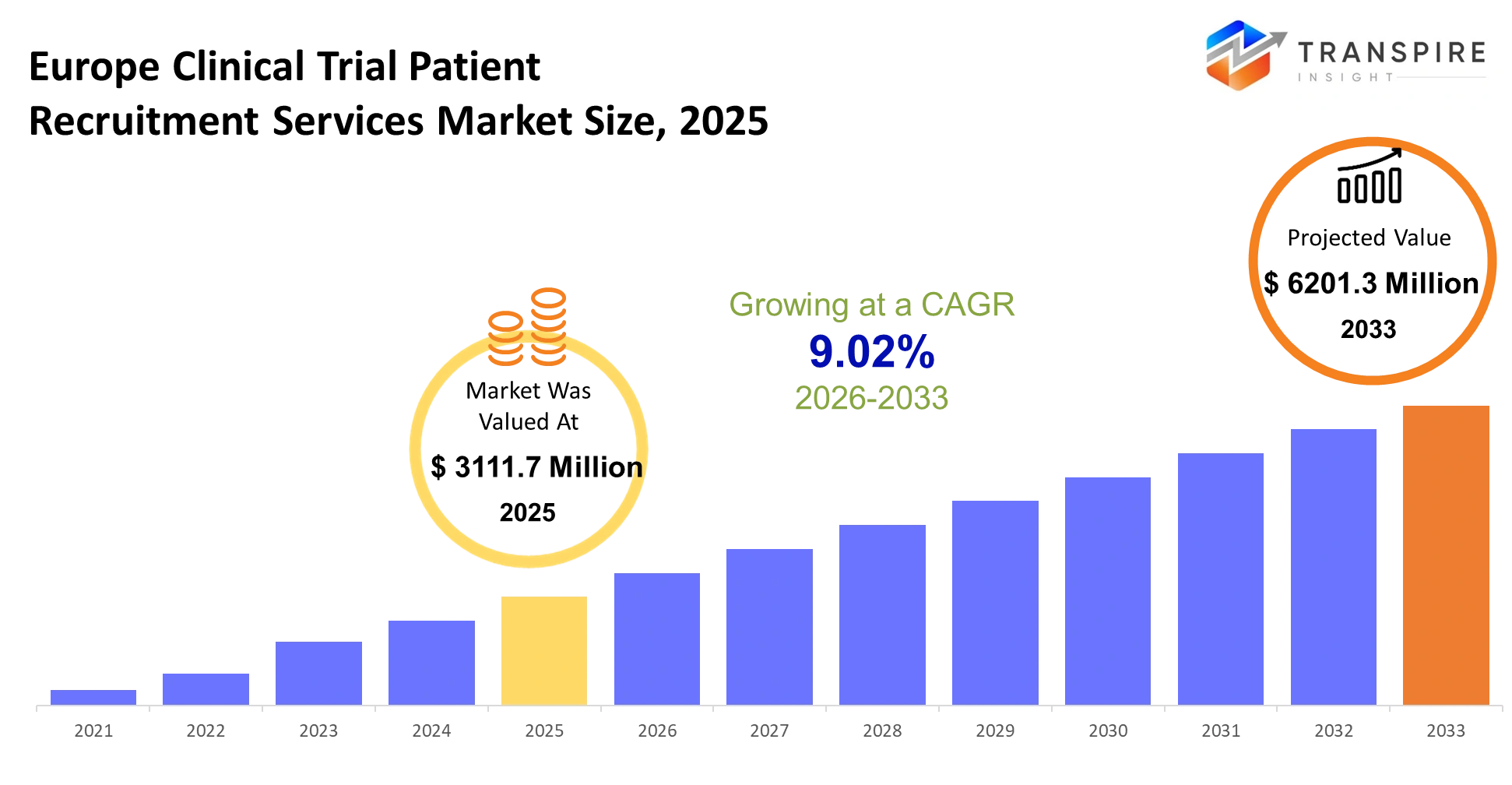
Europe Clinical Trial Patient Recruitment Services Market Size & Forecast:
- Europe Clinical Trial Patient Recruitment Services Market Size 2025: USD 3111.7 Million
- Europe Clinical Trial Patient Recruitment Services Market Size 2033: USD 6201.3 Million
- Europe Clinical Trial Patient Recruitment Services Market CAGR: 9.02%
- Europe Clinical Trial Patient Recruitment Services Market Segments: By Type (Patient Recruitment, Site Identification, Patient Retention, Digital Recruitment, Others); By Application (Oncology Trials, Cardiology Trials, Neurology Trials, Rare Disease Trials, Others); By End-User (Pharma Companies, CROs, Research Institutes, Biotech Firms, Others); By Service (Digital Recruitment, Traditional Recruitment, Hybrid Recruitment, Others)
To learn more about this report, Download Free Sample Report
Europe Clinical Trial Patient Recruitment Services Market Summary
The Europe Clinical Trial Patient Recruitment Services Market was valued at USD 3111.7 Million in 2025. It is forecast to reach USD 6201.3 Million by 2033. That is a CAGR of 9.02% over the period.
The clinical trial patient recruitment services across Europe establish connections between pharmaceutical sponsors and contract research organizations and suitable patients who participate in drug development tests by transforming existing healthcare systems into operational systems for finding study participants. The services function as a solution to an existing problem by enabling researchers to find suitable study participants while they conduct their screening process and maintain participant involvement throughout their studies which take place in various nations that have different healthcare systems.
The last 3–5 years have brought a market transformation that establishes decentralized recruitment through digital channels which use AI-based matching and real-world data platforms to replace existing site-only enrollment methods. The EU Clinical Trials Regulation (CTR) implementation together with COVID-19 pandemic disruption led to increased adoption of remote recruitment methods and hybrid trial designs which required sponsors to develop new patient access methods. The new process enables recruitment through data analysis while enabling researchers to select participants from different locations which increases the speed of enrollment and raises the number of people who can take part in clinical studies.
Key Market Insights
- Western Europe will control the European Clinical Trial Patient Recruitment Services Market in 2025 because it holds 52% market share which results from Germany France and the UK strong clinical infrastructure.
- Northern Europe will experience its most rapid development until 2030 because of its extensive adoption of digital health records and decentralized trial models.
- The Europe Clinical Trial Patient Recruitment Services Market operates with site-based recruitment services because established hospital networks provide them with approximate 41% market share.
- The second-most popular recruitment method involves patient database recruitment which benefits from the increasing adoption of real-world data platforms.
- The period between 2024 and 2030 will see digital and AI-enabled recruitment emerge as the fastest developing market segment because its growth depends on automation and predictive matching systems.
- Oncology trials account for 38% of application usage because cancer therapeutics research leads to extensive drug development activities throughout the world.
- The application segment for rare disease trials shows the fastest expansion because of advancements in genetic screening methods and international patient discovery systems.
- The pharmaceutical industry constitutes the dominant market segment in the European Clinical Trial Patient Recruitment Services Market because it accounts for 55% of total market share.
- The fastest expansion in Contract Research Organizations (CROs) occurs because sponsors prefer to use outsourcing for recruitment operations to achieve better operational results.
- Companies make substantial financial commitments toward developing AI-powered patient matching systems that will help them finish recruitment processes more quickly.
What are the Key Drivers, Restraints, and Opportunities in the Europe Clinical Trial Patient Recruitment Services Market?
The structural shift toward decentralized and digitally enabled clinical trials serves as the primary driver which accelerates growth in the Europe Clinical Trial Patient Recruitment Services Market. The EU Clinical Trials Regulation (CTR) framework together with the COVID-19 period created a situation which compelled sponsors to develop new recruitment methods that enabled remote patient participation and cross-border patient access. The increase in AI-driven patient matching platforms and real-world data integration has occurred because this change enables faster enrollment processes and better trial management. The pharmaceutical industry partners with recruitment service providers to achieve faster product launches which results in increased contract agreements and extended service arrangements.
The healthcare data systems in European countries face their most important challenge because fragmentation exists between different systems which national privacy and regulatory frameworks maintain. The trial regulations have achieved standardization yet patient records still exist in incompatible formats which GDPR enforcement applies with strict control. The process of data exchange encounters obstacles which impede automatic patient identification and result in extended recruitment phases while raising operational expenses for service providers. The market loses efficiency gains because organizations need to complete digital adoption while advanced recruitment platforms remain unutilized which results in revenue losses.
The development of cross-border real-world evidence networks presents a significant opportunity for growth which will benefit Northern and Central Europe. Denmark and the Netherlands have established national health data interoperability platforms which will help these countries identify patient cohorts more effectively.The company develops a scalable base for its AI-based recruitment services which enables businesses to create forecasting models and increase their testing range. The European Clinical Trial Patient Recruitment Services Market will enter its next growth stage when these ecosystems reach maturity because they will enable more accurate recruitment processes and shorter trial start times.
What Has the Impact of Artificial Intelligence Been on the Europe Clinical Trial Patient Recruitment Services Market?
The combination of artificial intelligence with advanced digital technologies creates new operational methods for scrubber performance systems and exhaust gas cleaning technology which European maritime operators use to monitor their systems through automated data analysis. The shipping industry uses scrubber systems which contain sensors and connect to cloud analytics to monitor their sulfur oxide emissions and fuel sulfur levels while complying with IMO 2020 and EU MRV standards. The automated system decreases required manual checks while it enhances the accuracy of regulatory reports which fleet operators need to produce across different regions.
The industry increasingly applies machine learning models to develop predictive maintenance systems which detect the initial signs of corrosion and scaling and pressure loss in scrubber systems before they reach failure points. The models enable emissions forecasting and voyage optimization capabilities which help operators decrease fuel use while they stay within required compliance limits. The shipping companies achieved measurable progress in system availability and business productivity while reducing unexpected repair expenses and enhancing their fuel consumption results.
The high costs required for system integration together with the challenges involved in upgrading existing vessels to use digital sensor networks create obstacles which prevent technology adoption. The limited satellite coverage in open ocean areas creates additional challenges for transmitting information in real time and the lack of sufficient labeled data regarding failures in extreme marine environments leads to decreased accuracy for predictive modeling.
Key Market Trends
- Recruitment strategies shifted from site-centric models in 2020 to hybrid decentralized systems after EU Clinical Trials Regulation rollout and COVID-19 disruption.
- AI-powered patient matching tools replaced manual database screening which resulted in nearly 30% faster enrollment cycle times during large oncology trials across Europe.
- The real-world evidence platforms started to grow after 2022 because sponsors used electronic health record connections to identify eligible patients.
- Pharmaceutical companies increasingly outsourced recruitment functions to CROs like IQVIA and ICON plc to improve trial scalability and geographic reach.
- Cross-border patient recruitment increased because GDPR-compliant data frameworks enabled better data sharing between Western and Northern European healthcare systems.
- Digital-first recruitment strategies became the main method for oncology and rare disease trials because of the historical pattern of low patient availability combined with high screening failure rates.
- Traditional advertising-based patient recruitment decreased because targeted digital outreach campaigns produced better results at lower costs.
- Machine learning adoption in feasibility analysis improved protocol design accuracy which resulted in fewer mid-trial amendments throughout Phase II and Phase III studies.
- Northern Europe emerged as a digital adoption hub because Denmark and Sweden developed national health data platforms to enhance recruitment processes.
Europe Clinical Trial Patient Recruitment Services Market Segmentation
By Type:
The Europe Clinical Trial Patient Recruitment Services Market operates mainly through patient recruitment which serves as its primary functional type because that process enables trial sponsors to complete their studies. Pharmaceutical sponsors prioritize this segment because delays in recruitment directly inflate clinical development costs and extend time-to-market for new therapies.
The segment experiences growth because organizations now practice precision recruitment which uses AI-based patient matching and real-world data to identify suitable candidates. Site identification and patient retention services are gaining relevance, but recruitment continues to absorb the highest investment as sponsors aim to reduce screening failures. The category experiences its fastest growth through digital recruitment which uses platforms to replace traditional physician referral pathways.
The development of patient recruitment services will proceed to full automation through data-driven systems which will connect to national health databases during the upcoming forecast period. The shift will create operational advantages for sponsors while driving higher demand for technology-based service providers.
To learn more about this report, Download Free Sample Report
By Application:
The leading application segment for oncology trials exists because there are multiple worldwide cancer drug development pipelines and there are strict rules about which patients can participate in trials. Cardiology and neurology trials maintain steady demand, but oncology dominates recruitment needs because of high trial volume and frequent protocol complexity.
The development of rare disease trials has become the fastest growing application because of better genetic testing methods and increased diagnostic resources throughout Europe. The regulatory environment for orphan drug development provides companies with various incentives which together drive development costs downward while maintaining all relevant operational requirements.
Future demand will concentrate heavily on precision medicine applications, especially oncology and rare diseases. The requirement for advanced recruitment analytics and cross-border patient identification systems will increase because this shift needs better enrollment efficiency.
By End-User:
Pharmaceutical companies hold the largest share in the Europe Clinical Trial Patient Recruitment Services Market due to their control over late-stage drug development pipelines and large-scale trial budgets. The companies must improve recruitment processes because they need to finish their development work faster while increasing their research-based financial returns.
Contract Research Organizations (CROs) represent the fastest-growing end-user segment as outsourcing becomes a preferred strategy for managing complex multi-country trials. Research institutes and biotech firms contribute steadily, particularly in early-phase and niche therapeutic areas where specialized recruitment is required.
CROs will expand their recruitment work because sponsors adopt more adaptable outsourcing methods. The requirement for unified recruitment systems that include data analysis capabilities and tools for patient interaction will increase.
By Service:
Digital recruitment services lead the market shift due to widespread adoption of AI-driven patient matching, social media outreach, and electronic health record integration. The manual recruitment methods of traditional recruitment exist in the market but have lost their market presence because digital tools provide superior speed and accuracy compared to manual methods.
Organizations use hybrid recruitment models because sponsors need to combine their digital outreach methods with their site-based verification processes to achieve better enrollment results. The method has gained particular significance in oncology and rare disease trials because these studies deal with complex eligibility requirements.
The future development of healthcare services will proceed through two main channels which include complete digital models and hybrid service models as healthcare data interoperability progresses throughout Europe. Service providers who dedicate resources to automation combined with predictive analytics will achieve better market advantages because trial durations decrease.
What are the Key Use Cases Driving the Europe Clinical Trial Patient Recruitment Services Market?
The Europe Clinical Trial Patient Recruitment Services Market exists primarily because oncology drug development serves as its most important application. The healthcare industry requires patient identification services which deliver immediate results because clinical trials need to identify patients who meet their strict eligibility requirements while their testing procedures update frequently. Pharmaceutical companies rely heavily on recruitment services to reduce screening failures and shorten study start-up timelines.
Contract Research Organizations and biotech companies that oversee their first development stage are using neurology and rare disease research studies as their second application area. The trials require access to specialized patient databases and genomic screening tools which help them find small patient populations that exist throughout multiple European healthcare systems.
Real-world evidence-based recruitment systems and predictive analytics-based identification of high-risk groups for preventive trial enrollment have now become potential use cases. The national health data platform integration in Denmark and the Netherlands enables health professionals to identify patients earlier while they develop recruitment plans for their adaptive and decentralized clinical trial methods.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 3111.7 Million |
|
Market size value in 2026 |
USD 3388.5 Million |
|
Revenue forecast in 2033 |
USD 6201.3 Million |
|
Growth rate |
CAGR of 9.02% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
IQVIA, Parexel, Syneos Health, PPD, ICON, Covance, PRA Health Sciences, Medpace, Worldwide Clinical Trials, Chiltern, Veristat, Novotech, KCR, ClinChoice, TrialSpark |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Patient Recruitment, Site Identification, Patient Retention, Digital Recruitment, Others); By Application (Oncology Trials, Cardiology Trials, Neurology Trials, Rare Disease Trials, Others); By End-User (Pharma Companies, CROs, Research Institutes, Biotech Firms, Others); By Service (Digital Recruitment, Traditional Recruitment, Hybrid Recruitment, Others) |
Which Regions are Driving the Europe Clinical Trial Patient Recruitment Services Market Growth?
The European Clinical Trial Patient Recruitment Services Market gets its leading position through Western Europe which has both a comprehensive pharmaceutical industry and its sophisticated clinical research system. The three countries of Germany France and the United Kingdom operate as hubs for multinational pharmaceutical companies and academic medical institutions which deliver constant streams of new patients for research purposes. The EU Clinical Trials Regulation establishes strong regulatory alignment which enables better trial management across international borders while creating uniform methods for participant recruitment. The existing digital health infrastructure and common use of electronic health records systems enable organizations to improve their patient identification processes. The system creates conditions for ongoing management of recruitment service requirements.
Northern Europe holds the second position, but its strength lies in stability rather than scale. The countries of Sweden, Denmark and Finland operate their public healthcare systems through digital technology which receives continuous support from their governments to promote life science research. The region attracts demand through its need for high-quality patient registries which contain extensive data that research teams need to conduct their studies. National health databases receive permanent funding which guarantees consistent participant enrollment for clinical trials. The pharmaceutical sponsors use this dependable information to conduct their studies which require complex research processes that last for extended periods.
Eastern Europe shows the highest growth rate because of its recent development of clinical trial facilities and its reduced costs for research sponsors. Since 2021, Poland and Hungary have made significant investments to transform their medical facilities and create regulatory systems that comply with European Union requirements. Pharmaceutical companies from Western Europe have accelerated their patient recruitment operations through increased outsourcing to this region.This growth signals strong opportunity for service providers and investors targeting scalable, cost-efficient trial expansion between 2026 and 2033.
Who are the Key Players in the Europe Clinical Trial Patient Recruitment Services Market and How Do They Compete?
The Europe Clinical Trial Patient Recruitment Services Market shows a moderately consolidated structure, led by a small group of global CROs and data-driven service providers. The competition has increased because sponsors now select their suppliers according to technological capabilities and especially AI technologies which support patient matching and real-world data integration. The existing companies maintain their market position through extended pharmaceutical contracts while new digital companies create disturbances in conventional recruitment methods which depend on physical locations. Service providers now compete through their ability to enroll patients quickly and exchange data between systems and treat patients from different countries rather than through their ability to operate in different regions.
IQVIA strengthens its position through data-centric recruitment strategies built on one of the largest real-world healthcare databases in Europe. The company uses predictive analytics to find eligible patients earlier during the trial process and connects recruitment with clinical data systems. The company aims to expand its operations through partnerships with hospital networks and national health systems which will enable it to access more data and enhance its ability to match patients in oncology and rare disease trials.
ICON plc competes through full-service CRO integration, combining recruitment with trial management to reduce sponsor coordination complexity. Syneos Health focuses on hybrid commercialization models, linking recruitment with patient engagement campaigns across digital channels. Parexel International uses its regulatory knowledge and extensive experience in conducting complex international studies to establish itself as a leader in multi-country trials which operate under EU CTR regulations. Medpace expands selectively into specialized therapeutic areas, using niche expertise in oncology and rare diseases to secure high-value recruitment contracts across Europe.
Company List
- IQVIA
- Parexel
- Syneos Health
- PPD
- ICON
- Covance
- PRA Health Sciences
- Medpace
- Worldwide Clinical Trials
- Chiltern
- Veristat
- Novotech
- KCR
- ClinChoice
- TrialSpark
Recent Development News
“In January 2026, ICON plc announced expansion of its Europe-focused patient recruitment network through additional bolt-on acquisitions of AI-enabled clinical trial technology assets. The development strengthened decentralized recruitment capabilities and improved cross-border patient matching efficiency across oncology and rare disease studies.https://www.proclinical.com
What Strategic Insights Define the Future of the Europe Clinical Trial Patient Recruitment Services Market?
The Europe Clinical Trial Patient Recruitment Services Market is moving toward complete development of recruitment systems which use health data networks to find and enroll patients while keeping them in clinical studies. The European Union Clinical Trial Regulation for EU CTR requires standardization of its regulations and the widespread use of electronic health record systems which creates a unified system for patient recruitment that costs less than current methods while showing better results through advanced analytical methods and artificial intelligence for trial design.
The risk of patient data control becoming centralized in the hands of major CROs and technology companies creates a hidden danger for the industry. The major companies will control access to essential recruitment databases which they have built through their data collection efforts thus creating challenges for new companies to enter the market and leading to prolonged dependency problems for sponsors. This situation will create pricing disparities that affect all parts of the business.
An emerging opportunity exists for genomic screening programs to combine with national healthcare databases in Northern Europe which will enable faster identification of eligible rare disease and oncology patients. The capability exists but current commercial application remains limited although pilot programs are driving its regional expansion. Market participants should focus on establishing partnerships with public health systems while developing AI platforms which can function across different regulatory environments to maintain their market dominance.
Europe Clinical Trial Patient Recruitment Services Market Report Segmentation
By Type
- Patient Recruitment
- Site Identification
- Patient Retention
- Digital Recruitment
- Others
By Application
- Oncology Trials
- Cardiology Trials
- Neurology Trials
- Rare Disease Trials
- Others
By End-User
- Pharma Companies
- CROs
- Research Institutes
- Biotech Firms
- Others
By Service
- Digital Recruitment
- Traditional Recruitment
- Hybrid Recruitment
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Europe Clinical Trial Patient Recruitment Services Market size is USD 6201.3 Million in 2033.
Key segments for the Europe Clinical Trial Patient Recruitment Services Market are By Type (Patient Recruitment, Site Identification, Patient Retention, Digital Recruitment, Others); By Application (Oncology Trials, Cardiology Trials, Neurology Trials, Rare Disease Trials, Others); By End-User (Pharma Companies, CROs, Research Institutes, Biotech Firms, Others); By Service (Digital Recruitment, Traditional Recruitment, Hybrid Recruitment, Others).
Major Europe Clinical Trial Patient Recruitment Services Market players are IQVIA, Parexel, Syneos Health, PPD, ICON, Covance, PRA Health Sciences, Medpace, Worldwide Clinical Trials, Chiltern, Veristat, Novotech, KCR, ClinChoice, TrialSpark.
The Europe Clinical Trial Patient Recruitment Services Market size is USD 3111.7 Million in 2025.
The Europe Clinical Trial Patient Recruitment Services Market CAGR is 9.02% from 2026 to 2033.
- IQVIA
- Parexel
- Syneos Health
- PPD
- ICON
- Covance
- PRA Health Sciences
- Medpace
- Worldwide Clinical Trials
- Chiltern
- Veristat
- Novotech
- KCR
- ClinChoice
- TrialSpark
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)