France Biopharmaceutical Market Size & Forecast:
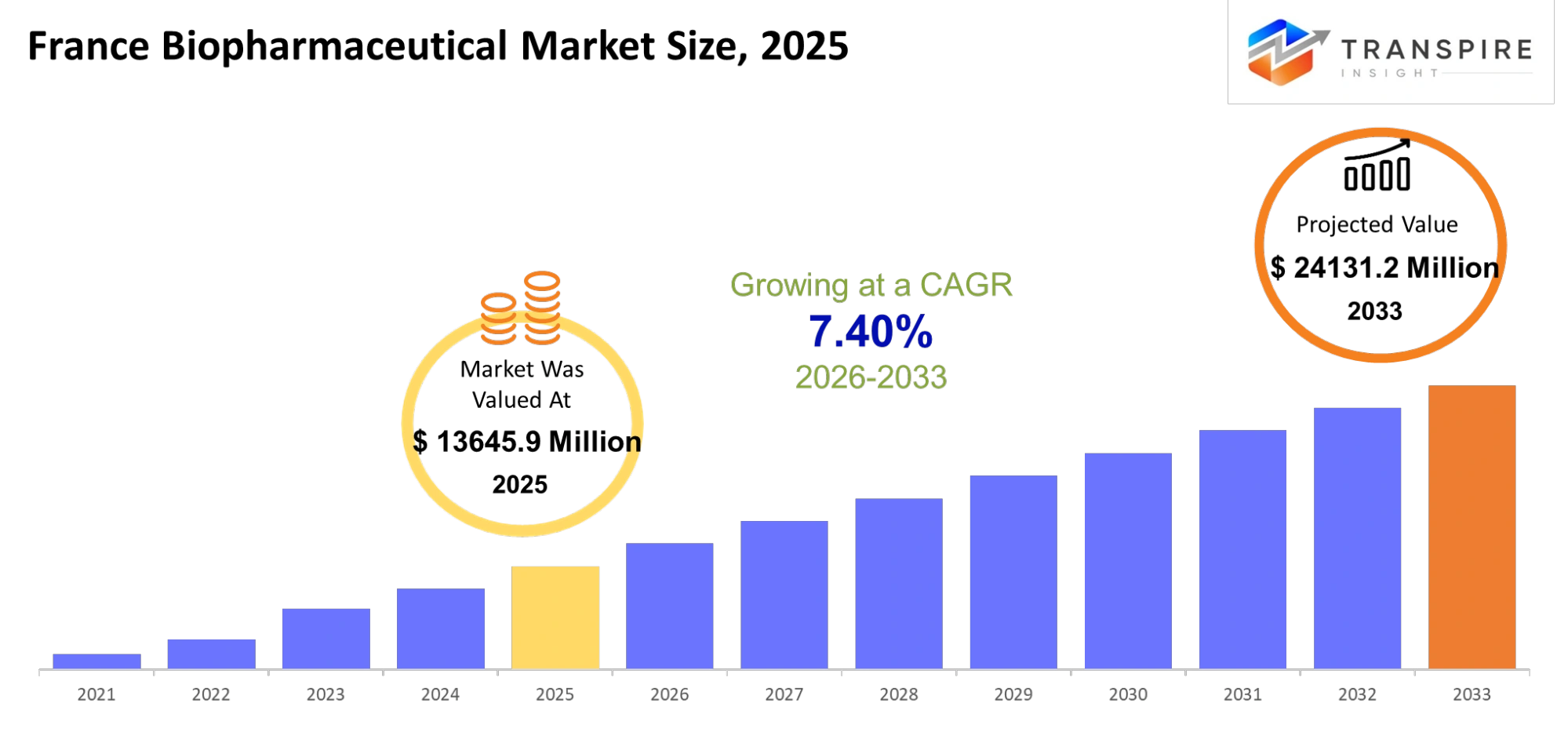
- France Biopharmaceutical Market Size 2025:USD 13645.9 Million
- France Biopharmaceutical Market Size 2033: USD 24131.2 Million
- France Biopharmaceutical Market CAGR: 7.40%
- France Biopharmaceutical Market Segments: By Type (Monoclonal Antibodies, Vaccines, Recombinant Proteins, Gene Therapy, Others); By Application (Oncology, Infectious Diseases, Autoimmune Disorders, Rare Diseases, Others); By End-User (Hospitals, Clinics, Pharma Companies, Research Institutes, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Others)
To learn more about this report, Download Free Sample Report
France Biopharmaceutical Market Summary
The France Biopharmaceutical Market was valued at USD 13645.9 Million in 2025. It is forecast to reach USD 24131.2 Million by 2033. That is a CAGR of 7.40% over the period.
The France biopharmaceutical market serves as a basis for developing treatments which medical practitioners use to treat complex diseases that occur in their patients who have oncological and autoimmune and rare genetic disorders. The system enables doctors to use targeted therapies and biologics and advanced treatments which conventional drugs do not provide so they can achieve better patient outcomes while hospitals maintain their operational efficiency and manage chronic illnesses.
The past three to five years have shown a clear change in drug development which now focuses on biologics and cell and gene therapies because precision medicine systems have advanced and research and development activities have combined better with clinical data systems. The COVID-19 pandemic served as the primary catalyst because it established mRNA platform validation processes which led to the development of faster regulatory pathways and manufacturing operations for these technologies.
This transformation resulted in a new focus on investment priorities. Companies now allocate more capital toward advanced therapy pipelines and localized production capacity. The company achieves growth through its development of high-value treatments which gain faster regulatory approvals through its partnerships with research institutions and healthcare providers which help it create more revenue opportunities while delivering medical services to patients at earlier stages.
Key Market Insights
- The biopharmaceutical market of France will reach its highest point in 2025 when Île-de-France maintains over 40percent market share because it contains both strong research institutes and multiple biotech companies.
- The region of Auvergne-Rhône-Alpes will experience its highest development rate between 2023 and 2033 because of its growing production facilities and effective governmental research and development initiatives.
- Monoclonal antibodies will achieve a market share of 35percent in 2025 because of their extensive application in cancer and autoimmune disorders treatment which is driven by increasing demand for targeted treatment options.
- The second largest market segment after vaccines benefits from public health initiatives together with infrastructure development that happened during the pandemic.
- The cell and gene therapy market segment represents the fastest growth area which will experience major growth until 2033 because of improvements in precision medicine.
- The oncology field will lead the market in 2025 with almost 45 percent market share because targeted biologics are changing cancer treatment methods throughout France.
- The fastest-growing application segment in the market develops rare diseases because orphan drug incentives and genomic research expansion continue to drive its growth.
- The France Biopharmaceutical Market shows hospitals as its leading segment because they account for more than 50 percent of market share through their centralized treatment systems and their ability to administer advanced therapies.
- The End User Segment of specialty clinics shows the highest growth rate because decentralized care systems and personalized treatment methods support their development.
- The research partnerships with strategic institutes enable organizations to speed up their clinical trial processes while reducing their drug development duration.
- Companies establish local manufacturing facilities to protect their supply chains from disruptions while they fulfill regulatory requirements.
- The advanced technologies of AI-based drug discovery and actual data analysis tools help organizations compete better and increase their market share.
What are the Key Drivers, Restraints, and Opportunities in the France Biopharmaceutical Market?
The France biopharmaceutical market experiences its most significant growth because precision medicine is becoming more widespread after genomic research and data-driven clinical studies established its effectiveness. The government-supported innovation initiatives together with their expedited advanced therapy regulatory processes have reduced the time required to commercialize biologics since the past five years. The new treatment method has increased adoption rates because targeted therapies produce results which benefits both oncology and rare disease treatment. The pharmaceutical industry now focuses on high-value biologics because these products offer superior financial returns compared to conventional pharmaceuticals.
The production processes of biopharmaceuticals create dual challenges because they require expensive resources and complex technical execution methods.
Biologic products need specialized production sites and rigorous quality assurance procedures and highly trained workers, which creates difficulties for companies entering the market. The solution to these limitations requires a lengthy process because it depends on both capital investment cycles and the time needed for regulatory validation. The manufacturing limitations result in decreased production capacity, which makes treatments more expensive. Healthcare systems experience reimbursement difficulties, which result in treatment delays for patients and reduce the total market access.
French cell and gene therapy production facilities present a critical opportunity for growth. The combination of localized production hubs with research institution partnerships and biotech firms establishes a framework that can be expanded. The mRNA and viral vector platforms development project produces reusable technology components that can be applied to various therapies. This development method accelerates product creation while establishing France as the primary center for next-generation product development, which will generate additional income sources and international sales opportunities.
What Has the Impact of Artificial Intelligence Been on the France Biopharmaceutical Market?
Artificial intelligence is transforming the French biopharmaceutical industry because it improves the efficiency of research activities and manufacturing processes and regulatory procedures. Artificial intelligence platforms in drug discovery use automated systems to find targets and screen molecules which results in shorter development times during the initial research phase. The companies use machine learning models to study genomic information and clinical trial results which helps them find suitable drug candidates more quickly while creating better trial designs. Advanced analytics in manufacturing processes enable organizations to optimize bioprocess parameters through advanced analytics which results in improved production efficiency and consistent batch quality while reducing operational waste.
Organizations are implementing predictive capabilities as their operational core function. The organizations use artificial intelligence models to predict when equipment will fail in bioreactors and downstream processing units which helps them decrease unexpected operational interruptions while enhancing their facility efficiency. Predictive analytics assist manufacturers in estimating both demand and supply chain requirements which enables them to synchronize their manufacturing processes with the requirements of hospitals and pharmacies. The applications deliver measurable benefits through better cost management and decreased time required for product launches and increased success rates during clinical research.
Artificial intelligence implementation at present encounters a major obstacle because of difficulties in data integration. The biopharmaceutical industry faces challenges with biopharmaceutical datasets because these datasets become divided between old systems and they must follow strict regulations. The process of building unified high-quality datasets necessary for accurate model training becomes challenging because of this issue which leads to delays in implementing large-scale artificial intelligence solutions.
Key Market Trends
- French regulators established shorter approval times to advance therapy approval processes from 2020, which expedited biopharmaceutical product launches and provided multiple oncology and rare disease patients with treatment access.
- Biologics have replaced small-molecule drugs in essential treatment fields because hospitals have increased their usage of monoclonal antibodies between 2021 and 2025.
- Post-pandemic companies changed their manufacturing operations to establish domestic production facilities which enabled them to decrease their dependency on international supply networks while enhancing their capacity to handle unexpected events.
- The period from 2021 until 2024 saw mRNA platforms extend their application beyond vaccine development to create new therapeutic pipelines which transformed research and development funding decisions made by leading pharmaceutical companies.
- Since 2022, biotechnology startups and academic institutions have formed more partnerships which led to faster clinical trials and better research results during the initial phases of drug development.
- Companies implemented AI-powered drug discovery systems after 2023, which led to shorter preclinical testing periods and better results in selecting drug candidates.
- After 2022, pricing and reimbursement systems became more strict which required manufacturers to present better real-world evidence along with cost-effectiveness proof for their premium biologic products.
- Since 2021, clinical oncology treatment planning has adopted personalized medicine through genomic testing which has increased into clinical practice.
- Contract development and manufacturing organizations saw their responsibilities expand after 2020 when they started to provide production and regulatory support to small biotech firms.
- After 2023, sustainability considerations became more important as companies started to invest in environmentally friendly bioprocessing technologies while decreasing their energy-intensive manufacturing processes.
France Biopharmaceutical Market Segmentation
By Type
The revenue from monoclonal antibodies exceeds all other treatment types because oncology and autoimmune therapies depend on targeted biologics that demonstrate validated clinical efficacy. The combination of high precision and extensive reimbursement coverage drives hospitals to implement this solution. The healthcare industry will achieve growth through biosimilars and next-generation antibody technologies which will lower treatment costs for medical facilities.
The vaccines market ranks second because of strong national immunization programs and post-pandemic infrastructure enhancements. The development of rapid-response platforms and public health funding have created stable demand for extended periods. The future of growth will rely on mRNA and multivalent vaccine platforms which provide quick solutions to new diseases and regular seasonal outbreaks.
To learn more about this report, Download Free Sample Report
By Application
The oncology field holds the largest application share because targeted biologics and immunotherapies have created new standards for cancer treatment. The business generates stable profits because high disease prevalence combined with ongoing product development efforts. The market segment will maintain its top position because of active work on combination therapies and biomarker-based treatment solutions.
The second most important medical application exists in infectious diseases because of high demand for vaccines and antiviral biologics. Pandemic preparedness policies have reinforced investment in rapid-response platforms. Future growth will depend on two factors which are surveillance systems and the capacity to expand production during outbreaks.
By End-User
Hospitals spend their resources on advanced biologics and gene therapies which need both controlled environments and expert supervision to function. Centralized treatment delivery supports safety and compliance. The ongoing hospital infrastructure investments will maintain the hospital network's position throughout the upcoming forecast period.
Clinics represent the second-largest segment as outpatient care models expand across chronic disease management. Decentralization of treatment improves patient access and reduces system burden. The industry will expand as additional treatments receive approval for use in facilities outside of hospitals.
By Distribution
Hospital pharmacies maintain distribution control because they need to store and handle and administer biologics during clinical procedures with controlled storage requirements. Treatment facilities need to establish integration systems because they want to ensure patient safety and treatment compliance. Advanced therapies will maintain this distribution channel as institutional delivery systems become their primary method of treatment.
Retail pharmacies hold the second-largest share as certain biologics and chronic therapies transition to outpatient settings. Patients experience better convenience because insurance coverage has expanded which enables more people to receive medical treatment. Future growth will depend on cold chain improvements and pharmacist training for complex therapies.
What are the Key Use Cases Driving the France Biopharmaceutical Market?
The main application of the France biopharmaceutical industry focuses on cancer treatment, which uses targeted biologics and immunotherapies to achieve better patient outcomes with fewer adverse reactions than standard treatment methods. The need for hospitals to administer complex cancer treatments through specialized staff and controlled administration systems drives this requirement. The industry maintains strong clinical usage and high treatment costs because antibody-based treatment methods and their combination therapies continue to see research progress.
Biologics now show potential for use in long-term care to treat both autoimmune diseases and infectious diseases. Specialty clinics increasingly administer these treatments, improving patient access outside large hospitals. Public health agencies and centralized procurement systems support vaccine deployment, which operates through national immunization programs as a vital use case.
The development of new use cases is occurring in gene therapy together with rare disease treatment because these therapies provide permanent solutions that eliminate the need for ongoing medication. Research institutes and biotech firms are advancing these therapies through clinical trials. The growing field of genomic diagnostics enables faster detection of suitable patients, which results in new adoption opportunities.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 13645.9 Million |
|
Market size value in 2026 |
USD 14641.8 Million |
|
Revenue forecast in 2033 |
USD 24131.2 Million |
|
Growth rate |
CAGR of 7.40%from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Roche, Pfizer, Novartis, Sanofi, AbbVie, Amgen, Bristol Myers Squibb, AstraZeneca, Merck, GSK, Takeda, Eli Lilly, Johnson & Johnson, Bayer, Biogen |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Monoclonal Antibodies, Vaccines, Recombinant Proteins, Gene Therapy, Others); By Application (Oncology, Infectious Diseases, Autoimmune Disorders, Rare Diseases, Others); By End-User (Hospitals, Clinics, Pharma Companies, Research Institutes, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Others) |
Which Regions are Driving the France Biopharmaceutical Market Growth?
The France biopharmaceutical market depends on Île-de-France because the region possesses numerous research facilities and international pharmaceutical company offices and receives backing from national health authorities. The Paris region functions as the core for this ecosystem because it possesses advanced clinical trial capabilities and provides access to leading academic experts. The presence of regulatory authorities and funding organizations in the same area enables organizations to obtain approvals faster which results in speedier innovation processes. The integrated system enables organizations to achieve research superiority because it helps them quickly develop their research into marketable medical solutions.
The biomanufacturing and industrial-scale production base make Auvergne-Rhône-Alpes a reliable second region which maintains its position as the second strongest biomanufacturing region. The city of Lyon maintains ongoing operations through its established supply networks and its highly skilled technical personnel. The area directs its activities toward establishing dependable distribution networks which operate at different levels of production instead of investigating new scientific developments. This balance makes it a dependable contributor to revenue, especially for large-scale biologics manufacturing.
The Occitanie region has become the most rapidly growing area because biotech startups receive more investments and research facilities expand in Toulouse and Montpellier. The recent public funding programs and academic partnerships have improved the innovation capacity of the organization through their implementation. The organization has expanded its operations since 2022 by opening new facilities and incubators which help develop new drugs. The current momentum establishes paths for investors who want to enter the rapidly expanding markets of gene therapy and advanced biologics.
Who are the Key Players in the France Biopharmaceutical Market and How Do They Compete?
The France biopharmaceutical market shows a semi-consolidated structure where global pharmaceutical companies control high-value market segments and smaller biotech companies drive early-stage development. The competition between companies depends on their technological superiority and their ability to conduct clinical trials instead of their pricing methods because biologics and advanced therapies maintain high market prices. Existing companies maintain their market position through extensive product development and regulatory knowledge yet new companies are transforming innovation processes through their specialized platforms which include gene therapy and mRNA technologies. The research institutes and large corporations now engage in strategic partnerships which have become essential for gaining competitive advantages.
Sanofi establishes its unique position through its domestic production capabilities and its vaccination expertise which the company supports through its ongoing investments in mRNA technology and its biotech company alliances to develop new products. Roche competes on precision medicine through its diagnostic and therapeutic solutions which create specialized oncology treatments that enhance patient results. Novartis establishes its market leadership in high-value treatments through its advanced therapy development which focuses on gene therapy.
Pfizer expands through strategic collaborations and platform-based innovation, especially after scaling mRNA capabilities during the pandemic. AstraZeneca develops its oncology and biologics departments through data-driven clinical trials and international partnerships which help it achieve faster drug approval results. The companies achieve competitive advantages through their ability to combine research and development resources with regulatory expertise and strategic funding for next-generation therapies which enables them to enter markets more quickly while generating continuous revenue growth.
Company List
- Roche
- Pfizer
- Novartis
- Sanofi
- AbbVie
- Amgen
- Bristol Myers Squibb
- AstraZeneca
- Merck
- GSK
- Takeda
- Eli Lilly
- Johnson & Johnson
- Bayer
- Biogen
Recent Development News
In April 2026, Sanofi reported late-stage progress in immunology and respiratory biologics, including positive trial outcomes and regulatory milestones. The updates strengthen its leadership in rare diseases and next-generation antibody therapies.
Source:https://www.sanofi.com
In July 2025, Sanofi acquired UK biotech Vicebio for up to $1.15 billion to expand its respiratory vaccine pipeline and combination vaccine technology. The deal strengthens its RSV and respiratory disease portfolio.
Source:https://www.reuters.com
What Strategic Insights Define the Future of the France Biopharmaceutical Market?
The France biopharmaceutical market is heading toward multiple years of upcoming development through genomics and regulatory acceleration and increasing demand for curative therapies which need complex precision medical treatments. The industry is transitioning from pharmaceutical manufacturing that relies on producing large quantities to producing high-value biological products and advanced treatment systems which create profits through fewer but more valuable medical solutions.
The supply chain dependency on specialized materials which include viral vectors and bioreactor-grade components carries hidden dangers because there are only a few worldwide suppliers who handle these materials. The advancement of gene and cell therapies faces a particular restriction which will result in postponed commercialization despite successful clinical development.
Decentralized biomanufacturing systems which use modular production modules and AI-driven process management systems have become an emerging opportunity that is especially developing in French regional biotech hubs. The approach will enable manufacturers to establish their own production facilities while achieving faster manufacturing processes. Market participants should prioritize investments in scalable manufacturing technologies and strategic partnerships with CDMO networks to secure long-term capacity and reduce operational risk exposure.
France Biopharmaceutical Market Report Segmentation
By Type
- Monoclonal Antibodies
- Vaccines
- Recombinant Proteins
- Gene Therapy
- Others
By Application
- Oncology
- Infectious Diseases
- Autoimmune Disorders
- Rare Diseases
- Others
By End-User
- Hospitals
- Clinics
- Pharma Companies
- Research Institutes
- Others
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure is USD 24131.2 Million.
Key segments for the France Biopharmaceutical Market are By Type (Monoclonal Antibodies, Vaccines, Recombinant Proteins, Gene Therapy, Others); By Application (Oncology, Infectious Diseases, Autoimmune Disorders, Rare Diseases, Others); By End-User (Hospitals, Clinics, Pharma Companies, Research Institutes, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Others).
Major France Biopharmaceutical Market players are Roche, Pfizer, Novartis, Sanofi, AbbVie, Amgen, Bristol Myers Squibb, AstraZeneca, Merck, GSK, Takeda, Eli Lilly, Johnson & Johnson, Bayer, Biogen.
The France Biopharmaceutical Market size is USD 13645.9 Million in 2025.
The France Biopharmaceutical Market CAGR is 7.40% from 2026 to 2033.
- Roche
- Pfizer
- Novartis
- Sanofi
- AbbVie
- Amgen
- Bristol Myers Squibb
- AstraZeneca
- Merck
- GSK
- Takeda
- Eli Lilly
- Johnson & Johnson
- Bayer
- Biogen
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)