Japan Stem Cell Therapy Market Size & Forecast:
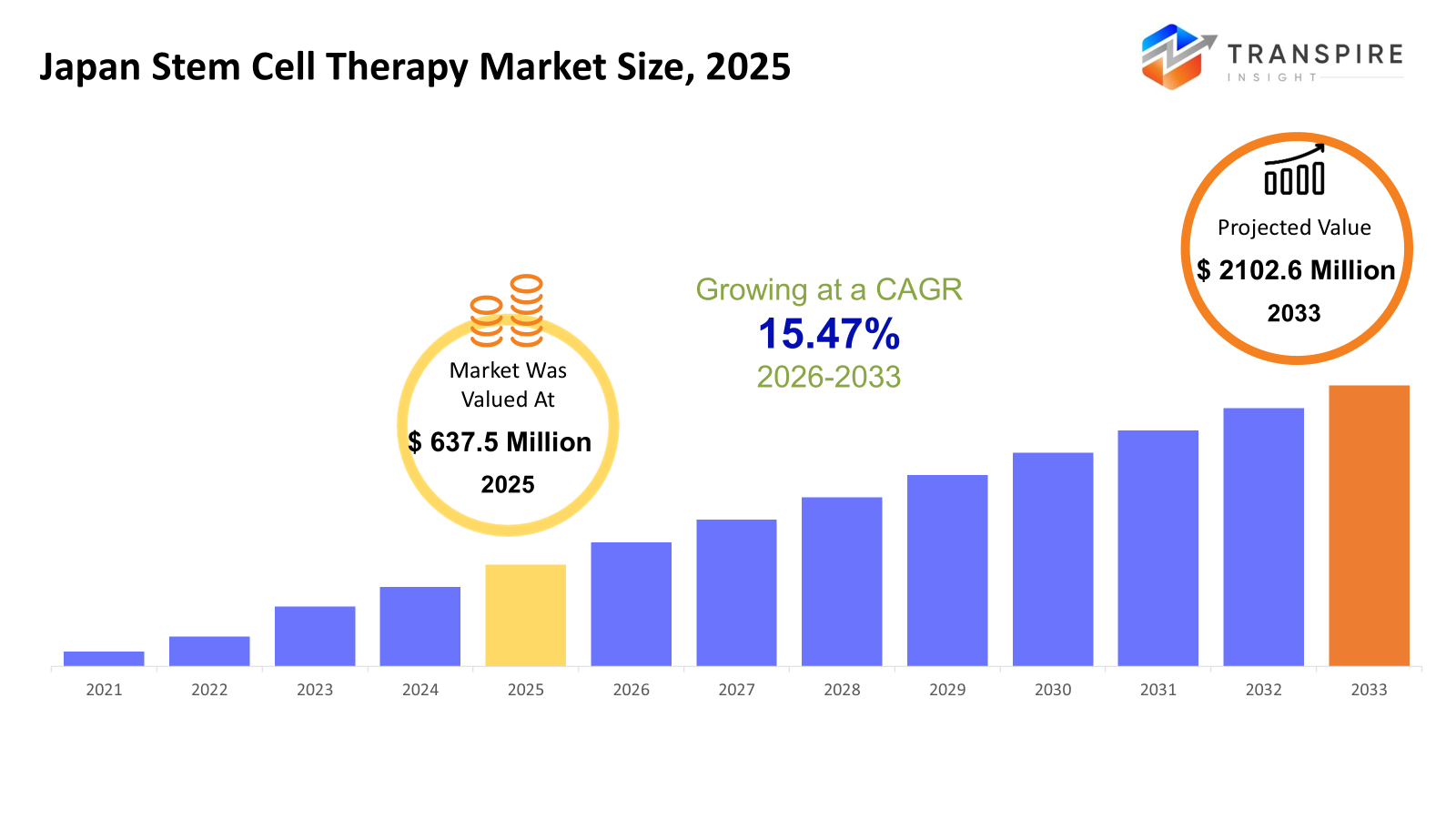
- Japan Stem Cell Therapy Market Size 2025: USD 637.5 Million
- Japan Stem Cell Therapy Market Size 2033: USD 2102.6 Million
- Japan Stem Cell Therapy Market CAGR: 15.47%
- Japan Stem Cell Therapy Market Segments:By Therapy Type (Autologous Stem Cell Therapy, Allogeneic Stem Cell Therapy), By Application (Oncology, Orthopedics, Neurology, Cardiovascular), By End User (Hospitals, Research Institutes).
To learn more about this report, Download Free Sample Report
Japan Stem Cell Therapy Market Summary:
The Japan Stem Cell Therapy Market size is estimated at USD 637.5 Million in 2025 and is anticipated to reach USD 2102.6 Million by 2033, growing at a CAGR of 15.47% from 2026 to 2033. Japan's stem cell therapy market is experiencing strong growth driven by the country's advanced biotechnology ecosystem and its supportive regulatory framework. Japan has established itself as the first nation worldwide to develop regenerative medicine through its fast-track approval system for new medical treatments. The market expansion is being driven by increasing needs for medical treatments that address chronic diseases and ageing-related disorders and orthopedic conditions. The ongoing research activities of universities and biotech companies, together with healthcare organisations, will develop new clinical uses for their findings. Japan is becoming a central centre for stem cell research and business development as both investment and collaborative efforts continue to rise.
Key Market Trends & Insights:
- The Government of Japan supports regenerative medicine through its comprehensive funding programmes and progressive regulatory framework, which accelerates research in this field. The fast-track approval system enables early patient access to safe stem cell therapies that show therapeutic potential.
- The increasing need for medical treatments for degenerative diseases stems from Japan's oldest population in the world. Stem cell therapies show potential to treat arthritis, neurological disorders and cardiovascular diseases.
- Japanese universities and hospitals and biotech companies are currently conducting clinical trials across the country. The goals of the research are the formation of new treatment methodologies based on stem cells for oncology or ophthalmology and tissue regeneration.
- Japan holds the top position in the world because it developed a technology which enables scientists to generate stem cells through the conversion of adult cells. This innovation opens new pathways to develop customised medical solutions which provide safer methods for regenerative medical procedures.
- Pharmaceutical companies, together with research institutes and biotech startups, are establishing partnerships to drive their innovation process. The collaborations enable companies to produce their products at larger volumes while achieving better scientific results and shorter timeframes for delivering new stem cell treatments.
Japan Stem Cell Therapy Market Segmentation
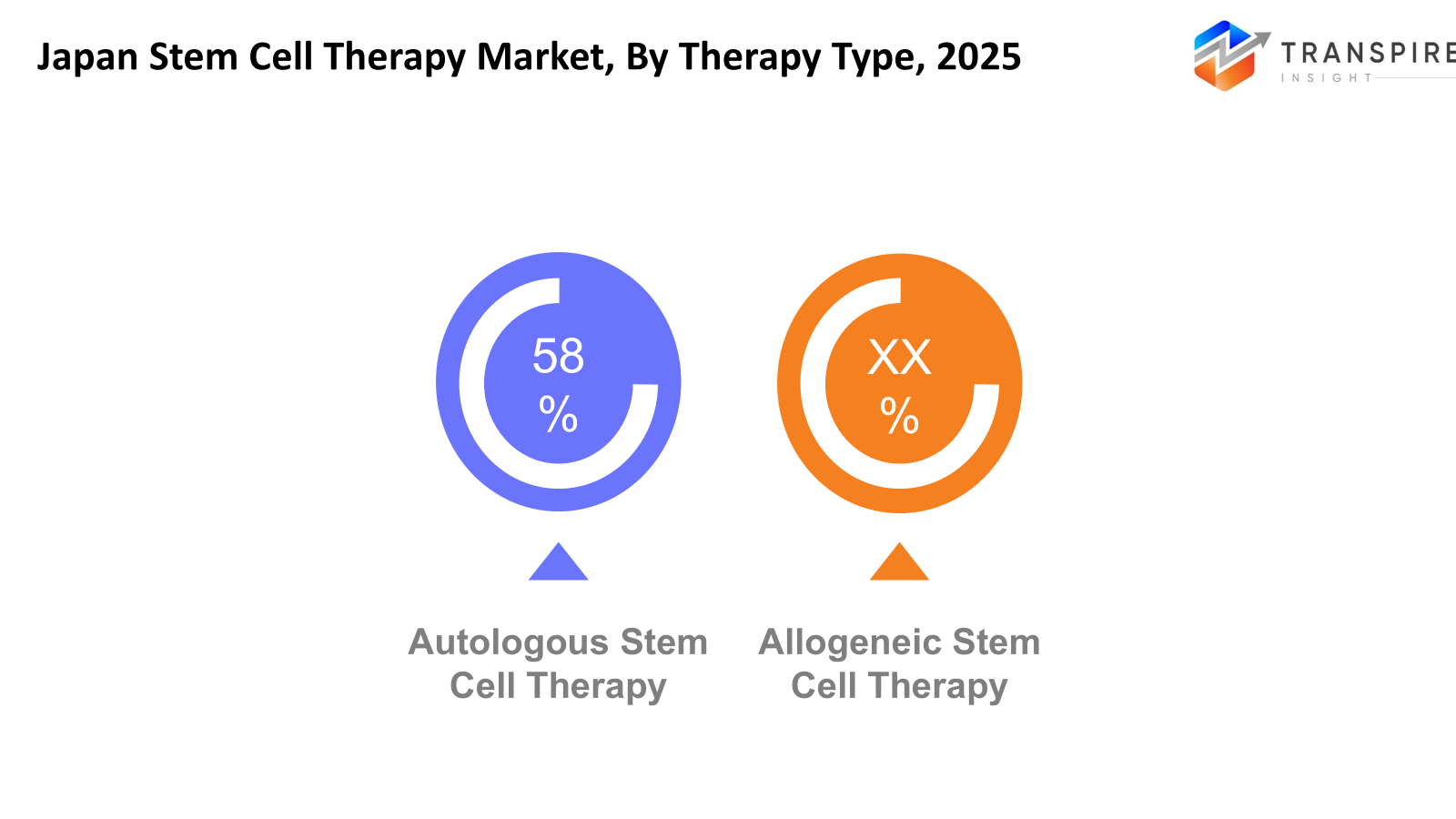
By Therapy Type
- Autologous Stem Cell Therapy: The procedure of autologous stem cell therapy requires patients to use their own stem cells, which results in a treatment that has minimal chances of their body rejecting the transplanted material. In Japan, this approach is widely explored for orthopaedic and regenerative treatments. Hospitals and clinics are expanding its medical uses because research activities and regulatory support work together to create new treatment possibilities.
- Allogeneic Stem Cell Therapy: Allogeneic stem cell therapy requires the use of stem cells that medical professionals obtain from healthy individuals. This method is gaining attention in Japan for treating blood disorders, cancers, and certain genetic conditions. The development of new cell processing and storage technologies results in better treatment results because ongoing research projects establish its importance to regenerative medicine practices.
To learn more about this report, Download Free Sample Report
By Application
- Oncology: In Japan, oncological medicine uses stem cell therapy to treat patients who need bone marrow transplants for leukaemia and other blood cancers. Researchers investigate stem cell-based therapies which enhance immune response and support tissue healing as new methods to increase survival rates and improve patient health.
- Orthopedics: Japan experiences fast growth in various medical fields which use stem cell therapy for orthopedic treatment purposes. The medical community uses these therapies to assist patients who need their cartilage and bone and joint injuries treated. Researchers investigate regenerative therapies as treatment choices for elderly patients who develop joint disorders instead of recommending them to undergo surgical procedures or use medication for extended periods.
- Neurology: Stem cell research in neurology has reached its maximum advancement stage in Japan. Scientists are developing treatments for Parkinson's disease, spinal cord injuries and stroke recovery. Researchers use advanced clinical trials and innovative technologies to investigate methods of restoring damaged neural tissues and enhancing patient recovery results.
- Cardiovascular: The Japanese healthcare system considers cardiovascular diseases as critical medical problems which result in increased research activities for stem cell treatments that restore heart tissue. Researchers investigate how regenerative cells can enhance heart function after heart attacks, which enables patients to recover quicker while decreasing their risk of long-lasting health issues.
By End User
- Hospitals: The hospitals in Japan deliver stem cell therapies as their primary medical service. The major medical facilities in the country maintain special treatment areas which their staff use to perform high-end regenerative medical procedures. The rising public understanding of stem cell treatments, together with their increased use in medical practice, drives hospitals to develop stem cell treatment protocols for their existing medical practices.
- Research Institutes: Japanese research institutes serve as the leading centres for stem cell research and development efforts. The universities and research centres conduct comprehensive research projects to design therapies which provide improved safety and efficacy. The research team develops induced pluripotent stem cells and regenerative medicine solutions which will create new directions for future industry development.
Country Insights
Japan has developed into a worldwide stem cell therapy market leader, which relies on its scientific research capabilities, its modern medical facilities and its forward-thinking regulatory framework. The country was among the first to create a fast-track approval system for regenerative medicines, which enables safe delivery of new treatments to patients at an accelerated pace. Japanese research on induced pluripotent stem cell (iPSC) technology has opened the door to the development of personalised regenerative medical treatments. Academic institutions, biotech companies and healthcare facilities are working together to develop clinical treatment methods that will benefit patients with cancer and neurological disorders, orthopaedic problems, and heart diseases. The increasing elderly population in Japan needs new medical treatments for their chronic progressive health conditions. The government provides funding and private sector investments to support research activities and commercialisation initiatives. Japan creates an innovation ecosystem that enables clinical advancements and establishes its position as a worldwide leader in stem cell therapy.
Recent Development News
Japan’s Regenerative Medicine Sector Gains Momentum with New Stem Cell Treatment Developments.
Japan Grants Conditional Approval to New Stem Cell Therapies for Parkinson’s and Heart Failure.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 637.5 Million |
|
Market size value in 2026 |
USD 768.2 Million |
|
Revenue forecast in 2033 |
USD 2102.6 Million |
|
Growth rate |
CAGR of 15.47% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Takeda Pharmaceutical Company, Fujifilm Cellular Dynamics, Astellas Pharma Inc., Mesoblast Limited, Vericel Corporation, Osiris Therapeutics, Pluristem Therapeutics, Gamida Cell Ltd., STEMCELL Technologies, Lonza Group AG, Thermo Fisher Scientific, Novartis AG, Sanofi SA, Bristol Myers Squibb, Roche Holding AG. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Therapy Type (Autologous Stem Cell Therapy, Allogeneic Stem Cell Therapy), By Application (Oncology, Orthopedics, Neurology, Cardiovascular), By End User (Hospitals, Research Institutes). |
Key Japan Stem Cell Therapy Company Insights
The Japan stem cell therapy market contains a vibrant ecosystem which includes biotechnology companies, pharmaceutical manufacturers and research-based startups who develop regenerative medicine solutions. Companies are working to create new treatments through their development of induced pluripotent stem cell technologies and their advanced cell processing capabilities. The development of clinical research and product development processes receives support through strategic partnerships between companies and educational institutions and healthcare research centres. In addition to their research efforts, companies develop production processes which include scalable manufacturing and quality control systems to prepare their products for market entry. The combination of advanced research capabilities and favourable regulatory frameworks enables Japanese industry stakeholders to develop new solutions and expand treatment options.
Company List
- Takeda Pharmaceutical Company
- Fujifilm Cellular Dynamics
- Astellas Pharma Inc.
- Mesoblast Limited
- Vericel Corporation
- Osiris Therapeutics
- Pluristem Therapeutics
- Gamida Cell Ltd.
- STEMCELL Technologies
- Lonza Group AG
- Thermo Fisher Scientific
- Novartis AG
- Sanofi SA
- Bristol Myers Squibb
- Roche Holding AG.
Japan Stem Cell Therapy Market Report Segmentation
By Therapy Type
- Autologous Stem Cell Therapy
- Allogeneic Stem Cell Therapy
By Application
- Oncology
- Orthopedics
- Neurology
- Cardiovascular
By End User
- Hospitals
- Research Institutes
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Japan Stem Cell Therapy Market size for the Market will be USD 2102.6 Million in 2033.
The key Segments of the Japan Stem Cell Therapy Market are By Therapy Type (Autologous Stem Cell Therapy, Allogeneic Stem Cell Therapy), By Application (Oncology, Orthopedics, Neurology, Cardiovascular), By End User (Hospitals, Research Institutes).
Major Players in the Japan Stem Cell Therapy Market are Takeda Pharmaceutical Company, Fujifilm Cellular Dynamics, Astellas Pharma Inc., Mesoblast Limited, Vericel Corporation, Osiris Therapeutics, Pluristem Therapeutics, Gamida Cell Ltd., STEMCELL Technologies, Lonza Group AG, Thermo Fisher Scientific, Novartis AG, Sanofi SA, Bristol Myers Squibb, Roche Holding AG.
The Current Market size of the Japan Stem Cell Therapy Market is USD 637.5 Million in 2025.
The Japan Stem Cell Therapy Market CAGR is 15.47%.
- Takeda Pharmaceutical Company
- Fujifilm Cellular Dynamics
- Astellas Pharma Inc.
- Mesoblast Limited
- Vericel Corporation
- Osiris Therapeutics
- Pluristem Therapeutics
- Gamida Cell Ltd.
- STEMCELL Technologies
- Lonza Group AG
- Thermo Fisher Scientific
- Novartis AG
- Sanofi SA
- Bristol Myers Squibb
- Roche Holding AG.
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


