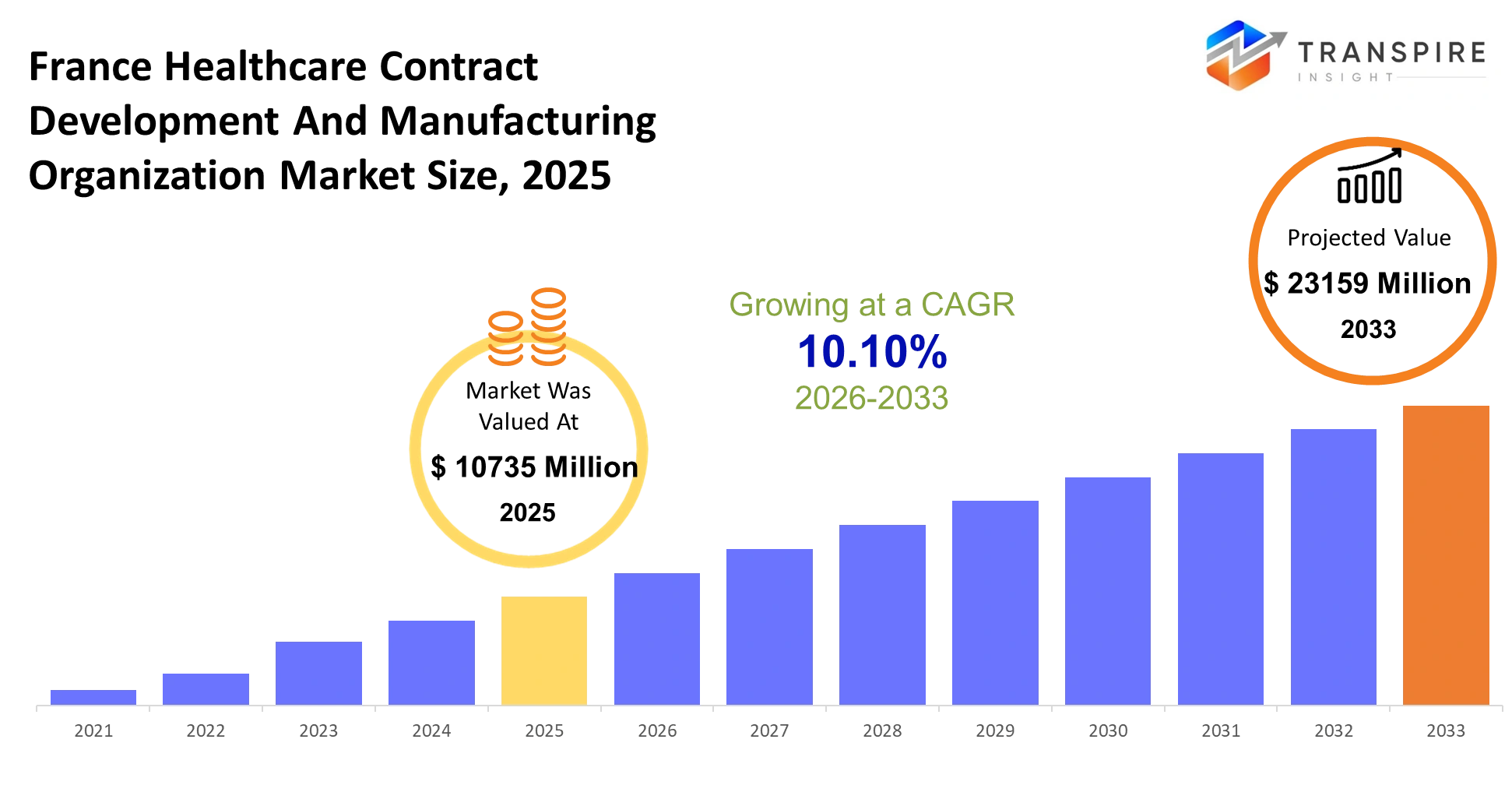
France Healthcare Contract Development And Manufacturing Organization Market Size & Forecast:
- France Healthcare Contract Development And Manufacturing Organization Market Size 2025: USD 10735 Million
- France Healthcare Contract Development And Manufacturing Organization Market Size 2033: USD 23159 Million
- France Healthcare Contract Development And Manufacturing Organization Market CAGR: 10.10%
- France Healthcare Contract Development And Manufacturing Organization Market Segments: By Type (API Manufacturing, Biologics Manufacturing, Packaging Services, Others); By Application (Drug Development, Clinical Trials, Commercial Production, Biologics, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Scale (Preclinical, Clinical, Commercial, Others)
To learn more about this report, Download Free Sample Report
France Healthcare Contract Development And Manufacturing Organization Market Summary
The France Healthcare Contract Development And Manufacturing Organization Market was valued at USD 10735 Million in 2025. It is forecast to reach USD 23159 Million by 2033. That is a CAGR of 10.10% over the period.
The France Healthcare Contract Development and Manufacturing Organization market supports pharmaceutical companies by handling drug development, clinical-scale production, and large-scale manufacturing under strict regulatory compliance. The system enables biotech companies and pharmaceutical developers to speed up their drug launch processes without needing to create their own manufacturing facilities. The system functions as an essential support network which enables vaccine production and biologics development and specialized drug manufacturing throughout the entire healthcare industry.
Biologics and advanced therapy manufacturing now dominate the market because production pipelines have shifted away from traditional small-molecule methods during the past three to five years. The COVID-19 pandemic acted as a major trigger, exposing supply chain vulnerabilities and accelerating outsourcing to CDMO partners for faster vaccine production and decentralized manufacturing capacity. The industry developed increased ties with manufacturing facilities that maintained advanced skills for meeting regulatory requirements. Pharmaceutical companies now drive industry expansion by using outsourced services and they require production facilities that can match their changing needs which has resulted in longer contract durations and better financial stability for CDMO companies.
Key Market Insights
- The market for healthcare contract development and manufacturing organizations in France shows significant growth because companies are outsourcing biologics production and the demand for advanced therapeutic manufacturing is increasing.
- The drug substance manufacturing sector will maintain a 42% market share in 2025 because it needs to produce large quantities of biologics and vaccines.
- The formulation scaling and packaging outsourcing trends establish drug product manufacturing as the second largest market segment.
- The cell and gene therapy manufacturing industry will experience its fastest growth rate until 2033 because personalized medicine development creates more demand for these therapies.
- Oncology applications dominate demand with over 35% share because cancer biologics development continues to grow.
- The fastest growing field of research focuses on infectious diseases because vaccine readiness initiatives are being implemented throughout Europe.
- Over 55% of total outsourcing requirements come from pharmaceutical companies which serve as the primary customer base.
- Biotech firms experience the quickest growth among end-user segments because they lack sufficient manufacturing capabilities for production.
- The main companies in this market include Lonza, Catalent, Recipharm, Samsung Biologics, Thermo Fisher Scientific, and Fareva.
- Companies are expanding their operations through three methods which include building new facilities and upgrading their EU sites and establishing partnerships for biologics development.
What are the Key Drivers, Restraints, and Opportunities in the France Healthcare Contract Development And Manufacturing Organization Market?
The France CDMO market develops because pharmaceutical and biotechnology companies choose to outsource their complex drug development and production activities. The industry shift started when biologics became more complex and European Medicines Agency regulations demanded companies to establish high-quality compliance systems. CDMO partners now provide companies with production capacity to meet their needs which leads to higher contract values and faster revenue growth for manufacturers who possess advanced biologics production skills.
The main limitation exists because GMP-compliant biologics manufacturing facilities need substantial financial investment to build. The production of cell therapies and monoclonal antibodies requires extended time periods for both equipment development and mandatory regulatory testing. The system creates an entry barrier which hinders market newcomers while delaying operational capacity growth. The system causes temporary supply shortages because customer demand increases faster than businesses can expand their operational capacity.
The manufacturing capacity of cell and gene therapy in France presents a significant opportunity which will benefit the biotech clusters located in Île-de-France and Lyon. The government life sciences investment programs along with EU funding dedicated to advanced therapies are driving infrastructure development progress. CDMOs that choose to invest in modular and flexible biologics production facilities will achieve a competitive edge by establishing contracts with emerging biotech companies which need manufacturing support.
What Has the Impact of Artificial Intelligence Been on the France Healthcare Contract Development And Manufacturing Organization Market?
The French CDMO market experiences transformation through artificial intelligence which enhances manufacturing accuracy and quality assessment and operational productivity throughout pharmaceutical supply chains. AI-driven automation operates throughout process monitoring systems which monitor batch consistency and equipment performance and regulatory compliance in real time. The systems automate production processes which enable facilities to maintain GMP compliance during biologics manufacturing operations.
Companies now use predictive analytics to enhance their equipment maintenance processes and optimize their production operations. Machine learning models use historical production data to forecast equipment breakdowns and determine optimal batch scheduling while minimizing unplanned downtime in manufacturing facilities. The system enables continuous operations while enhancing facility usage during periods of high demand for biologics production. AI helps optimize vaccine production processes and monoclonal antibody production processes through its ability to find efficiency gaps in fermentation and purification stages.
The operational enhancements produce three benefits which decrease production costs through enhanced yield consistency and faster quality release periods and decreased batch failure rates. The high integration expenses combined with existing fragmented manufacturing systems in older CDMO facilities create obstacles for AI implementation. The strict pharmaceutical audit standards demand regulatory validation to approve algorithm-driven decisions which creates delays in AI technology implementation. AI systems maintain their essential position as fundamental technologies which enable development of future pharmaceutical manufacturing methods.
Key Market Trends
- The share of biologics manufacturing increased by 18% from 2022 to 2025 because European markets experienced growing demand for monoclonal antibody production.
- The 2025 expansion of cell and gene therapy manufacturing capacity advanced because contract development and manufacturing organizations dedicated resources to building modular good manufacturing practice certified cleanroom spaces.
- Vaccine production contracts experienced growth after 2023 because governments established stronger pandemic emergency supply chain partnerships through their improved agreements.
- CDMO companies experienced a major increase in pharmaceutical outsourcing because companies decreased their internal manufacturing investments for production equipment.
- The European biologics production facilities of Lonza and Catalent expanded their capacity to produce complex drug substances because of the increasing demand from customers.
- Recipharm increased its budget for constructing sterile injectable manufacturing facilities throughout France to enable the company to develop high-value pharmaceutical products.
- The implementation of AI-based manufacturing monitoring systems resulted in CDMO plants achieving better batch quality control while decreasing production process errors.
- The European pharmaceutical market has increased its need for oncology drug manufacturing because cancer biologics pipelines from European pharmaceutical companies continue to expand.
- The EU GMP regulatory rules created harmonized regulations which allowed CDMO companies to work together across borders while standardizing their contract agreements.
- Samsung Biologics increased its global CDMO capabilities which created higher competition in the European biologics outsourcing industry.
France Healthcare Contract Development And Manufacturing Organization Market Segmentation
By Type
The France Healthcare Contract Development and Manufacturing Organization market by type is led by biologics manufacturing due to the strong shift toward complex therapies such as monoclonal antibodies, vaccines, and advanced biologics. The segment has obtained its greatest market share because pharmaceutical companies now prefer to hire specialized CDMO providers for their expensive biologics production needs which require extensive facilities. The production of active pharmaceutical ingredients maintains its vital role in small-molecule drug development while providing ongoing support to both branded and generic pharmaceutical manufacturing. The demand for packaging services remains constant because European Union pharmaceutical safety standards require companies to implement serialization, labeling accuracy, and compliance measures.
Biologics manufacturing continues to gain dominance as oncology and immunotherapy pipelines expand across Europe. The process of biologics production demands facilities with advanced technology which small-scale manufacturers cannot afford to operate and thus must depend on external service providers. The growth of API manufacturing will proceed because companies will implement cost reduction methods together with establishing supply chains that use multiple regions instead of relying on a single area. Packaging services develop consistently because new requirements for product traceability emerge while more customers demand packaging solutions that ensure their safety during medical treatment. The forecast period will show biologics manufacturing increasing its market dominance while packaging and active pharmaceutical ingredient services become essential components of complete value chains within CDMO networks.
By Application
Pharmaceutical companies now tend to outsource their initial drug creation work because it helps them decrease research and development time. The market segment establishes a strong position because it enables companies to move their research work into clinical studies at a quicker pace while they spend less on their research facilities. Clinical trials also provide essential support because their design for testing biologics and rare disease treatments has become more complex. Commercial production serves as an essential process for developing approved medicines especially in biologics, which require exact manufacturing standards, and specialty pharmaceuticals.
The fastest growth in biologics applications occurs because more people demand new medical solutions like cell and gene therapies. Biotech companies increasingly depend on CDMOs for clinical trial production because these organizations offer them both flexible batch manufacturing and needed regulatory documentation. Companies now choose to outsource their drug development work because it helps them develop new products faster and save money. The commercial production process will become more important because the number of biologics approvals increases while clinical and development areas will continue to serve as innovation pathways for new treatment options.
To learn more about this report, Download Free Sample Report
By End-User
Pharmaceutical companies maintain control over the end-user market because they outsource their manufacturing needs for both small-molecule drugs and biologic products. The industry maintains this dominance because companies use cost-saving methods while facing demands for faster drug development processes. The biotech industry maintains rapid expansion because these companies require contract manufacturing organizations to fulfill their clinical and commercial production needs. Research institutes also contribute to demand through their drug discovery activities and their cooperative development work with contract manufacturers.
The fastest growth in biotech firms occurs because they develop new treatments for oncology and immunology and rare diseases. Biotechs create new business opportunities through their use of CDMO solutions which provide both flexible and scalable manufacturing facilities. The pharmaceutical industry maintains its market dominance through its manufacturing agreements and international distribution networks. Research institutes conduct essential work to establish the foundation for validation and translational research processes. The forecast period will see biotech firms gain more power to direct CDMO service development while pharmaceutical companies will maintain their market share through volume-based methods.
By Scale
The France CDMO market size reaches its largest point through commercial scale manufacturing which needs high-volume production of approved pharmaceutical drugs and biologic medicines. The segment operates through long-term supply contracts which CDMOs establish with pharmaceutical companies to obtain required production capacity for their ongoing requirements. The manufacturing process for clinical-scale operations follows this because clinical trials have increased together with the demand for phase II and phase III production services. The preclinical manufacturing process serves a vital role because it supports essential activities in drug development and product formulation.
The need for sophisticated trial pipelines which handle both biologics and personalized therapies has led to rapid growth in clinical-scale manufacturing. Biotech startups use CDMOs to perform their initial formulation testing and feasibility assessments which drives consistent growth in preclinical service demand. Commercial-scale production remains the primary source of revenue because it relies on existing supply contracts and the high demand for biologics products. The importance of clinical and preclinical segments will increase as innovation pipelines develop while commercial manufacturing will stay as the core method for generating consistent revenue across the CDMO network.
What are the Key Use Cases Driving the France Healthcare Contract Development And Manufacturing Organization Market?
Biologics manufacturing serves as the primary application that drives the France CDMO industry because pharmaceutical companies need to produce monoclonal antibodies and vaccines at dedicated production facilities. The demand for this service exists because biotech companies and major pharmaceutical firms need to produce complex biologics but lack their own facilities that meet GMP standards. The use case generates the highest demand because production requires advanced bioreactors, strict regulatory validation, and continuous quality control.
The development of cell and gene therapies is an expanding field which biotechnology companies that focus on oncology and research institutions connected to hospitals support. CDMOs possess manufacturing capabilities that meet the requirements of these therapies because they need to produce small quantities of specialized products. Public health procurement programs create a reliable demand for vaccine production outsourcing through their ongoing vaccine purchasing activities.
The market now contains two emerging use cases which involve manufacturing personalized medicine and producing mRNA-based therapeutics. These applications are gaining traction in advanced research pipelines, particularly in precision oncology and rare disease treatment programs.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 10735 Million |
|
Market size value in 2026 |
USD 11809 Million |
|
Revenue forecast in 2033 |
USD 23159 Million |
|
Growth rate |
CAGR of 10.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Lonza, Catalent, Thermo Fisher, Recipharm, WuXi AppTec, Samsung Biologics, Boehringer Ingelheim, AGC Biologics, Evonik, Fareva, Piramal Pharma, Siegfried, Jubilant Pharmova, AbbVie, Pfizer CentreOne |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (API Manufacturing, Biologics Manufacturing, Packaging Services, Others); By Application (Drug Development, Clinical Trials, Commercial Production, Biologics, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Scale (Preclinical, Clinical, Commercial, Others) |
Which Regions are Driving the France Healthcare Contract Development And Manufacturing Organization Market Growth?
The France CDMO market centers around Île-de-France because the region has multiple pharmaceutical headquarters and biotech startups and regulatory institutions headquartered there. The region benefits from its capacity to implement EU pharmaceutical standards together with its capability to process approvals at a rapid pace. The advanced research hospitals and biotech clusters generate ongoing requirements for companies that provide clinical and commercial manufacturing services. The ecosystem enables production of high-value biologics while it quickens the process that innovators use to establish partnerships with CDMO service providers.
The Auvergne-Rhône-Alpes region functions as a dependable secondary center which receives its strength from its industrial resources and its historical background in pharmaceutical production. The region maintains its dedication to sterile injectable production together with its backing of small-molecule manufacturing support services. The region develops through industrial operations that take place over extended periods instead of utilizing research advancements to build new business activities which is different from Île-de-France. The CDMO operations maintain their efficiency throughout multiple production cycles because of the strong logistics network together with the established pharmaceutical workforce.
The fastest-growing area in Provence-Alpes-Côte d'Azur experienced growth through new life sciences investment incentives and expanded biotech incubation programs. The new funding programs which the region introduced after 2024 have financed facility improvements which support biologics and advanced therapy production. The industrial appeal of the area has increased because of improved transport options which connect with Mediterranean export routes. The upcoming growth period from 2026 to 2033 creates fresh entry points for CDMO providers who want to establish decentralized manufacturing capabilities.
Who are the Key Players in the France Healthcare Contract Development And Manufacturing Organization Market and How Do They Compete?
The France CDMO market maintains a moderate level of consolidation because global companies control the biologics manufacturing sector while European mid-sized companies pursue market share through specialized services and their development of facilities in particular areas. The primary factors that drive competition in biologics production include technological capabilities and the ability to meet regulatory requirements and the time needed to bring products to market. The existing market players develop their high-value production capabilities while new market players develop their businesses in specialized fields that include cell therapy treatments and sterile injectable products.
Lonza maintains its market position by operating advanced biologics and cell therapy production systems which handle complex production needs in GMP-certified facilities that enable late-stage clinical trials. Catalent establishes itself as a unique company through its adaptable drug product development services which assist biotech companies in developing their products more quickly. Thermo Fisher Scientific grows by providing complete manufacturing solutions which integrate product development and analytical testing with commercial production processes into one unified system.
Samsung Biologics uses its extensive biomanufacturing capabilities to compete in the market, operating one of the largest biologics production facilities in the world which draws pharmaceutical companies looking to outsource their production needs. Recipharm specializes in the production of sterile injectable products and small-molecule drugs throughout Europe, and the company enhances its market position by expanding its production facilities while maintaining strict regulatory standards. Fareva gains a market edge through its diverse contract manufacturing services for both pharmaceutical products and cosmetic items, which the company operates from its robust European manufacturing facilities. The companies grow their businesses by investing in new production facilities and upgrading existing ones while establishing long-term partnerships with biotech companies to provide manufacturing solutions that can expand their operations.
Company List
- Lonza
- Catalent
- Thermo Fisher
- Recipharm
- WuXi AppTec
- Samsung Biologics
- Boehringer Ingelheim
- AGC Biologics
- Evonik
- Fareva
- Piramal Pharma
- Siegfried
- Jubilant Pharmova
- AbbVie
- Pfizer CentreOne
Recent Development News
In March 2026, Lonza expanded its European biologics manufacturing capacity through additional GMP-certified production lines. The expansion strengthens its ability to support late-stage clinical and commercial biologics demand across Europe.https://www.lonza.com/news (Lonza)
In May 2026, Lonza reported continued expansion of its integrated biologics operations supported by new growth projects and upgraded manufacturing workflows across its global CDMO network. The development improves production scalability for mammalian and biotherapeutic drug programs across Europe.https://www.lonza.com
What Strategic Insights Define the Future of the France Healthcare Contract Development And Manufacturing Organization Market?
The France CDMO market is structurally moving toward high-complexity biologics and decentralized manufacturing networks which biotech pipeline expansion and EU GMP regulatory frameworks drive forward. Over the next 5–7 years, demand will shift toward flexible, modular production systems which can support personalized medicine and small-batch biologics manufacturing needs.
Biotech startups and pharmaceutical clients face dependency risks because advanced biologics capacity exists in limited supply among global CDMO providers. The system will experience major operational delays whenever capacity bottlenecks or compliance disruptions occur within clinical pipelines and commercial launches.
AI-enabled bioprocess optimization together with modular cell therapy manufacturing hubs create a significant opportunity for development in French biotech clusters. CDMOs which make early investments in flexible biologics infrastructure together with digital manufacturing systems will gain long-term contract benefits which will emerge from developing biotech companies. The organization should concentrate its strategic efforts on developing scalable GMP platforms while expanding its operations to multiple regions which will help the business maintain continuous manufacturing capabilities and establish enduring pharmaceutical collaborations.
France Healthcare Contract Development And Manufacturing Organization Market Report Segmentation
By Type
- API Manufacturing
- Biologics Manufacturing
- Packaging Services
- Others
By Application
- Drug Development
- Clinical Trials
- Commercial Production
- Biologics
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Others
By Scale
- Preclinical
- Clinical
- Commercial
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure is USD 23159 Million.
Key segments for the France Healthcare Contract Development And Manufacturing Organization Market are By Type (API Manufacturing, Biologics Manufacturing, Packaging Services, Others); By Application (Drug Development, Clinical Trials, Commercial Production, Biologics, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others); By Scale (Preclinical, Clinical, Commercial, Others).
Major France Healthcare Contract Development And Manufacturing Organization Market players are Lonza, Catalent, Thermo Fisher, Recipharm, WuXi AppTec, Samsung Biologics, Boehringer Ingelheim, AGC Biologics, Evonik, Fareva, Piramal Pharma, Siegfried, Jubilant Pharmova, AbbVie, Pfizer CentreOne.
The France Healthcare Contract Development And Manufacturing Organization Market size is USD 10735 Million in 2025.
The France Healthcare Contract Development And Manufacturing Organization Market CAGR is 10.10% from 2026 to 2033.
- Lonza
- Catalent
- Thermo Fisher
- Recipharm
- WuXi AppTec
- Samsung Biologics
- Boehringer Ingelheim
- AGC Biologics
- Evonik
- Fareva
- Piramal Pharma
- Siegfried
- Jubilant Pharmova
- AbbVie
- Pfizer CentreOne
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)