Japan Artificial Blood Substitutes Market Size & Forecast:
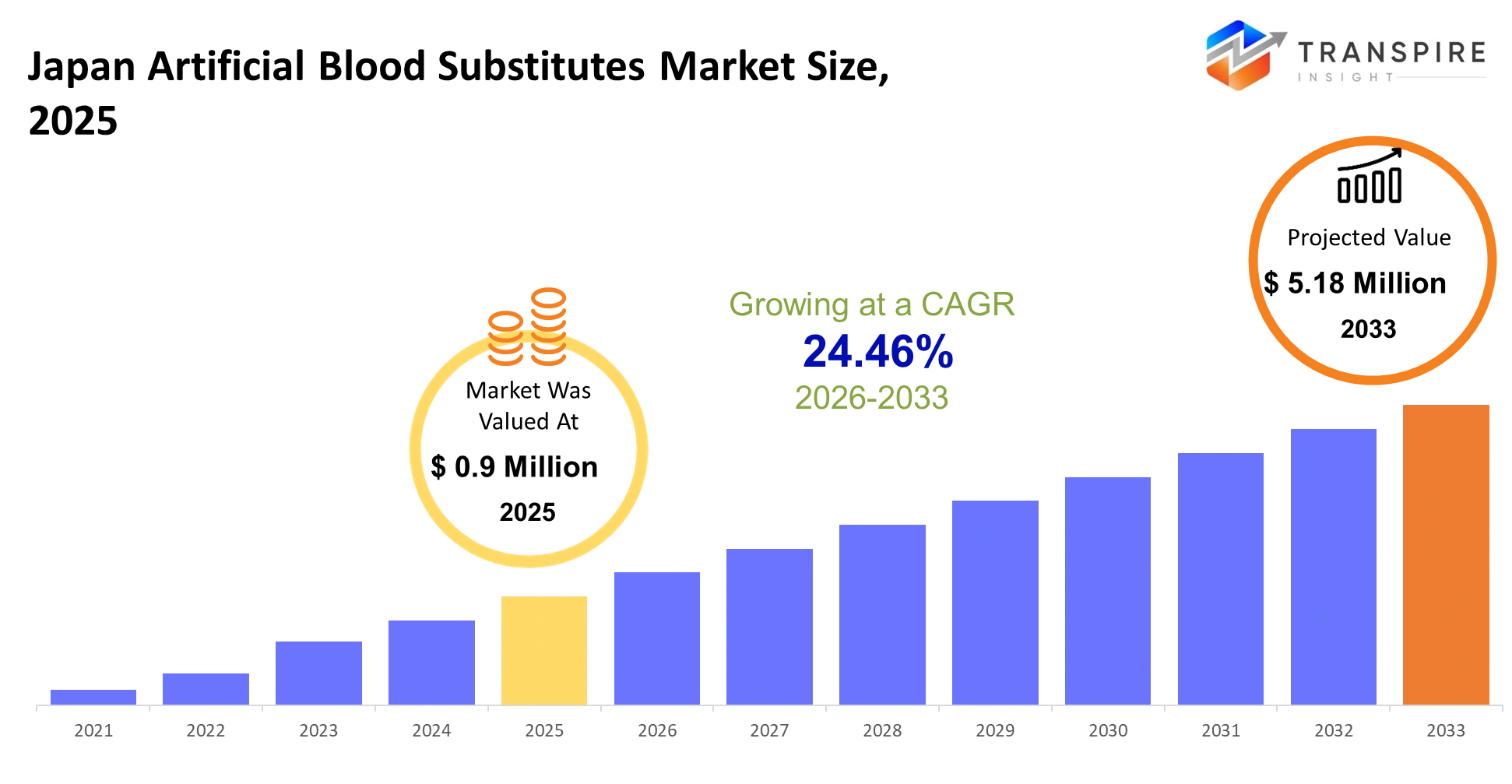
- Japan Artificial Blood Substitutes Market Size 2025: USD 0.9 Million
- Japan Artificial Blood Substitutes Market Size 2033: USD 5.18 Million
- Japan Artificial Blood Substitutes Market CAGR: 24.46%
- Japan Artificial Blood Substitutes Market Segments: By Type (Hemoglobin-based, Perfluorocarbon-based, Synthetic Oxygen Carriers, Recombinant Products), By Application (Trauma, Surgery, Emergency, Military), By End-User (Hospitals, Military, Research Institutes, Emergency Services)
To learn more about this report, Download Free Sample Report
Japan Artificial Blood Substitutes Market Summary:
The Japan Artificial Blood Substitutes Market size is estimated at USD 0.9 Million in 2025 and is anticipated to reach USD 5.18 Million by 2033, growing at a CAGR of 24.46% from 2026 to 2033. The artificial blood solutions market in Japan exists because three factors drive demand which include the need for advanced medical treatments and the increasing blood supply and transfusion safety concerns. The research field makes progress because advanced research institutions exist together with government institutions which support biomedical innovation.
The Japanese market has not reached full commercial potential because it remains in its development stage which requires businesses to meet strict regulatory standards while they conduct ongoing clinical trials for product safety and efficacy testing. The process of widespread adoption faces obstacles which include the need to spend significant resources on product development and the possibility of experiencing side effects and the need to address ethical issues. Pharmaceutical companies and research institutes and government bodies join forces to create new products which lead to faster development. The Japanese market will experience slow growth because technological advancements increase the safety and effectiveness of artificial blood substitutes. The emergency care field and military medicine and remote healthcare systems will drive market growth because these areas lack access to conventional blood supply systems.
Key Market Trends & Insights:
- The research teams in Japan are investigating HBOC and PFC technologies because they require blood alternatives which must be both safe and effective. The academic institutions and biotech companies work together to create better methods of oxygen delivery which will reduce negative effects.
- The country requires artificial blood substitutes because its aging population and decreasing number of blood donors create this need. The demographic changes require urgent development of permanent blood transfusion alternatives which will work in all medical environments.
- The Japanese market needs to follow strict regulatory standards which determine how fast companies can bring new products to market. The regulations require companies to meet high safety and efficacy standards which extend their clinical trial durations because organizations must spend money on compliance testing and validation processes.
- The pharmaceutical industry, research institutions, and government agencies establish strategic alliances to drive their research initiatives. The partnerships between organizations enable them to share their expertise, receive financial backing, and accelerate the development of advanced artificial blood products which will be used in surgical procedures and trauma care.
- Artificial blood substitutes became more stable and longer lasting because scientists used nanotechnology and biotechnology together to create better oxygen-carrying materials. The developments will make these solutions suitable for use in remote medical facilities, military applications, and emergency response operations which need immediate access to blood products.
Japan Artificial Blood Substitutes Market Segmentation
By Type
- Hemoglobin-based: This system uses modified hemoglobin as oxygen transport agents which function throughout human body systems. The system receives extensive research attention because it replicates natural blood functions yet its primary application exists in trauma and surgical environments. The system faces development challenges because of safety concerns and adverse effects which hinder its approval process by regulatory agencies.
- Perfluorocarbon-based: Synthetic compounds known as perfluorocarbon (PFC) products enable efficient oxygen delivery through their capacity to dissolve oxygen. Patients must breathe high oxygen concentrations to achieve maximum treatment results with these products. The products maintain emergency and military operational readiness through their extended shelf life and stable performance which helpwith blood storage and compatibility issues.
- Synthetic Oxygen Carriers: The system creates synthetic compounds which function as oxygen transport systems that do not use any human or animal materials. Their research aims to develop superior storage solutions which improve safety and material compatibility. The efficiency of new technologies in nanotechnology and biomaterials research has reached a level where these materials can be used in upcoming medical applications at large scale.
- Recombinant Products: Scientists use genetic engineering methods to create recombinant artificial blood substitutes which produce hemoglobin and other protein-based products. The products deliver high purity results which decrease the chance of contamination. The research process for these solutions currently exists at initial stages, yet they show potential to deliver safe oxygen delivery solutions which can expand to large operations while maintaining consistent performance.

To learn more about this report, Download Free Sample Report
By Application
- Trauma: The essential role of artificial blood substitutes in trauma treatment provides immediate oxygen delivery during situations that need immediate blood loss control. The product becomes perfect for emergency situations because it works with all blood types and provides fast access which eliminates the need for blood typing and cross-matching testing during urgent medical situations.
- Surgery: The surgical process depends on these substitutes to deliver oxygen during blood loss situations when donor blood remains inaccessible. The system provides advantages to complex surgeries because it improves patient stability while minimizing the complications linked to standard blood transfusions.
- Emergency : The use of artificial blood substitutes provides emergency medical services with a benefit because these products can be easily transported and they maintain their usability for extended periods. The product enables better survival rates for emergency medical situations which occur in ambulances and areas that lack quick access to donor blood.
- Military: The military sector depends on artificial blood substitutes because they allow medical treatment in combat zones where soldiers cannot store blood products. The product enables medical personnel to treat wounded soldiers on the battlefield because it maintains its strength and compatibility with all systems.
By End-User
- Hospitals: Hospitals serve as the main users because they need to provide surgical operations and treatment for accidents and emergency medical services. The use of artificial blood substitutes enables medical professionals to improve treatment outcomes while decreasing transfusion complications and maintaining stable oxygen delivery systems.
- Military: Military medical facilities use these blood substitutes for both their operational activities and their medical treatment operations. Their ability to function without strict storage conditions makes them highly suitable for emergency care in remote and combat environments.
- Research Institutes: Research institutions play a vital role in developing and testing artificial blood technologies. Their research work focuses on three main areas: safety improvements, efficacy enhancements, and scalability development. This research work will help to build future market innovations which will lead to commercial products.
- Emergency Services: Emergency responders use artificial blood substitutes for immediate medical treatment which includes their use by ambulance services and disaster response teams. The equipment operates effectively as life-saving tools because it can be deployed within seconds and requires almost no setup time during emergency situations.
Country Insights
The Japanese market for artificial blood substitutes derives its strength from two main factors which include the nation's advanced healthcare system and its dedication to biomedical research. The country maintains an established research ecosystem which includes academic institutions and pharmaceutical firms and government programs that work together to create new medical technologies. Japanese research in regenerative medicine and biotechnology has advanced research development for oxygen therapeutics which includes hemoglobin-based and recombinant medical products. Government sustainability projects for healthcare systems and emergency response training initiatives are driving funding toward new blood transfusion technologies which replace existing systems.
Japan's market development derives from its aging demographic situation which results in decreased blood donor availability. The existing blood supply system faces increased emergency danger because of the current treatment requirement which requires medical resources. Healthcare providers and policymakers increasingly evaluate artificial blood substitutes because these products provide dependable and easily implemented solutions. The market only permits high-security products which meet all safety and efficacy requirements according to strict regulatory standards that govern the approval process. Japan will strengthen its artificial blood research position through ongoing clinical trials and public-private partnerships which will lead to gradual market expansion.
Recent Development News
In 2025, Japan became the first country to start human clinical trials for artificial blood which Nara Medical University conducted. The innovation uses hemoglobin vesicles derived from expired donor blood which provides universal compatibility and extended shelf life to create a system that will transform emergency transfusion procedures.
In 2026, Japanese researchers started their Phase Ib clinical research in 2026 to evaluate the safety and drug absorption characteristics of hemoglobin vesicle-based artificial blood on healthy adult participants. The current research evaluates both the effectiveness and various medical applications of the technology, which brings the development process nearer to commercial availability.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 0.9 Million |
|
Market size value in 2026 |
USD 1.12 Million |
|
Revenue forecast in 2033 |
USD 5.18 Million |
|
Growth rate |
CAGR of 24.46% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Hemarina, Sanguine Biosciences, OPK Biotech, Hemopure, Biopure, Baxter, Pfizer, Roche, Takeda, Astellas, Otsuka, Terumo, Daiichi Sankyo, Mitsubishi Pharma, Sumitomo Pharma |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Hemoglobin-based, Perfluorocarbon-based, Synthetic Oxygen Carriers, Recombinant Products), By Application (Trauma, Surgery, Emergency, Military), By End-User (Hospitals, Military, Research Institutes, Emergency Services) |
Key Japan Artificial Blood Substitutes Market Company Insights
The Japan artificial blood substitutes market includes international biotechnology companies and pharmaceutical companies and local healthcare organizations which all work together to develop and market their products. The companies Hemarina and OPK Biotech through their link to Hemopure via Hemoglobin Oxygen Therapeutics lead the development of oxygen therapeutics by creating new hemoglobin-based and marine-derived oxygen carriers. The firms use their advanced research and development efforts together with innovative biomaterials and their clinical testing programs to create better oxygen delivery systems which provide safer and more effective ways to deliver oxygen. The companies Sanguine Biosciences and Biopure worked together to develop and market hemoglobin-based substitutes through their early development efforts but some of their products are restricted to controlled and compassionate use programs.
The major pharmaceutical and healthcare companies support research through their collaborative partnerships with clinical facilities while they provide funding to research projects. The Japanese companies Terumo and Takeda establish major market power through their extensive domestic operations which focus on medical device production and biopharmaceutical development. The involvement of these major companies in adjacent technologies and regulatory processes and large-scale manufacturing operations creates a stronger artificial blood product ecosystem for research. The market operates as a research-driven environment where specialized biotech companies create new innovations which established pharmaceutical companies use to develop safe artificial blood products for clinical applications.
Company List
- Hemarina
- Sanguine Biosciences
- OPK Biotech
- Hemopure
- Biopure
- Baxter
- Pfizer
- Roche
- Takeda
- Astellas
- Otsuka
- Terumo
- Daiichi Sankyo
- Mitsubishi Pharma
- Sumitomo Pharma
Japan Artificial Blood Substitutes Market Report Segmentation
By Type
- Hemoglobin-based
- Perfluorocarbon-based
- Synthetic Oxygen Carriers
- Recombinant Products
By Application
- Trauma
- Surgery
- Emergency
- Military
By End-User
- Hospitals
- Military
- Research Institutes
- Emergency Services
Frequently Asked Questions
Find quick answers to common questions.
The approximate Japan Artificial Blood Substitutes Market size for the market will be USD 5.18 Million in 2033.
Key segments for the Japan Artificial Blood Substitutes Market are By Type (Hemoglobin-based, Perfluorocarbon-based, Synthetic Oxygen Carriers, Recombinant Products), By Application (Trauma, Surgery, Emergency, Military), By End-User (Hospitals, Military, Research Institutes, Emergency Services).
Major Japan Artificial Blood Substitutes Market players are Hemarina, Sanguine Biosciences, OPK Biotech, Hemopure, Biopure, Baxter, Pfizer, Roche, Takeda, Astellas, Otsuka, Terumo, Daiichi Sankyo, Mitsubishi Pharma, Sumitomo Pharma.
The Japan Artificial Blood Substitutes Market size is USD 0.9 Million in 2025.
The Japan Artificial Blood Substitutes Market CAGR is 24.46%.
- Hemarina
- Sanguine Biosciences
- OPK Biotech
- Hemopure
- Biopure
- Baxter
- Pfizer
- Roche
- Takeda
- Astellas
- Otsuka
- Terumo
- Daiichi Sankyo
- Mitsubishi Pharma
- Sumitomo Pharma
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
May 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









