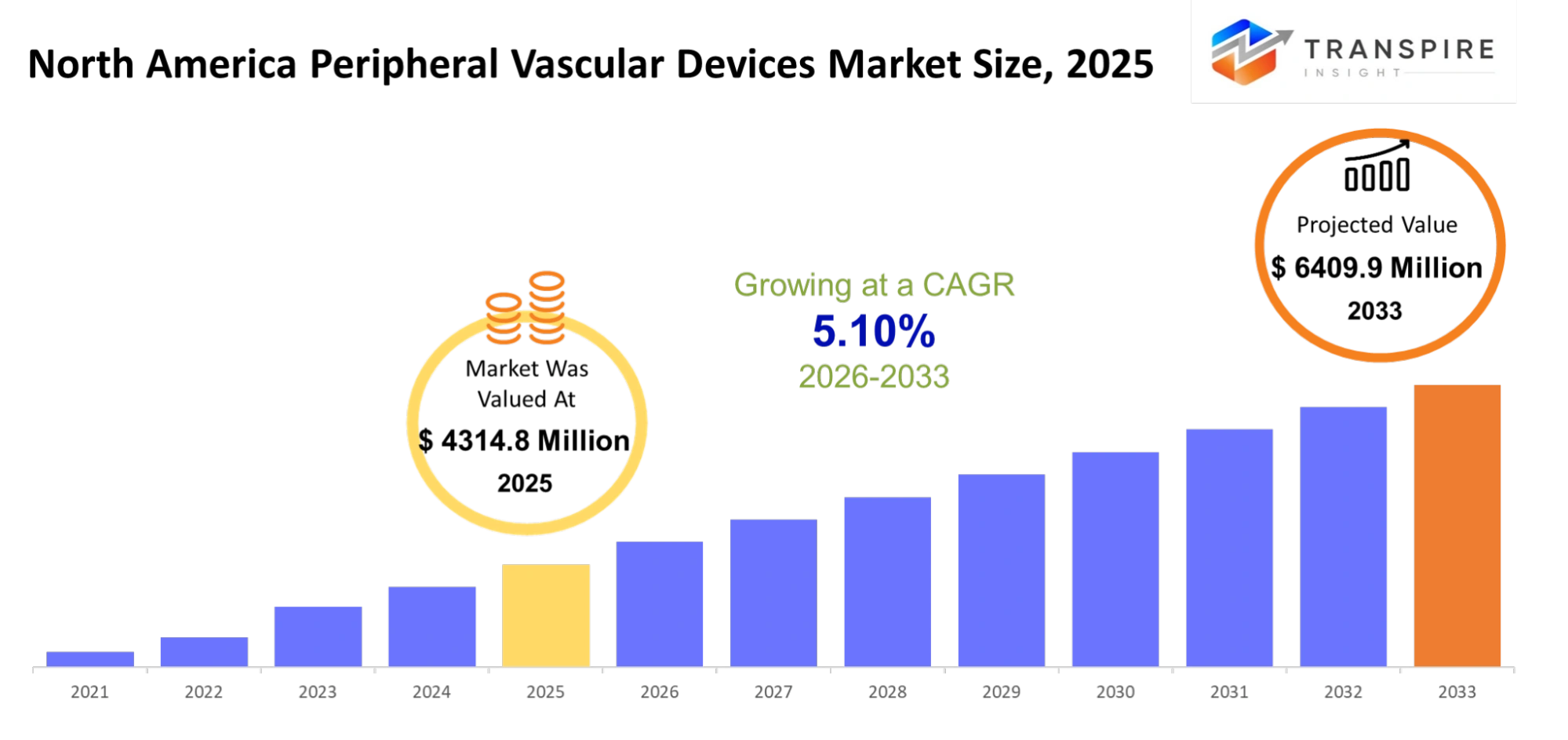
North America Peripheral Vascular Devices Market Size & Forecast:
- North America Peripheral Vascular Devices Market Size 2025: USD 4314.8 Million
- North America Peripheral Vascular Devices Market Size 2033: USD 6409.9 Million
- North America Peripheral Vascular Devices Market CAGR: 5.10%
- North America Peripheral Vascular Devices Market Segments: By Device Type (Angioplasty Balloons, Stents, Catheters, Atherectomy Devices), By Application (Peripheral Artery Disease, Venous Diseases), By End-User (Hospitals, Ambulatory Surgical Centers)
To learn more about this report, Download Free Sample Report
North America Peripheral Vascular Devices Market Summary:
The North America Peripheral Vascular Devices Market size is estimated at USD 4314.8 Million in 2025 and is anticipated to reach USD 6409.9 Million by 2033, growing at a CAGR of 5.10% from 2026 to 2033. The North America Peripheral Vascular Devices Market which includes Canada and the United States, and Mexico operates as a distinct market segment within the medical device industry that specializes in restoring blood flow through peripheral artery treatments. The market will develop through increased patient demand for minimally invasive procedures, which offer rapid recovery times and shorter hospital stays. The field of medical treatment will experience continuous change through advancements in catheter technology drug-coated devices and improved imaging methods. The increasing demand for safe products supported by empirical evidence will lead to more stringent regulatory requirements which medical device manufacturers must meet. The demand for medical devices will increase because of two factors: the aging population and the rise of vascular diseases that result from unhealthy lifestyles which will drive the creation of customized devices that prioritize individual patient needs throughout the entire region.
Key Market Trends & Insights:
The North America Peripheral Vascular Devices Market, which includes countries such as Canada, the USA, and Mexico, is now witnessing a stronger inclination towards vascular treatments through methods of minimal invasion. The field of medicine is also likely to witness a higher degree of approval for treatments that cause lesser harm to the patient while ensuring faster rates of healing. Advanced forms of catheters and stents along with imaging-based treatments are also likely to gain traction as hospitals and clinics have started to embrace these new forms of medical treatments and technologies.
The process of diagnosis and treatment for vascular diseases is also witnessing a digital transformation through various technological advancements. It is likely to see a rise in the use of smart imaging along with data-driven planning as a means of ensuring accuracy during treatments and surgeries. Physicians will be relying on the analysis of data in real-time, while manufacturers of these devices will be focusing on ensuring the integration of such features to assess and monitor the performance of these devices as per the current standards of healthcare technology.
Patient awareness about vascular health affects their choices regarding medical treatments and healthcare expenses. Patients will pursue early detection methods together with treatment options that cause less interruption which will lead hospitals to develop dedicated vascular treatment centers. Patients will drive demand for customized treatment methods which use medical devices that match their individual body structure and health background and future health effects.
The United States and Canada, and Mexico have established newer regulatory systems which require better proof of medical evidence. Authorities will require manufacturers to conduct testing and compliance procedures through their clinical validation process and post-market monitoring requirements. The current business landscape will create an environment that enables new ideas to develop while maintaining safety standards which will determine the process for bringing medical devices into the market and keeping them there.
North America Peripheral Vascular Devices Market Segmentation
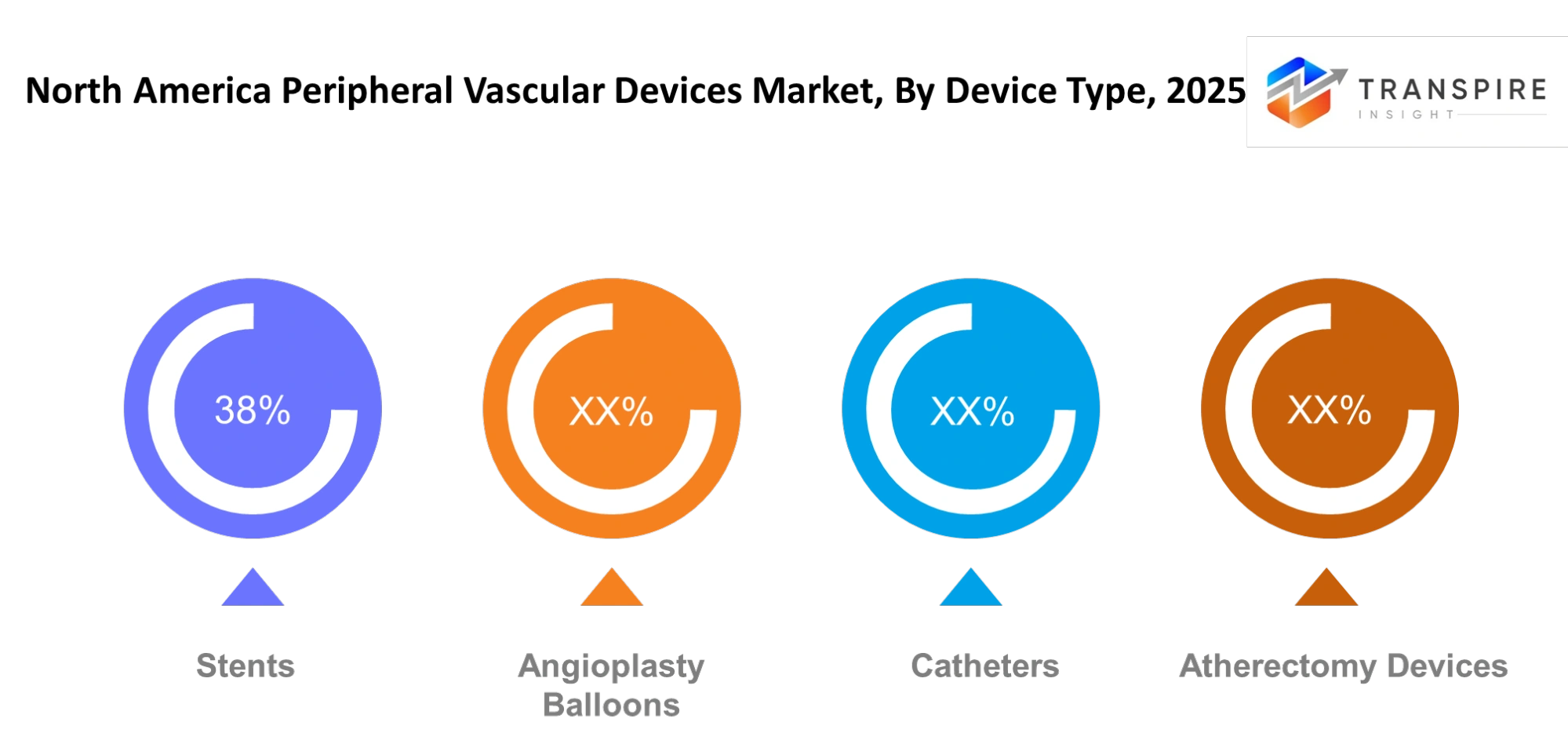
By Device Type
Angioplasty Balloons- Angioplasty balloons will remain an important tool for restoring circulation through narrowed vessels. The design of the balloon will continue to be improved, making it more flexible and having a drug-coated surface, thus preventing restenosis. Physicians will increasingly use these devices to perform exact dilation procedures which are necessary for handling difficult peripheral lesions that need controlled expansion without harming blood vessels.
Stents- Stents will also gain increasing clinical preference as long-term vessel support becomes essential in peripheral interventions. New developments, such as self-expanding and drug-eluting stents, will help increase durability and reduce repeat interventions. 3 Physicians will select stents based on lesion type and vessel location, which will result in the best treatment results and continuous blood circulation throughout the affected sections.
Catheters- Catheters will serve as the foundation for most vascular procedures, which enables medical professionals to traverse through complex arterial pathways. The improved steerability of devices together with their enhanced imaging capabilities will result in better accuracy during medical procedures. Healthcare providers will increasingly rely on special catheter designs which medical teams developed for particular medical procedures to minimize complications while ensuring successful medical device operations.
Atherectomy Devices- The clinical application of atherectomy devices will increase because they provide effective solutions for plaque removal from peripheral arteries. These tools will allow targeted plaque excision while preserving healthy tissue. The development of rotational and laser-based systems will provide higher accuracy for treating heavily calcified lesions which require advanced treatment beyond standard balloon angioplasty methods.
To learn more about this report, Download Free Sample Report
By Application
Peripheral Artery Disease- Peripheral artery disease will remain the principal field of research because its cases increase with the aging population and changing lifestyle patterns. The medical field will focus on two treatment methods which include early medical treatment and methods that require less invasive medical procedures. The selection of devices will depend on the level of disease progression which allows doctors to treat circulation problems while minimizing both potential complications and permanent physical impairment.
Venous Diseases- Venous diseases will represent a steadily expanding application segment as awareness improves and diagnostic capabilities advance. The medical field will use less invasive techniques to treat conditions that include both varicose veins and deep vein thrombosis. Surgical devices will develop to achieve three goals which include better control and improved patient comfort and faster recovery times compared to current surgical methods.
By End-User
Hospitals- Hospitals will maintain their position as primary end-users because they possess advanced infrastructure and highly trained specialists. Complex vascular procedures will largely be performed in these settings, where multidisciplinary teams can manage high-risk patients. The hospital's capacity to conduct peripheral interventions will expand through its investment in imaging systems and hybrid operating room systems.
Ambulatory Surgical Centers- The demand for ambulatory surgical centers will increase because patients want to receive medical treatment that costs less and takes less time to complete. The centers will specialize in conducting minimally invasive surgical procedures which enable patients to leave the hospital within a short period after their operations. The hospital will expand its operations because improved device efficiency and simplified techniques enable physicians to perform vascular procedures with less operational workload while delivering greater convenience to patients.
Regional Insights
The United States will achieve superior regional performance through its commitment to developing vascular care infrastructure and implementing advanced clinical practices. Hospitals will establish advanced imaging systems together with medical devices to enhance their operational effectiveness during procedures. The healthcare industry will use reimbursement policies to determine which devices will get approval for use in urban and rural medical facilities, but patients will continue to prefer minimally invasive treatment options.
Canada will achieve gradual advancement through its publicly funded healthcare system, which provides patients with accessible medical services and prioritizes their health results over time. The medical field will decide which peripheral vascular devices to adopt after studying their clinical performance and economic benefits. Healthcare providers will focus on diagnosing conditions at early stages while establishing preventive healthcare programs, which will enable patients to access modern medical treatments, through controlled financial resources for health services in different provinces.
Healthcare modernization initiatives in Mexico will create better access to vascular treatment services, which will drive healthcare system growth in the country. The private sector will take the lead in bringing innovative medical devices to the market, while public medical facilities will develop their technology capabilities at a slower pace. More patients will begin treatment at medical facilities because they understand vascular diseases better, which will lead to increased adoption of less invasive treatments in major urban areas.
North American healthcare organizations, regulatory bodies, and device manufacturers will work together to develop better treatment options for patients. Device development and approval processes will be influenced by knowledge sharing and clinical research between countries. The different healthcare systems across the region will create distinct adoption trends, but all countries will maintain their dedication to patient safety and better health results, which will drive regional changes forward.
Recent Development News
United States: Device Approvals and Commercial Launch Momentum-The recent activities of the United States demonstrate that advanced vascular technologies receive strong regulatory approval while their initial commercial use begins. The FDA approved new carotid stent systems and iliac branch devices for clinical use in 2025 which enhanced treatment accuracy for difficult medical situations. The developments establish the country as an advanced medical center which will launch upcoming peripheral medical treatments.
Canada: Evidence-Driven Adoption and Clinical Integration- Canada will maintain its vascular treatment system development by following scientific research evidence on treatment methods. The healthcare system will start using new minimally invasive medical devices while assessing their patient results and financial efficiency. Innovative medical devices which enter the North American market will create standardized treatment procedures across Canadian provinces because clinical research results and actual product performance data will determine procurement choices.
Mexico: Expanding Access Through Private Healthcare Investment- Private hospitals in Mexico will increase their operational activities through their facility upgrades and investment projects. Major urban areas will receive advanced vascular devices because diagnostic methods have improved and public knowledge has increased about these medical tools. The change will make minimally invasive medical treatments more accessible to patients who want to recover quickly and have better treatment experiences in private medical facilities.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 4314.8 Million |
|
Market size value in 2026 |
USD 4526.4 Million |
|
Revenue forecast in 2033 |
USD 6409.9 Million |
|
Growth rate |
CAGR of 5.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Terumo Corporation, Becton Dickinson and Company, Cook Medical LLC, Cardinal Health Inc., Biotronik SE & Co. KG, Koninklijke Philips N.V., Siemens Healthineers AG, Stryker Corporation, AngioDynamics Inc., Penumbra Inc., Teleflex Incorporated, Merit Medical Systems Inc. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Device Type (Angioplasty Balloons, Stents, Catheters, Atherectomy Devices), By Application (Peripheral Artery Disease, Venous Diseases), By End-User (Hospitals, Ambulatory Surgical Centers) |
Key North America Peripheral Vascular Devices Market Company Insights
Key players shaping the competitive landscape of the North America (Canada, the United States, and Mexico) Peripheral Vascular Devices Market will include major medical technology firms such as Medtronic, Boston Scientific, Abbott, and Becton Dickinson, alongside specialized companies like Cook Medical and Terumo. The organizations will develop their business through three main strategies which include expanding their minimally invasive product lines and enhancing their clinical research capabilities and making strategic acquisitions to improve their product offerings and market coverage.
Company List
- Medtronic plc
- Boston Scientific Corporation
- Abbott Laboratories
- Terumo Corporation
- Becton Dickinson and Company
- Cook Medical LLC
- Cardinal Health Inc.
- Biotronik SE & Co. KG
- Koninklijke Philips N.V.
- Siemens Healthineers AG
- Stryker Corporation
- AngioDynamics Inc.
- Penumbra Inc.
- Teleflex Incorporated
- Merit Medical Systems Inc.
North America Peripheral Vascular Devices Market Report Segmentation
By Device Type
- Angioplasty Balloons
- Stents
- Catheters
- Atherectomy Devices
By Application
- Peripheral Artery Disease
- Venous Diseases
By End-User
- Hospitals
- Ambulatory Surgical Centers
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America Peripheral Vascular Devices Market size for the market will be USD 6409.9 Million in 2033.
Key segments for the North America Peripheral Vascular Devices Market By Device Type (Angioplasty Balloons, Stents, Catheters, Atherectomy Devices), By Application (Peripheral Artery Disease, Venous Diseases), By End-User (Hospitals, Ambulatory Surgical Centers).
Major players in the North America Peripheral Vascular Devices Market are Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Terumo Corporation, Becton Dickinson and Company, Cook Medical LLC, Cardinal Health Inc., Biotronik SE & Co. KG, Koninklijke Philips N.V., Siemens Healthineers AG, Stryker Corporation, AngioDynamics Inc., Penumbra Inc., Teleflex Incorporated, Merit Medical Systems Inc.
The North America Peripheral Vascular Devices Market size is USD 4314.8 Million in 2025.
The North America Peripheral Vascular Devices Market CAGR is 5.10%.
- Medtronic plc
- Boston Scientific Corporation
- Abbott Laboratories
- Terumo Corporation
- Becton Dickinson and Company
- Cook Medical LLC
- Cardinal Health Inc.
- Biotronik SE & Co. KG
- Koninklijke Philips N.V.
- Siemens Healthineers AG
- Stryker Corporation
- AngioDynamics Inc.
- Penumbra Inc.
- Teleflex Incorporated
- Merit Medical Systems Inc.
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









