Europe Regenerative Medicine Market Size & Forecast:
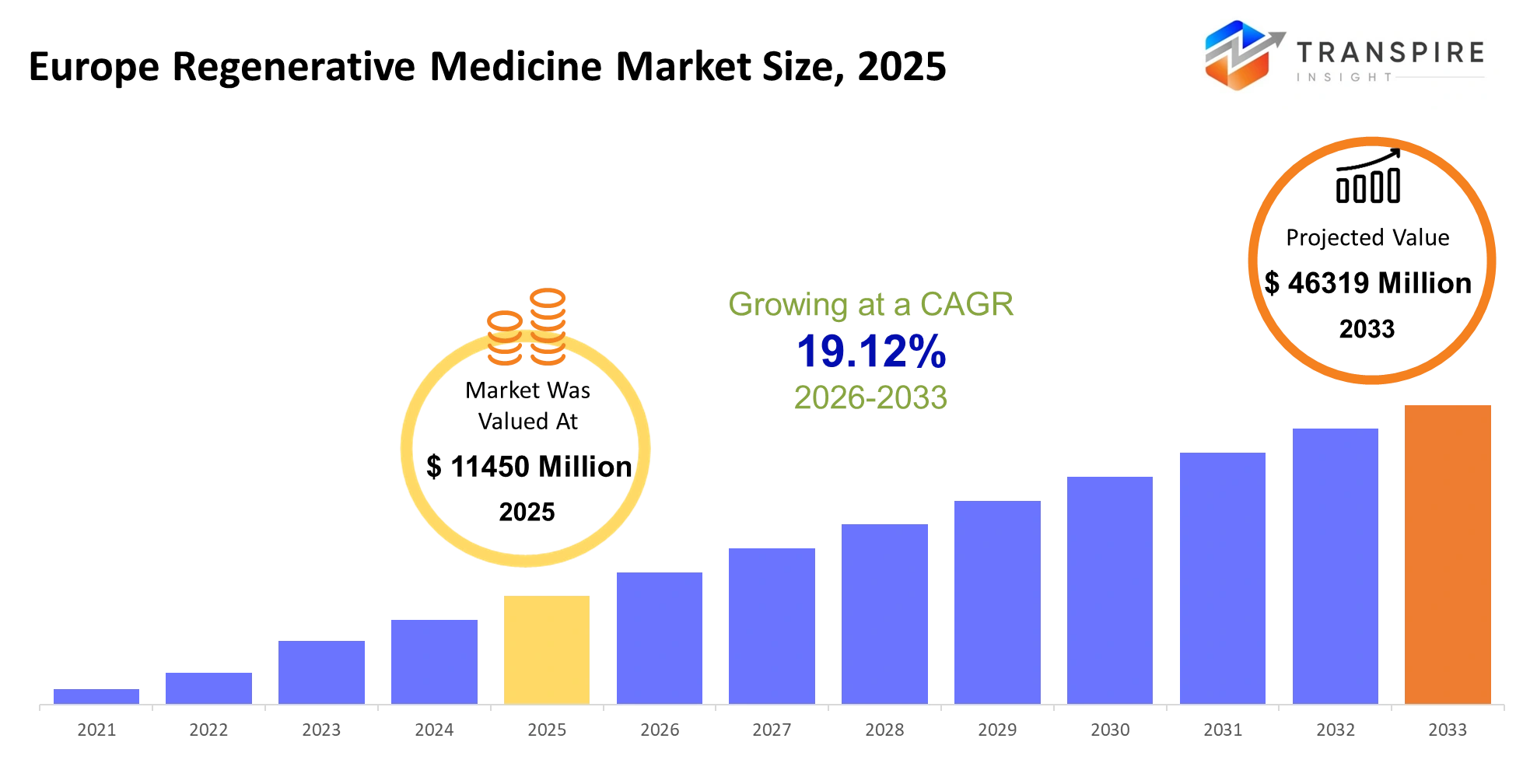
- Europe Regenerative Medicine Market Size 2025: USD 11450 Million
- Europe Regenerative Medicine Market Size 2033: USD 46319 Million
- Europe Regenerative Medicine Market CAGR: 19.12%
- Europe Regenerative Medicine Market Segments: By Type (Cell Therapy, Gene Therapy, Tissue Engineering, Stem Cell Therapy, Biomaterials, Others); By Application (Orthopedics, Oncology, Dermatology, Cardiology, Neurology, Others); By End-User (Hospitals, Clinics, Research Institutes, Biotech, Pharma, Others); By Product (Therapies, Devices, Consumables, Scaffolds, Biologics, Others).
To learn more about this report, Download Free Sample Report
Europe Regenerative Medicine Market Summary:
The Europe Regenerative Medicine Market size is estimated at USD 11450 Million in 2025 and is anticipated to reach USD 46319 Million by 2033, growing at a CAGR of 19.12% from 2026 to 2033. The europe regenerative medicine market in the advanced healthcare space will undoubtedly transform the approaches and methods for therapy development and delivery in the region. The patients will become more demanding and seek more personalized and non-invasive treatment approaches, which will encourage the use of cell therapies and bioengineered tissues by the hospitals and biotechnology companies. The regulatory agencies will strengthen their scrutiny on one hand and hasten the process of drug approval on the other hand. Advancements in the realms of stem cells, genetic engineering, and bioprinting will revolutionize the way things will be done in the field.
What Has the Impact of Artificial Intelligence Been on the Europe Regenerative Medicine Market?
Artificial intelligence is rapidly reshaping the europe regenerative medicine market by introducing data-driven precision into research, development, and commercialization. AI in europe regenerative medicine market environments is enabling deeper insights from complex biological datasets, allowing companies to identify viable cell therapies and regenerative pathways faster than traditional methods. Advanced machine learning models are streamlining market research, uncovering hidden patterns in clinical outcomes, and supporting predictive analytics that refine demand forecasting and investment decisions.
Artificial intelligence in europe regenerative medicine market operations is also improving production efficiency through smart automation. From optimizing bioprocessing conditions to reducing human error in laboratory workflows, AI-driven systems are helping manufacturers scale regenerative solutions with greater consistency and lower operational costs. At the same time, supply chain optimization is becoming more agile, as predictive tools anticipate material shortages and align distribution with real-time demand signals across Europe.
Beyond operational gains, AI is strengthening innovation and personalization within the europe regenerative medicine market. It is enabling tailored therapies based on patient-specific genetic and clinical data, creating a competitive edge for companies that adopt intelligent systems early. As market trends continue to favor precision medicine, AI will remain central to accelerating breakthroughs, enhancing decision-making, and driving sustainable growth across the sector.
Key Market Trends & Insights:
- Germany dominates the europe regenerative medicine market with ~28% share in 2024, driven by strong biotech infrastructure and clinical research funding.
- The UK is the fastest-growing region, expected to expand at ~17% CAGR through 2030 due to favorable regulatory frameworks and R&D incentives.
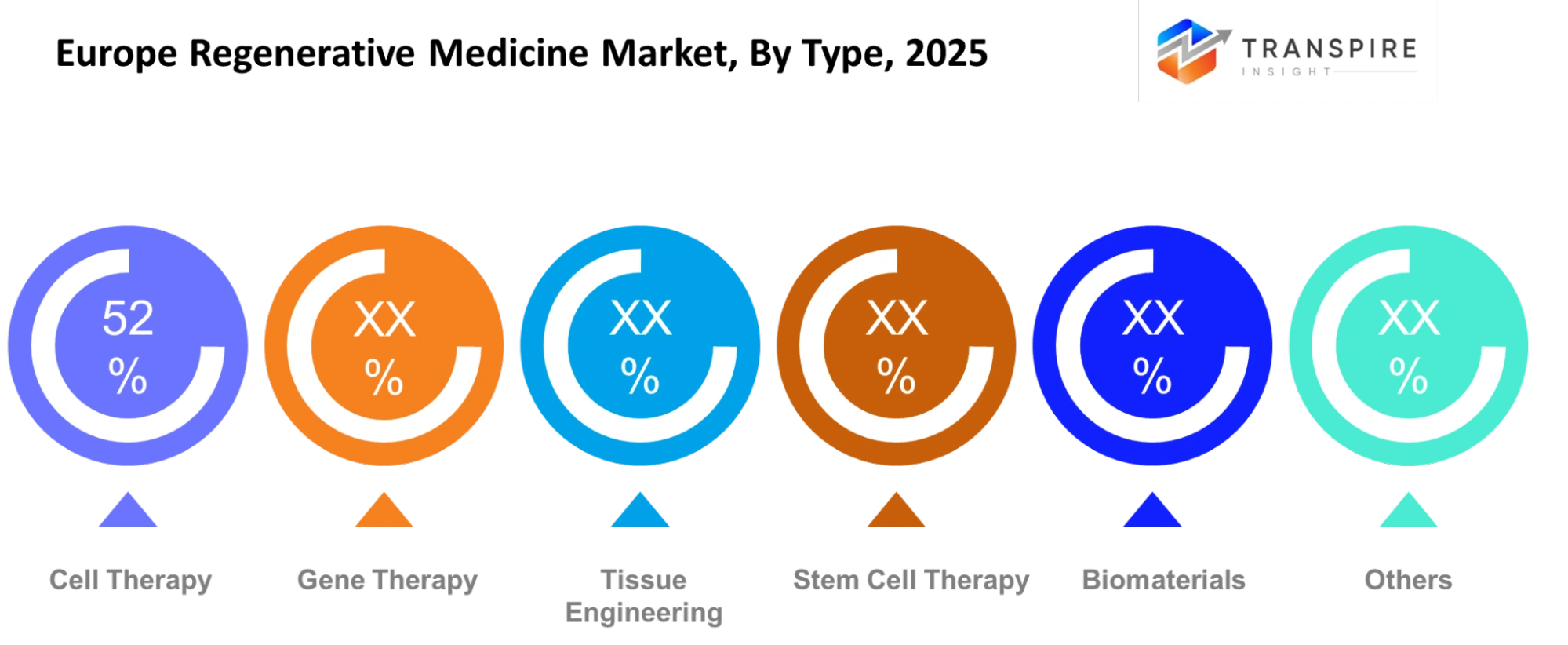
- Cell therapy leads with over 45% market share in 2024, supported by increasing clinical approvals and adoption in oncology treatments.
- Gene therapy holds the second-largest share (~30%), benefiting from advancements in CRISPR and viral vector technologies.
- Tissue engineering is the fastest-growing segment, projected to grow at ~18% CAGR through 2030 due to rising demand for organ repair solutions.
- Oncology dominates with ~40% share, driven by increasing use of regenerative therapies in cancer treatment and immune system enhancement.
- Orthopedics is the fastest-growing application, fueled by rising cases of musculoskeletal disorders and demand for minimally invasive solutions.
- Hospitals account for ~50% of the europe regenerative medicine market share, supported by advanced infrastructure and high patient inflow.
- Research institutes are rapidly growing, expected to expand at ~16% CAGR due to increased funding and innovation partnerships.
Europe Regenerative Medicine Market Segmentation
By Type
Cell therapy will focus on using living cells to repair or replace damaged tissues, especially in conditions where traditional treatments show limited success. Gene therapy will aim to correct or modify genetic material to treat inherited and chronic diseases. Tissue engineering will combine cells and biomaterials to create functional tissue structures for medical use. Stem cell therapy will continue to gain attention due to the ability to develop into multiple cell types for regenerative purposes.
Biomaterials will support these treatments by providing structural support and improving compatibility within the human body. Research efforts will improve material quality to ensure better integration and durability. Each type will play a role in building a more advanced treatment ecosystem. Growth in this segment will depend on clinical validation and scalability of these technologies.
To learn more about this report, Download Free Sample Report
By Application
Orthopedics will use regenerative solutions for bone and joint repair, helping reduce the need for invasive surgeries. Oncology will apply regenerative approaches to support immune-based treatments and tissue recovery after intensive therapies. Dermatology will focus on skin repair and wound healing, especially in burn care and cosmetic procedures. Cardiology will explore tissue repair for heart-related conditions, aiming to improve long-term patient survival.
Neurology will present new possibilities in treating nerve damage and degenerative conditions through regenerative techniques. Each application will grow based on disease prevalence and clinical success rates. Healthcare systems will adopt these solutions as evidence of effectiveness increases. Demand will remain strong in areas where traditional treatments offer limited recovery outcomes.
By End-User
Hospitals will remain key users due to access to advanced infrastructure and patient volume. Clinics will gradually adopt regenerative treatments as procedures become more standardized and accessible. Research institutes will play a central role in developing and testing new therapies, supported by funding and collaborations. Biotech firms will drive innovation by focusing on specialized treatment development.
Pharma companies will expand involvement through partnerships and investment in regenerative technologies. Each end-user group will contribute to the growth of treatment availability and accessibility. Collaboration between these entities will improve development timelines and clinical outcomes. Expansion in this segment will depend on infrastructure readiness and skilled workforce availability.
By Product
Therapies will form the core offering, focusing on direct patient treatment through advanced regenerative methods. Devices will support delivery and monitoring of these treatments, improving precision and safety. Consumables will include essential materials used during procedures, ensuring consistency in clinical practice. Scaffolds will provide structural frameworks that support tissue growth and regeneration.
Biologics will include products derived from living organisms, playing a major role in treatment effectiveness. Product development will focus on improving quality, efficiency, and patient outcomes. Innovation will continue to shape how these products are designed and used in clinical settings. Market progress will depend on regulatory approval and the ability to scale production efficiently.
What are the Main Challenges for the Europe Regenerative Medicine Market Growth?
The Europe regenerative medicine market faces several technical and operational challenges that limit consistent progress. Product stability remains a major concern, as cell-based therapies require strict handling conditions and controlled environments. Variability in biological materials often leads to inconsistent performance, affecting treatment reliability. In addition, complex supply chains for sensitive materials create delays and increase operational risks, highlighting key challenges in europe regenerative medicine market expansion.
Manufacturing and commercialization barriers continue to slow down large-scale adoption in the europe regenerative medicine market. Production processes are highly specialized and must comply with strict Good Manufacturing Practice standards, increasing time and cost pressures. Many companies struggle with scaling up from clinical trials to commercial production without compromising quality. These market limitations often delay product approvals and reduce the speed at which innovative therapies reach patients.
Adoption challenges also impact the europe regenerative medicine market, particularly due to limited infrastructure and a shortage of skilled professionals. Advanced facilities are required to deliver regenerative treatments, yet many regions lack the necessary setup. Training gaps in handling complex therapies further restrict widespread use. Funding constraints, especially in smaller economies, reduce access to cutting-edge treatments and slow overall market penetration.
Competitive pressure and future risks add another layer of complexity to the europe regenerative medicine market. Alternative treatment technologies, including conventional biologics, continue to compete on cost and accessibility. Pricing pressure remains high as healthcare systems demand cost-effective solutions. At the same time, evolving regulatory frameworks and rapid technological changes create uncertainty, reinforcing long-term growth barriers for the market.
Regional Insights
The Europe regenerative medicine market will continue to expand as healthcare systems focus on advanced treatment approaches that restore damaged tissues and improve long-term recovery outcomes. Rising demand for personalized care and minimally invasive procedures will influence development strategies. Strong research support and clinical advancements will shape future progress across major European economies.
Investment in biotechnology and collaboration between research institutes and healthcare providers will strengthen innovation pipelines. Regulatory bodies will maintain strict approval frameworks while supporting faster access to breakthrough therapies. Growth will remain linked to clinical success rates, scalability of production, and integration of new technologies into mainstream healthcare systems.
Western Europe will dominate due to established healthcare infrastructure, strong funding support, and presence of leading biotech companies. Countries such as Germany, France, and the United Kingdom will drive innovation through research initiatives and early adoption of regenerative therapies. Demand will remain high due to aging populations and rising chronic disease cases.
Eastern Europe will witness gradual growth supported by improving healthcare systems and increasing investment in medical research. Adoption rates will rise as awareness increases and infrastructure develops. Regional expansion will depend on government initiatives, funding availability, and access to skilled professionals, which will influence long-term market penetration.
Recent Development News
In April 2026, UCB announced acquisition of Neurona Therapeutics. The deal, valued at over $1 billion, will expand UCB’s pipeline into regenerative cell therapies for neurological disorders.
Source: https://www.ucb.com
In April 2026, UCB reported acquisition agreement of Neurona Therapeutics for $650 million upfront with additional milestone payments. The acquisition will strengthen regenerative medicine capabilities through development of NRTX-1001 cell therapy for epilepsy treatment.
Source: https://www.fiercebiotech.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 11450 Million |
|
Market size value in 2026 |
USD 13614 Million |
|
Revenue forecast in 2033 |
USD 46319 Million |
|
Growth rate |
CAGR of 19.12% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Novartis, Roche, Pfizer, AstraZeneca, Johnson & Johnson, Smith & Nephew, Medtronic, Zimmer Biomet, AbbVie, Sanofi, Vericel, Organogenesis, Mesoblast, Takeda, Bayer. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Cell Therapy, Gene Therapy, Tissue Engineering, Stem Cell Therapy, Biomaterials, Others); By Application (Orthopedics, Oncology, Dermatology, Cardiology, Neurology, Others); By End-User (Hospitals, Clinics, Research Institutes, Biotech, Pharma, Others); By Product (Therapies, Devices, Consumables, Scaffolds, Biologics, Others). |
How Can New Companies Establish a Strong Foothold in the Europe Regenerative Medicine Market?
New entrants can build a strong position in the europe regenerative medicine market by focusing on clearly defined niches where unmet clinical needs remain high. Instead of competing broadly, companies will benefit from targeting areas such as rare diseases, orthopedic repair, or personalized cell therapies. This approach allows efficient use of limited resources while aligning with current industry trends that favor precision medicine. In the europe regenerative medicine market, niche specialization also improves the chances of faster regulatory approval and early clinical success.
Innovation strategies will play a decisive role in differentiation. Startups that integrate artificial intelligence, predictive analytics, and advanced bioprocessing will improve treatment accuracy and reduce development timelines. For example, companies like Cellino are advancing automated stem cell manufacturing, while Celyad Oncology is focusing on engineered cell therapies for cancer treatment. Such emerging players demonstrate how technology-led innovation can address key pain points in scalability and consistency within the europe regenerative medicine market.
Strategic partnerships will further strengthen market entry. Collaborations with research institutes, hospitals, and established biotech firms will provide access to infrastructure, funding, and clinical expertise. New companies can also solve manufacturing and supply chain challenges by adopting smart automation and modular production systems. In the europe regenerative medicine market, firms that combine focused innovation, strong alliances, and scalable solutions will secure long-term competitive advantage.
Key Europe Regenerative Medicine Market Company Insights
The europe regenerative medicine market will continue to expand as healthcare systems shift toward advanced therapies focused on tissue repair and long-term recovery. Demand will rise due to increasing chronic conditions and aging populations. Strong research ecosystems and clinical trials will support development across major European countries, improving treatment accessibility and effectiveness.
Investment will increase in biotechnology firms working on cell and gene-based solutions. Regulatory frameworks will balance safety with faster approvals for innovative treatments. Growth will depend on scalable production methods and consistent clinical outcomes. Integration of digital tools and data-driven systems will further improve efficiency and patient-specific treatment planning.
Competition will remain strong as established pharmaceutical companies and emerging biotech firms continue to invest in advanced therapies. Companies such as Novartis, Roche, and AstraZeneca will strengthen positions through product innovation and strategic collaborations. Focus on personalized treatments and advanced research will help maintain competitive advantage in key European markets.
Emerging players will challenge established firms by introducing cost-effective and technology-driven solutions. Partnerships with research institutes and healthcare providers will accelerate development and commercialization. Competitive strategies will include regional expansion, improved manufacturing processes, and adoption of automation to enhance efficiency and reduce operational costs.
Company List
- Novartis
- Roche
- Pfizer
- AstraZeneca
- Johnson & Johnson
- Smith & Nephew
- Medtronic
- Zimmer Biomet
- AbbVie
- Sanofi
- Vericel
- Organogenesis
- Mesoblast
- Takeda
- Bayer
What are the Key Use-Cases Driving the Growth of the Europe Regenerative Medicine Market?
The europe regenerative medicine market is gaining traction through strong clinical use-cases that address unmet medical needs in high-burden diseases. One of the most impactful applications is in oncology, where cell-based immunotherapies are improving cancer treatment outcomes and reducing relapse rates. Hospitals across Europe are adopting these therapies to enhance survival rates, which is directly supporting the expansion of the europe regenerative medicine market.
Orthopedic care is another major use-case driving adoption in the europe regenerative medicine market. Regenerative solutions such as stem cell therapies and tissue engineering are being used to repair damaged joints, cartilage, and bones, reducing the need for invasive surgeries. This approach not only shortens recovery time but also lowers long-term healthcare costs, making it attractive for both providers and patients.
Cardiology and neurology are emerging as high-potential areas within the europe regenerative medicine market. Regenerative therapies are being explored for repairing heart tissues after cardiac events and restoring nerve function in degenerative conditions. These applications are still developing but show strong scalability due to rising cases of cardiovascular and neurological disorders across Europe.
Beyond healthcare, research institutions and biotech firms are using regenerative technologies to develop advanced biomaterials and personalized treatments. This innovation-driven use-case is accelerating clinical trials and improving treatment precision. As these applications expand, the europe regenerative medicine market will continue to grow through increased adoption, technological progress, and broader therapeutic impact.
Europe Regenerative Medicine Market Report Segmentation
By Type
- Cell Therapy
- Gene Therapy
- Tissue Engineering
- Stem Cell Therapy
- Biomaterials
- Others
By Application
- Orthopedics
- Oncology
- Dermatology
- Cardiology
- Neurology
- Others
By End-User
- Hospitals
- Clinics
- Research Institutes
- Biotech
- Pharma
- Others
By Product
- Therapies
- Devices
- Consumables
- Scaffolds
- Biologics
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Regenerative Medicine Market size for the market will be USD 46319 Million in 2033.
Key segments for the Europe Regenerative Medicine Market are By Type (Cell Therapy, Gene Therapy, Tissue Engineering, Stem Cell Therapy, Biomaterials, Others); By Application (Orthopedics, Oncology, Dermatology, Cardiology, Neurology, Others); By End-User (Hospitals, Clinics, Research Institutes, Biotech, Pharma, Others); By Product (Therapies, Devices, Consumables, Scaffolds, Biologics, Others).
Major Europe Regenerative Medicine Market players are Novartis, Roche, Pfizer, AstraZeneca, Johnson & Johnson, Smith & Nephew, Medtronic, Zimmer Biomet, AbbVie, Sanofi, Vericel, Organogenesis, Mesoblast, Takeda, Bayer.
The Europe Regenerative Medicine Market size is USD 11450 Million in 2025.
The Europe Regenerative Medicine Market CAGR is 19.12%.
- Novartis
- Roche
- Pfizer
- AstraZeneca
- Johnson & Johnson
- Smith & Nephew
- Medtronic
- Zimmer Biomet
- AbbVie
- Sanofi
- Vericel
- Organogenesis
- Mesoblast
- Takeda
- Bayer
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031


