North America Catheter Stabilization Device Market Size & Forecast:
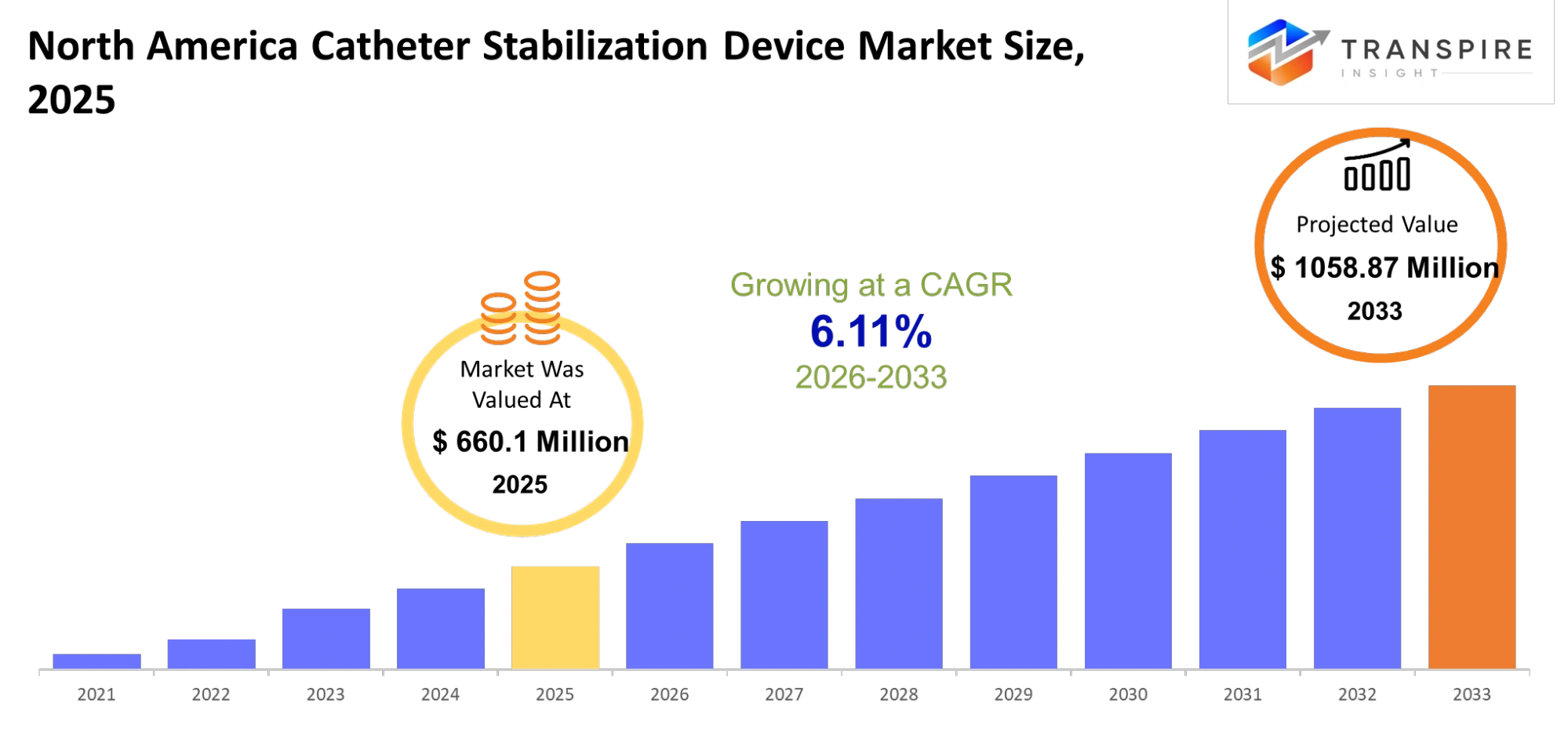
- North America Catheter Stabilization Device Market Size 2025: USD 660.1 Million
- North America Catheter Stabilization Device Market Size 2033: USD 1058.87 Million
- North America Catheter Stabilization Device Market CAGR: 6.11%
- North America Catheter Stabilization Device Market Segments: By Type (Adhesive Devices, Sutureless Devices, Securement Devices, Anchor Devices, Others), By Application (IV Catheters, Urinary Catheters, Central Venous Catheters, PICC Lines, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Material (Silicone, Foam, Plastic, Composite, Others).
To learn more about this report, Download Free Sample Report
North America Catheter Stabilization Device Market Summary:
The North America Catheter Stabilization Device Market size is estimated at USD 660.1 Million in 2025 and is anticipated to reach USD 1058.87 Million by 2033, growing at a CAGR of 6.11% from 2026 to 2033. North America Catheter Stabilization Device Market shows strong growth because hospitals and outpatient facilities need safe effective methods to manage vascular access.
Hospital devices which hospitals use to stabilize catheters help decrease the number of dislodged catheters and catheter-related infections and skin irritation problems which lead to better patient results.. The adoption of advanced stabilization solutions has been increasing because healthcare systems now prioritize both patient safety and cost efficiency.
The North America Catheter Stabilization Device Market has gained from increased clinician knowledge about securement methods which clinicians use for extended medical treatments. The North America Catheter Stabilization Device Market has received another major driving force through the increasing rate of chronic diseases which create a need for extended catheterization. The market expands because new technological developments bring double-sided adhesives which safely attach to skin and provide modern methods to secure medical equipment.
What Has the Impact of Artificial Intelligence Been on the North America Catheter Stabilization Device Market?
AI technology is reshaping the North America Catheter Stabilization Device Market because it enables companies to analyze their market presence and develop solutions for upcoming medical needs. The North America Catheter Stabilization Device Market uses AI technology to perform data mining which enables organizations to evaluate hospital and clinical data more precisely.
Through predictive analytics and machine learning systems businesses use their market knowledge to forecast upcoming market developments while determining product development needs. The data-driven system helps North America Catheter Stabilization Device Market stakeholders make better decisions which results in measurable market advantages.
North America Catheter Stabilization Device Market operations use artificial intelligence for process improvement through automation systems that enhance both manufacturing and supply chain operations. AI-driven systems enhance manufacturing accuracy which results in reduced material waste and better quality control while decreasing costs and improving product delivery speed.
Inventory optimization and demand forecasting use predictive analytics to decrease supply chain interruptions. The North America Catheter Stabilization Device Market will experience continuous market expansion because of these innovations which create new personalized device solutions for healthcare providers who adopt intelligent technologies.
Key Market Trends & Insights:
- The north america catheter stabilization device market size is expanding due to increasing chronic disease prevalence and higher demand for long-term catheterization solutions across healthcare facilities.
- The need to minimize catheter-related infections drives medical facilities to implement advanced securement devices which provide better safety features and improved patient results.
- North America catheter stabilization device market development now shows increased competitive advantage because technological advancements create better skin-safe adhesives together with antimicrobial protection material.
- The demand for portable stabilization devices which are easy to use has grown because outpatient services and home healthcare services have increased during recent times.
- The United States controls the north america catheter stabilization device market because it holds more than 85 of market share which will reach 2025 because of its developed healthcare system.
- Canada represents the fastest-growing region, with a projected CAGR above 7% through 2030, driven by expanding healthcare access and rising investments.
- Arterial securement devices control the market with their existing share which exceeds 40 because they receive common application in both critical care settings and surgical operations.
- The second largest market share for central venous catheter stabilization devices exists because more patients require ICU treatment together with extended medical procedures.
- The section which includes peripheral catheter stabilization devices will grow at the fastest rate between now and 2030 because outpatient procedures will increase.
- The hospital sector operates as the primary application field because it delivers more than 65 of market share through its capacity to handle high patient traffic and perform numerous medical procedures.
North America Catheter Stabilization Device Market Segmentation
By Type
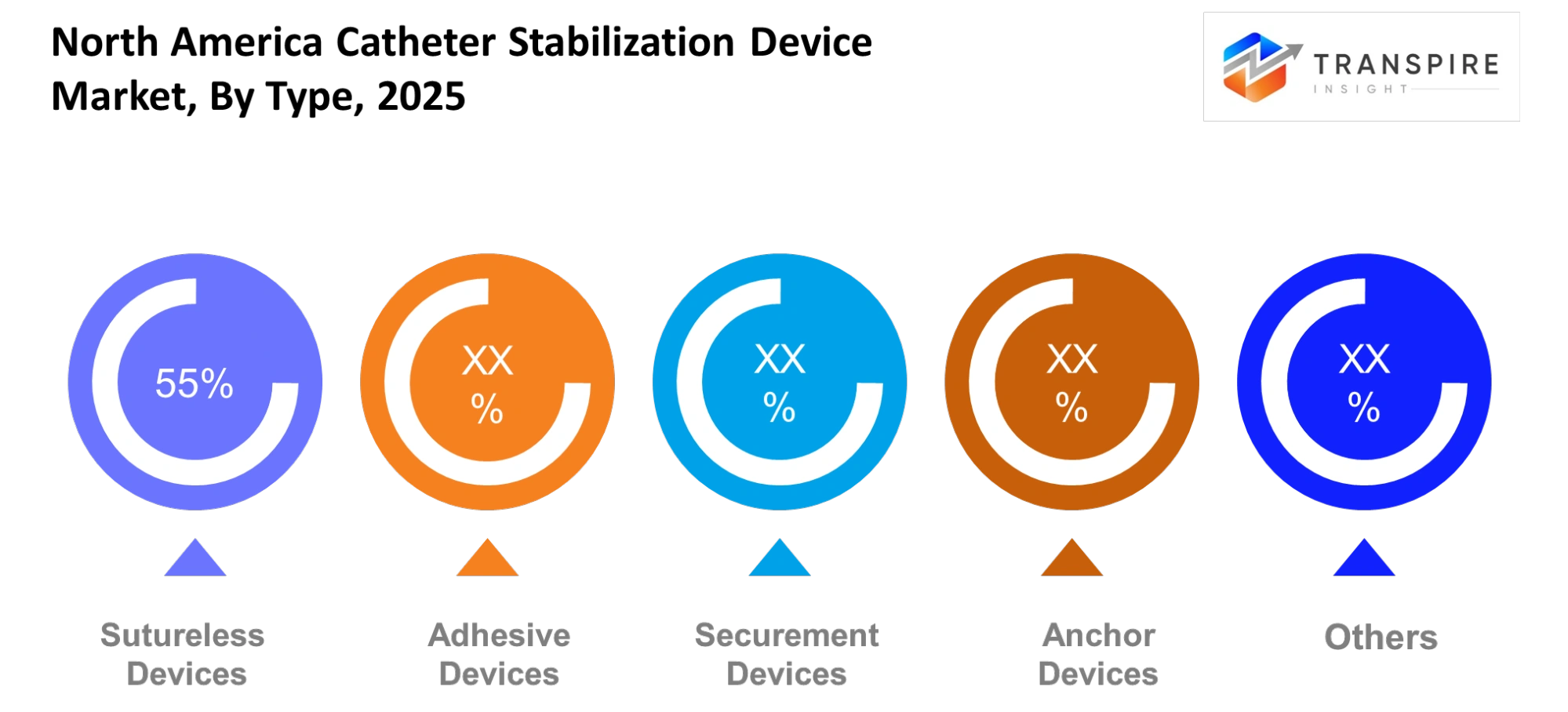
Adhesive devices maintain strong market demand because they can be applied easily while their use results in lower risk of skin damage during catheter fixation. Healthcare facilities now prefer sutureless devices because they help reduce infection risks while providing better patient comfort. Securement devices maintain their stability during extended catheter use which enables them to function properly in clinical environments.
Anchor devices establish secure placement under high movement conditions while other devices meet specific clinical requirements in specialized medical procedures. The segment will experience growth because people become more aware of infection prevention methods which lead to improved patient health outcomes.
Product development for adhesives and skin-friendly materials will enhance their usability while maintaining safety standards. Healthcare providers will choose solutions which decrease complications and streamline their operational processes. The demand patterns show that all device categories will experience continuous growth while customers prefer non-invasive solutions.
To learn more about this report, Download Free Sample Report
By Application
IV catheters account for a major share because hospitals use them widely to deliver fluids and administer medication. Urinary catheters maintain steady demand in long-term care and post-surgical recovery cases. Central venous catheters enable critical care treatments through their capability to provide intensive patient monitoring and drug administration. PICC lines show rising adoption for extended therapies while other applications serve specific medical procedures that need secure catheter placement.
The increase in patient admissions together with the rise of chronic disease cases will result in higher usage across all application areas. Treatment durations together with complex therapies will create a demand for stabilization solutions that need to provide reliable performance. Healthcare systems will focus on improving safety and efficiency through their catheter management procedures. The growth trends show that both acute and long-term care settings have begun to adopt new technologies at high rates.
By End User
The primary reason hospitals lead in service usage is their capacity to handle numerous patients who need their advanced facilities to perform various catheter-based medical procedures. The combination of outpatient services and regular treatments at clinics creates a continuous need for clinics which require efficient methods to maintain patient stability.
The number of patients receiving homecare treatment continues to grow because medical facilities now permit more people to obtain care at home. Other end-users include specialized care centers which manage medical needs that require special treatment. The healthcare system requires more space because patients now prefer outpatient services and cost-effective treatment solutions which drive up their need for homecare and office-based treatment facilities.
The healthcare system requires more space because hospitals need advanced medical equipment to perform complicated procedures which require multiple medical devices. The healthcare system requires more space because hospitals need advanced medical equipment to perform complicated procedures which require multiple medical devices. The healthcare system requires more space because hospitals need advanced medical equipment to perform complicated procedures. The healthcare system needs continuous market growth which shows balanced development across all user groups.
By Material
Patients with delicate skin requirements and those who need extended wear time can use silicone materials because they provide both adaptable properties and skin-safe features. The use of foam-based products enables users to experience cushioning effects which help them maintain comfortable conditions while reducing pressure complications during catheter insertion. Plastic materials maintain their widespread application because they offer both strong durability and economical performance across common usage scenarios.
Composite materials provide multiple advantages through their combination of different material properties while other materials deliver essential performance requirements for specific applications.Material science advancements will create better product performance results which will enhance patient safety throughout this entire product category.
The market will see increased demand for materials which provide better skin comfort protection while improving adhesive strength. Healthcare providers will choose materials which enable extended usage periods while maintaining product effectiveness throughout their entire duration. The development of new business possibilities will result from advancements in composite materials and hybrid material technologies.
What are the Main Challenges for the North America Catheter Stabilization Device Market Growth?
The north america catheter stabilization device market encounters substantial technical and operational problems which impede delivery of reliable product results. The system suffers from multiple problems which include insufficient adhesion and skin irritation and device displacement that restrict its dependable operation in medical environments. The system faces additional challenges because it needs to work with multiple catheter designs and different patient treatment requirements. The north america catheter stabilization device market experiences its major operational challenges because supply chain disturbances and raw material shortages create efficiency drops.
The north america catheter stabilization device market experiences pressure from manufacturing and commercialization barriers. The strict regulatory requirements demand compliance with quality standards and safety protocols which results in extended production timeframes and increased production expenses. Manufacturers need to spend large amounts of their resources for testing and validation work because this testing process serves as the main requirement for product approval. The combination of cost limitations and pricing competition creates challenges for smaller companies who want to expand their business operations.
The north america catheter stabilization device market continues to face adoption problems which create major obstacles for healthcare facilities that lack advanced technology. Device usage decreases when healthcare professionals lack device knowledge and training programs do not provide consistent instruction. The availability of infrastructure and budget constraints in specific facilities result in slow adoption processes. The market needs better education programs which should accompany funding assistance and simplified implementation methods.
Market competition and future risks continue to influence the north america catheter stabilization device market outlook. The existence of alternative securement methods together with new technological developments creates market competition which affects both pricing and product innovation. Manufacturers face operational challenges because of two factors which include rapid technological advancements and changing regulatory environments. The business requires ongoing product development together with strategic market positioning in order to achieve sustainable growth throughout the upcoming years.
Country Insights
The United States maintains the highest market share because its healthcare systems deliver advanced medical services and its hospitals treat many patients and its infection control measures combat the spread of diseases. The demand will continue to grow because chronic disease rates and surgical procedure needs will rise. The ongoing development of new products together with positive reimbursement systems will generate continuous market expansion for hospitals and outpatient medical facilities.
Canada maintains a steady growth pattern because healthcare access increases and people become more aware of patient safety standards. The demand for medical services will rise because hospitals will improve their infrastructure and work to minimize complications that stem from catheter use. The government will enhance stabilization device usage in medical facilities and home healthcare settings through its healthcare service funding programs.
The remaining North American regions show slow development because healthcare facilities improve and patients want better treatment results. The use of modern medical devices will grow because people learn about these devices and they become more accessible. The market expansion and sustained demand will depend on both economic growth and healthcare funding.
Recent Development News
In April 2026, Merit Medical acquires imaging oncology company View Point Medical for $140M: Merit Medical Systems completed the acquisition of View Point Medical in a deal valued at approximately $140 million, strengthening its position in interventional and diagnostic device solutions. The acquisition adds advanced tumour localisation technologies, supporting expansion in catheter-related procedures and oncology care.
Source: https://www.massdevice.com
In January 2026, Smith & Nephew to Acquire U.S.-Based Integrity Orthopaedics in $450 Million Deal: Smith & Nephew announced the acquisition of U.S.-based Integrity Orthopaedics to strengthen its orthopaedic and catheter-related treatment portfolio. The deal is expected to enhance innovation in surgical repair systems and improve patient outcomes across U.S. healthcare settings.
Source: https://www.reuters.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 660.1 Million |
|
Market size value in 2026 |
USD 699.05 Million |
|
Revenue forecast in 2033 |
USD 1058.87 Million |
|
Growth rate |
CAGR of 6.11% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
3M, BD, Medtronic, B. Braun, Smith & Nephew, Cardinal Health, Baxter, Teleflex, ConvaTec, Coloplast, Cook Medical, Merit Medical, AngioDynamics, Vygon, ICU Medical |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Adhesive Devices, Sutureless Devices, Securement Devices, Anchor Devices, Others), By Application (IV Catheters, Urinary Catheters, Central Venous Catheters, PICC Lines, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Material (Silicone, Foam, Plastic, Composite, Others). |
How Can New Companies Establish a Strong Foothold in the North America Catheter Stabilization Device Market?
New entrants can build a strong position in the north america catheter stabilization device market by targeting unmet clinical needs and focusing on niche applications such as pediatric care or long-term home treatment. The company will establish its unique market position through innovation strategies which include skin-friendly adhesives and antimicrobial features to solve infection risk and device displacement problems.
The company will achieve faster product launch times and increased market credibility through its early strategy creation process which starts with industry trend analysis and regulatory requirement development. The emerging players need to develop focused strategies because established competitors in their markets create challenges that they must overcome. The north america catheter stabilization device market will experience growth through technology-driven differentiation.
Smart automation with data-driven capabilities which include usage tracking and predictive maintenance functions enables companies to enhance their product value. AdheSafe Medical (illustrative) will create an AI-powered adhesive system that adapts to different patient skin conditions while SecureCath Innovations (illustrative) will develop sustainable materials that maintain exceptional strength. The innovation-led strategies will establish powerful market positions that will enable sustainable business development throughout time.
The north america catheter stabilization device market will experience market expansion through strategic partnerships with hospitals and research institutions and distributors. The speed of clinical validation processes will increase through partnerships while healthcare providers will develop greater confidence in the results. The combination of flexible manufacturing systems with local supply chain networks will enable companies to decrease expenses and better meet changing customer demands.
Key North America Catheter Stabilization Device Market Company Insights
The established companies use their broad product offerings and their successful distribution networks and their established relationships with doctors to keep their dominant market share. The organization will maintain its position as an industry leader through ongoing dedication to safety requirements and clinical excellence. The organization will improve product reliability through its continuous research development efforts which will result in positive outcomes for its brand image.
The leading companies in the market use their advanced adhesive technologies and their antimicrobial product attributes and their designs which provide ease of use to patients in order to enhance their products. The organization will create research-based solutions which will decrease infection problems and improve user experience during long-term catheter use. The organization will develop effective and durable products which will enable it to maintain its existing market position while competing against other companies.
The organization will expand its market presence through improved product testing when it establishes partnerships with hospitals and research institutions and distribution companies. The partnerships will fast track new technology adoption while building confidence with medical professionals. Through strong partnerships companies can improve their supply chain processes while managing their growing customer demand.
New competitors enter the market because they offer affordable specialized solutions which create higher competitive challenges for the entire industry. Small companies will attract more customers through their specialized niche products and their ability to produce items through adaptable manufacturing methods. Existing businesses must improve their pricing methods while delivering steady product quality to compete against growing market competition.
Company List
- 3M
- BD
- Medtronic
- B. Braun
- Smith & Nephew
- Cardinal Health
- Baxter
- Teleflex
- ConvaTec
- Coloplast
- Cook Medical
- Merit Medical
- AngioDynamics
- Vygon
- ICU Medical
What are the Key Use-Cases Driving the Growth of the North America Catheter Stabilization Device Market?
The north america catheter stabilization device market exists because hospitals need these devices to maintain their patients' intravenous catheter systems for continuous medication and fluid treatment. The medical field requires dependable stabilization products because hospitals need to treat more patients and perform more surgical operations. The devices protect against dislodgement and infections which creates better clinical results and operational productivity for intensive care environments.
The north america catheter stabilization device market exists because patients with chronic diseases like cancer and cardiovascular disorders need secure catheter systems for their treatment needs. Patients who need central venous catheters and PICC lines for their medical treatment need advanced securement devices to provide secure catheter positioning for extended treatment duration. The home healthcare industry expansion has resulted in more patients receiving medical treatment in their homes which creates a need for products that combine easiness of use with strong performance.
The north america catheter stabilization device market exists because hospitals need these devices to manage urinary catheters for their post-surgical patients and elder patients. The right stabilization techniques help patients recover better because they reduce the risk of both irritation and accidental catheter removal. Hospitals and long-term care facilities now adopt infection prevention protocols because healthcare workers better understand these protocols.
The North America catheter stabilization device market is experiencing growth because outpatient clinics and ambulatory surgical centers create new market possibilities. The need for efficient catheter management solutions has increased because hospitals now prioritize affordable healthcare delivery and brief patient stays. The development of materials and design innovations will enable healthcare facilities to expand their operations into new business opportunities.
North America Catheter Stabilization Device Market Report Segmentation
By Type
- Adhesive Devices
- Sutureless Devices
- Securement Devices
- Anchor Devices
- Others
By Application
- IV Catheters
- Urinary Catheters
- Central Venous Catheters
- PICC Lines
- Others
By End-User
- Hospitals
- Clinics
- Homecare
- Others
By Material
- Silicone
- Foam
- Plastic
- Composite
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America Catheter Stabilization Device Market size for the market will be USD 1058.87 Million in 2033.
The key segments of the North America Catheter Stabilization Device Market are By Type (Adhesive Devices, Sutureless Devices, Securement Devices, Anchor Devices, Others), By Application (IV Catheters, Urinary Catheters, Central Venous Catheters, PICC Lines, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Material (Silicone, Foam, Plastic, Composite, Others).
Major players in the North America Catheter Stabilization Device Market are 3M, BD, Medtronic, B. Braun, Smith & Nephew, Cardinal Health, Baxter, Teleflex, ConvaTec, Coloplast, Cook Medical, Merit Medical, AngioDynamics, Vygon, ICU Medical.
The current market size of the North America Catheter Stabilization Device Market is USD 660.1 Million in 2025.
The North America Catheter Stabilization Device Market CAGR is 6.11%.
- 3M
- BD
- Medtronic
- B. Braun
- Smith & Nephew
- Cardinal Health
- Baxter
- Teleflex
- ConvaTec
- Coloplast
- Cook Medical
- Merit Medical
- AngioDynamics
- Vygon
- ICU Medical
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)