development risks while implementing co-investment systems to speed up their product introduction process.
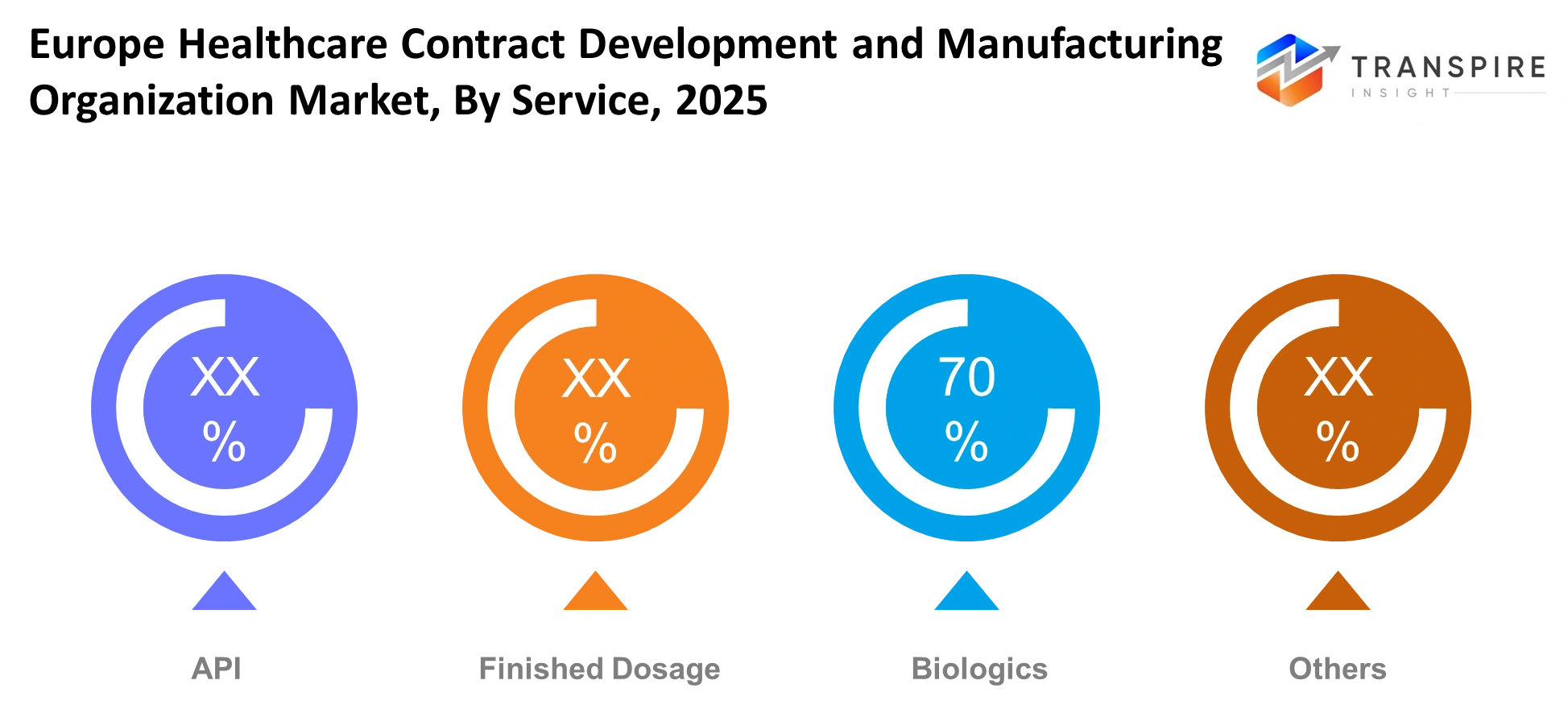
By Service
The fastest evolving segment of the industry operates through biologics manufacturing which serves as the main service of the business because customers demand monoclonal antibodies and vaccines and advanced therapies. The active pharmaceutical ingredient manufacturing process maintains its strong market position because customers require high-quality active ingredients while finished dosage formulation remains essential for final product delivery. Biologics growth occurs because companies develop more valuable therapeutic products which require advanced production processes that prevent them from manufacturing these products internally.
The need for specialized containment systems and quality assurance processes drives organizations to make capital investments in their advanced facility developments. The industry will experience continuous growth in biologics production capacity while companies implement continuous manufacturing methods and single-use production systems. Service providers who combine their API capabilities with their biologics and finished dosage services will obtain more valuable contracts which will enhance their market competitiveness.
To learn more about this report, Download Free Sample Report
What are the Key Use Cases Driving the Europe Healthcare Contract Development and Manufacturing Organization Market?
The main application focuses on producing large quantities of biologics which include monoclonal antibodies used in cancer treatment. Pharmaceutical companies outsource work to this area because they face three major challenges which include handling intricate production processes and adhering to strict GMP standards and needing to invest in expensive equipment.
Biotech companies use contract development manufacturing organizations for their clinical trial material production and their small-batch manufacturing needs. Contract development manufacturing organizations enable early-stage companies to grow in their rare disease and neurology-related research areas without requiring them to build their own facilities.
The new applications specifically target the production of cell and gene therapies and the development of mRNA platforms. Personalized medicine development and European regulatory pathways will create future capacity needs in these fields which currently suffer from limited operational capacity.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 54.38 Billion |
|
Market size value in 2026 |
USD 59.87 Billion |
|
Revenue forecast in 2033 |
USD 117.41 Billion |
|
Growth rate |
CAGR of 10.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Lonza, Catalent, Recipharm, Thermo Fisher, Samsung Biologics, WuXi AppTec, Siegfried, Almac, Piramal Pharma, Cambrex, Evotec, Charles River, AGC Biologics, Boehringer Ingelheim, Famar. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Drug Development, Manufacturing, Packaging, Others), By Application (Oncology, Cardiology, Neurology, Infectious Diseases, Others), By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others), By Service (API, Finished Dosage, Biologics, Others). |
Which Regions are Driving the Europe Healthcare Contract Development and Manufacturing Organization Market Growth?
The market in Western Europe reaches its highest point because Germany France and Switzerland operate their pharmaceutical industries through effective systems which enable both regulatory control and market development. The combination of strict GMP standard enforcement together with a centralized approval process system creates an environment which manufacturers high-value products. The region maintains continuous innovation while using its extensive network of biotech clusters and research institutions together with its skilled workforce. Established CDMOs achieve their market leadership position through their long-term agreements with international pharmaceutical companies which operate in their respective regions.
Northern Europe serves as a stable contributor to economic development because Sweden Denmark and Ireland focus on achieving operational excellence through their transparent organizational procedures. The region depends on specialized training for biologic products and specialized medical solutions which differs from Western European countries that use their larger territories to gain advantages. International companies establish operations in the region because governments maintain stable regulatory frameworks which provide tax benefits for their businesses. The region maintains its stability against market disturbances because of its continuous capital investments and its methodical business growth approach.
The fastest developing area in Central and Eastern Europe advances through its cost effective advantages and its latest pharmaceutical infrastructure development projects. Poland and Hungary have modernized their production facilities so they can meet European Union compliance requirements. The region has experienced a rise in nearshoring activities because companies shifted their outsourcing work following supply chain disruptions. The new CDMO market expansion creates opportunities for market entry while providing investors reduced operating expenses which will rise to higher technical standards until 2033.
Who are the Key Players in the Europe Healthcare Contract Development and Manufacturing Organization Market and How Do They Compete?
The Healthcare Contract Development and Manufacturing Organization market in Europe exhibits a semi-consolidated competitive structure because multiple major companies hold most important biologics production capacity while smaller organizations focus on specific services. The existing companies maintain their market position through their established contractual partnerships and their proven history of meeting regulatory requirements which result in substantial client retention costs. Technology capability and integrated service models have become the primary basis of competition, replacing pure cost advantages. New companies struggle to achieve rapid growth because of the need for substantial financial resources and their requirement to meet compliance standards, but dedicated companies continue to emerge in the cell and gene therapy market.
Lonza Group uses technology-based methods to create unique manufacturing solutions for both biologics and cell therapy production. Lonza obtains high-value contracts through its advanced technological platforms and its worldwide approved certification system. Recipharm AB operates its business based on two core strengths: flexible production capabilities and a solid presence throughout Europe which helps mid-sized customers optimize their growth potential. Siegfried Holding AG establishes its market position through its ability to produce high-potency active pharmaceutical ingredients combined with its comprehensive development capabilities for complex small molecule products. The two companies use strategic acquisitions together with partnerships to extend their service offerings while focusing on their core business activities.
Company List
- Lonza
- Catalent
- Recipharm
- Thermo Fisher
- Samsung Biologics
- WuXi AppTec
- Siegfried
- Almac
- Piramal Pharma
- Cambrex
- Evotec
- Charles River
- AGC Biologics
- Boehringer Ingelheim
- Famar
Recent Development News
In February 2026, Charles River raises annual profit view as it sells underperforming assets: Charles River announced divestment of parts of its European CDMO and drug discovery assets as part of a strategic restructuring. The deal is expected to reshape its European outsourcing footprint in advanced drug manufacturing services.
Source: https://www.reuters.com
In January 2026, contract drug manufacturer Lonza forecasts slower sales growth for 2026: Lonza, one of Europe’s largest CDMOs, reported a slowdown in expected CDMO growth for 2026 despite strong 2025 performance. The company also highlighted margin expansion plans and continued strength in biologics manufacturing platforms.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the Europe Healthcare Contract Development and Manufacturing Organization Market?
The Europe Healthcare Contract Development and Manufacturing Organization market is developing integrated service platforms which provide high-complexity solutions throughout the next five to seven years. The industry shift occurs because biologics and cell therapies and personalized medicines now require development and manufacturing operations to work together under one company. As pipelines become more specialized, sponsors will favor fewer but deeper partnerships, concentrating revenue among technologically advanced CDMOs.
The market concentration risk operates through the way businesses build their operations around a few leading companies which maintain advanced capabilities. The rising demand for advanced therapies has created a situation where qualified facilities for serving this need operate at limited capacity, which gives major companies the ability to dictate prices. This phenomenon will create obstacles for smaller biotech companies who will face difficulties accessing funding, which will result in decreased innovation activities despite the overall industry growth.
The industrialization of modular and decentralized manufacturing systems presents an opportunity in Central and Eastern Europe because infrastructure developments are underway. A strategic decision for market participants should involve early investment in flexible small-scale biologics platforms which enable rapid growth in high-demand therapy areas while minimizing capital requirements.
Europe Healthcare Contract Development and Manufacturing Organization Market Report Segmentation
By Type
- Drug Development
- Manufacturing
- Packaging
- Others
By Application
- Oncology
- Cardiology
- Neurology
- Infectious Diseases
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Others
By Service
- API
- Finished Dosage
- Biologics
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Healthcare Contract Development and Manufacturing Organization Market size for the market will be USD 117.41 Billion in 2033.
The key segments of the Europe Healthcare Contract Development and Manufacturing Organization Market are By Type (Drug Development, Manufacturing, Packaging, Others), By Application (Oncology, Cardiology, Neurology, Infectious Diseases, Others), By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others), By Service (API, Finished Dosage, Biologics, Others).
Major players in the Europe Healthcare Contract Development and Manufacturing Organization Market are Lonza, Catalent, Recipharm, Thermo Fisher, Samsung Biologics, WuXi AppTec, Siegfried, Almac, Piramal Pharma, Cambrex, Evotec, Charles River, AGC Biologics, Boehringer Ingelheim, Famar.
The current market size of the Europe Healthcare Contract Development and Manufacturing Organization Market is USD 54.38 Billion in 2025.
The Europe Healthcare Contract Development and Manufacturing Organization Market CAGR is 10.10%.
- Lonza
- Catalent
- Recipharm
- Thermo Fisher
- Samsung Biologics
- WuXi AppTec
- Siegfried
- Almac
- Piramal Pharma
- Cambrex
- Evotec
- Charles River
- AGC Biologics
- Boehringer Ingelheim
- Famar
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









