Europe CRO Mass Spectroscopy Services Market Size & Forecast:
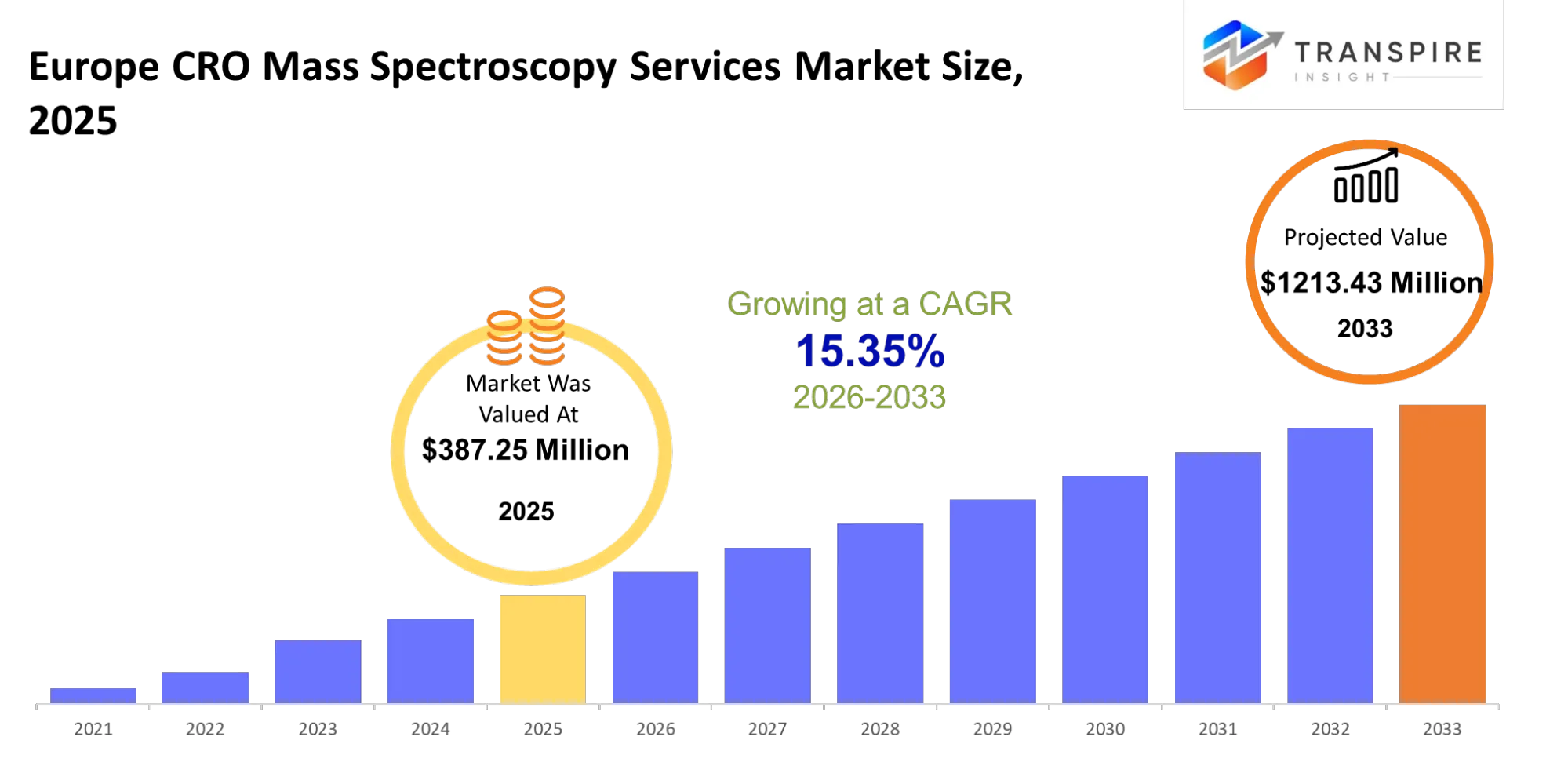
- Europe CRO Mass Spectroscopy Services Market Size 2025: USD 387.25 Million
- Europe CRO Mass Spectroscopy Services Market Size 2033: USD 1213.43 Million
- Europe CRO Mass Spectroscopy Services Market CAGR: 15.35%
- Europe CRO Mass Spectroscopy Services Market Segments: By Type (Small Molecule Analysis, Large Molecule Analysis, Others), By Application (Drug Discovery, Clinical Trials, Proteomics, Others), By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others), By Technology (LC-MS, GC-MS, MALDI-TOF, Others).
To learn more about this report, Download Free Sample Report
Europe CRO Mass Spectroscopy Services Market Summary:
The Europe CRO Mass Spectroscopy Services Market size is estimated at USD 387.25 Million in 2025 and is anticipated to reach USD 1213.43 Million by 2033, growing at a CAGR of 15.35% from 2026 to 2033. The Europe CRO mass spectroscopy services market depends on specialized laboratories which assist pharmaceutical and biotech companies with their chemical and biological compound identification and quantification and validation needs throughout the drug discovery and development and regulatory submission processes. The services provide a solution which enables companies to obtain precise molecular data without the need to invest in costly laboratory equipment for their internal analytical operations.
The market has evolved during the last 3 to 5 years from basic assay outsourcing to advanced high-resolution LC-MS platforms which can perform complex biological analyses of products from cell and gene therapy. The pharmaceutical industry has undergone a structural transformation which transfers analytical testing processes from internal laboratory settings to dedicated CRO testing facilities.
The post-COVID supply chain disruptions together with new EU regulations which control nitrosamine impurities triggered this acceleration because they required companies to perform process revalidation at an urgent pace. The company needs specialized mass spectrometry services because its advanced mass spectrometry needs have become essential for company growth which results in ongoing revenue increases.
Key Market Insights
- The Europe CRO Mass Spectroscopy Services Market shows its strongest presence in Western Europe because Germany, the UK, and France serve as major pharmaceutical centers.
- The Eastern European market experiences rapid growth because CRO companies expand at low costs while clinical research outsourcing becomes more popular.
- Germany possesses the largest market share because its biotech facilities support extensive testing activities.
- The Europe CRO Mass Spectroscopy Services Market uses LC-MS-based analytical services because they provide high sensitivity and meet regulatory standards.
- The second-largest market share belongs to sample preparation and bioanalytical testing services which assist with standard pharmaceutical operations.
- The segment with the fastest growth until 2030 consists of high-resolution mass spectrometry services which scientists require to study biologics and complex molecules.
- The Europe CRO Mass Spectroscopy Services Market shows drug development and validation applications as the main market driver for its services.
- The application area with the fastest growth rate belongs to proteomics and biomarker discovery because of the rising need for precision medicine.
- The European Union has implemented more rigorous safety testing standards which has resulted in increased demand for toxicology testing services.
- The pharmaceutical industry stands as the primary customer group for CRO mass spectrometry services which generates most of the service demand.
- Biotechnology firms are the fastest-growing end-user group due to rapid biologics and cell therapy pipelines.
What are the Key Drivers, Restraints, and Opportunities in the Europe CRO Mass Spectroscopy Services Market?
The **Europe CRO Mass Spectroscopy Services Market** experiences its main growth through the increasing trend of pharmaceutical companies to outsource their analytical processes. The European Union established stricter impurity and bioanalytical validation requirements which led to organizations needing to prove their medical product safety through enhanced regulatory standards. The operational costs and capital expenses of in-house laboratories increased because of rising compliance demands for high-resolution LC-MS equipment. Pharmaceutical and biotech companies now prefer to work with CROs that offer specialized mass spectrometry services which generate additional income through ongoing projects and permanent business agreements throughout Europe.
The field suffers from a major structural limitation because there is an ongoing deficit of analytical scientists who possess advanced skills for using and understanding complex mass spectrometry equipment. The development of expertise creation proves difficult because it requires multiple years of training together with ongoing practice on new equipment. Organizations that provide contract research services need to expand their capabilities to meet client needs because their current capacity does not match demand which results in extended delivery times and limited business growth. The organization faces revenue challenges because it lacks sufficient skilled workers despite having multiple projects in the pipeline.
The integration of AI-enabled data processing into mass spectrometry workflows creates a major opportunity for scientific advancement. Thermo Fisher Scientific and other CROs are funding machine-learning-based spectral analysis systems to enhance identification accuracy while decreasing testing duration. The biotechnology industry in Eastern Europe is developing cost-effective outsourcing centers through its expansion into Poland and Hungary. The European CRO Mass Spectroscopy Services Market will experience its next growth stage through the combination of automation with regional market expansion.
What Has the Impact of Artificial Intelligence Been on the Europe CRO Mass Spectroscopy Services Market?
Artificial intelligence transforms the **Europe CRO Mass Spectroscopy Services Market** because it changes how analytical data is created and handled and understood in pharmaceutical and biotech operations. AI-based automation solutions in current contract research organization laboratories enable efficient operation of LC-MS systems through their peak detection abilities and spectral deconvolution capabilities and robotic sample preparation functions. The systems ensure that testing activities require less human input while maintaining testing accuracy during high-speed testing procedures which are essential for drug development and bioanalysis work.
Predictive capabilities are also gaining traction, with machine learning models applied to instrument health monitoring and predictive maintenance of mass spectrometry systems. The CROs use performance drift and calibration pattern analysis to identify equipment problems which lead to unexpected downtime while they improve their ability to operate instruments. The company achieves faster client service delivery through this method while establishing better capacity planning for analytical work throughout Europe contract research ecosystem.
AI systems bring operational advantages which include decreased need for reanalysis testing and improved efficiency of high-resolution LC-MS processes and reduced testing expenses in extensive research projects. The current structure of spectral data usage in laboratories creates an obstacle which prevents organizations from implementing their AI systems.AI model deployment faces delays because of data inconsistency and high integration expenses and the need for regulatory validation. The Europe CRO Mass Spectroscopy Services Market shows increasing demand for advanced analytical testing systems.
Key Market Trends
- The pharmaceutical industry transferred more than 40 percent of its testing work to contract research organizations which became necessary because European Union regulations became more complex and the industry experienced higher cost requirements.
- European countries saw a major increase in high-resolution mass spectrometry usage which occurred after 2022 because the region developed multiple biologics and gene therapy programs.
- The 2023 implementation of stricter regulations for nitrosamine impurities resulted in companies increasing their validation study outsourcing to dedicated contract research organization labs.
- The demand for services that use proteomics to discover biomarkers experienced substantial growth since 2021 because precision medicine research became more common in oncology studies.
- The capacity of Eastern Europe contract research organizations grew quickly between 2022 and 2026 because their operational expenses decreased and they received European Union research infrastructure funding.
- Eurofins Scientific and SGS expanded their investments in automated LC-MS systems to achieve better processing capacity and faster response times for regulatory requirements.
- The development of AI-driven spectral analysis tools gained popularity after 2024 because they decreased the time needed for manual study assessments and enabled researchers to obtain consistent results during extensive research projects.
- Top contract research organizations achieved a 20 percent reduction in sample reanalysis rates because their advanced laboratories used better instrument calibration and predictive maintenance technologies.
- Biotech companies increased their outsourcing activities after 2023 because their internal mass spectrometry equipment proved too expensive for them to expand their research operations.
Europe CRO Mass Spectroscopy Services Market Segmentation
By Type
The Europe CRO Mass Spectroscopy Services Market is primarily controlled by small molecule analysis which has maintained its status as the main analysis method for conventional drug development and regulatory testing procedures. The field of large molecule analysis is experiencing rapid growth because biologics and monoclonal antibodies and cell-based therapies become more popular in European drug development pipelines. The testing volume for established analytical protocols in both generic and branded pharmaceutical markets allows small molecule services to maintain their leading market position.
The field of large molecule analysis has gained increased traction because regulatory bodies now require organizations to conduct more in-depth structural analysis and impurity examination. This transformation causes CRO service offerings to develop towards hybrid functions. The segment will evolve into integrated platforms that support both molecular types because CROs need to handle different types of therapeutic pipelines while maintaining steady income during changing drug development processes.
By Application
The drug discovery process remains the leading segment because pharmaceutical pipelines need ongoing support for their initial compound discovery and testing processes. The clinical trial segment has grown because European Union regulations now require development phases to undergo complete safety and bioanalytical testing procedures. The field of proteomics has become the fastest expanding application area because precision medicine and biomarker-based diagnostics have become more widely used.
Drug discovery maintains its top position because companies need to outsource their analytical work while their testing processes require consistent testing. Oncological research and the creation of personalized treatments drive the development of proteomics. The segment is developing multi-omics data analysis capabilities to enable CROs to deliver more detailed answers which will help them secure higher-value agreements with pharmaceutical and biotechnology clients.
To learn more about this report, Download Free Sample Report
By End-User
The End-User pharmaceutical companies create demand for Europe CRO Mass Spectroscopy Services because their drug pipelines and testing requirements for regulations form the foundation of their business operations. The end-user group with the fastest growth rate consists of biotech firms which now outsource their analytical workflows to reduce costs while they speed up their process of developing new products. Research institutes sustain consistent output through their academic and government-funded projects which investigate disease biology and molecular research.
The testing requirements of pharmaceutical companies and their established outsourcing systems drive market demand, while laboratory biotech companies create demand through their adaptable project work. Research institutes support early-stage innovation but operate with limited budgets. The segment is gradually shifting towards biotechnology companies which will create service requirements through their particular medical needs that they will meet by working with contract research organizations.
By Technology
The Europe CRO Mass Spectroscopy Services Market recognizes LC-MS as the leading technology because its high sensitivity combined with regulatory acceptance and its ability to support all stages of drug development processes. The field maintains its current status through GC-MS, which serves as the primary tool for analyzing volatile compounds and testing environmental and toxicology samples. The MALDI-TOF system has become increasingly popular because its fast and precise analysis capabilities enable scientists to conduct proteomic and microbial research on complicated biological materials.
The scientific community prefers LC-MS as their primary testing method because it can examine both small and large molecules, while GC-MS remains in use for specific testing areas that develop at a slower rate. CROs currently choose MALDI-TOF systems as their preferred method for conducting high-volume tests on biological samples. The segment is progressing toward multi-platform integration, where CROs combine complementary technologies to improve analytical depth, reduce turnaround time, and strengthen competitiveness in high-value pharmaceutical outsourcing contracts.
What are the Key Use Cases Driving the Europe CRO Mass Spectroscopy Services Market?
The primary function of the European CRO Mass Spectroscopy Services Market exists for drug discovery because pharmaceutical companies need their high-precision LC-MS workflows to meet the demands of EU regulations when they seek to identify, validate, and optimize lead compounds. The testing process reaches its peak volume at this stage because researchers must conduct multiple tests of structural confirmation and impurity profiling for every new molecule before they can advance it to clinical development.
The field of clinical trial bioanalysis and proteomics-based biomarker validation experiences rapid growth currently because biotechnology companies and major pharmaceutical companies are adopting these services. The use cases enable researchers to identify patient groups who require different types of treatment while they monitor treatment safety and assess treatment effectiveness throughout their multi-phase clinical studies. Biotech companies are increasing their outsourcing activities which has created a stronger demand for analytical services that provide rapid results with high sensitivity while reducing the need for companies to build their own testing facilities which will help them meet regulatory requirements.
The emerging applications of cell and gene therapy characterization and multi-omics data integration work together to develop precision medicine solutions. The areas have not yet reached complete market acceptance but their growth is accelerating because oncology research activities have increased and the European Union provides funds for the development of new advanced therapeutic solutions. CROs are developing specialized workflows to analyze complex biological samples and high-dimensional datasets which represents their movement toward adopting next-generation analytical testing methods.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 387.25 Million |
|
Market size value in 2026 |
USD 446.69 Million |
|
Revenue forecast in 2033 |
USD 1213.43 Million |
|
Growth rate |
CAGR of 15.35% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Thermo Fisher, Agilent, Danaher, Bruker, Waters, Shimadzu, PerkinElmer, Eurofins, Charles River, SGS, Intertek, Covance, Labcorp, WuXi AppTec, Syneos Health |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Small Molecule Analysis, Large Molecule Analysis, Others), By Application (Drug Discovery, Clinical Trials, Proteomics, Others), By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others), By Technology (LC-MS, GC-MS, MALDI-TOF, Others) |
Which Regions are Driving the Europe CRO Mass Spectroscopy Services Market Growth?
Western Europe leads the Europe CRO Mass Spectroscopy Services Market because its pharmaceutical industry operates under strict EU drug safety regulations. The pharmaceutical research and development centers in Germany, France, and the United Kingdom create ongoing requirements for exact scientific measurements. The academic institutes and biotech clusters and CRO networks form strong connections which help drive service outsourcing to outside companies. Advanced LC-MS systems together with established compliance procedures enable organizations to use their premium mass spectrometry services. The ecosystem enables Western Europe to develop its drug testing operations while conducting specialized biologics research, which establishes Western Europe as the leading region for scientific testing services.
Northern Europe serves as a dependable market segment that has reached full development for the Europe CRO Mass Spectroscopy Services Market because it benefits from strong healthcare systems and ongoing financial backing from established pharmaceutical companies. The region does not follow the Western European trend of expanding through scale but instead establishes its presence through enduring research and development collaborations and stable international regulations in Sweden and Denmark and the Netherlands. The shipping industry and research institutions require analytical testing services, which include environmental assessments and life sciences research. The strong partnership between public and private sectors creates a stable environment for CRO work in Northern Europe, which functions as a dependable revenue source that does not depend on fluctuating market conditions for its business growth.
Eastern Europe experiences the highest growth rate because it develops its affordable CRO facilities and receives more international funding for scientific research. The EU-funded programs and national efforts in Poland and Hungary have improved laboratory capabilities which resulted in Western European pharmaceutical companies outsourcing their work to these countries. The emerging biotech clusters have adopted mass spectrometry services at a faster rate because they now have better skilled workers and lower business expenses. The European testing network for analytical work has started to shift its operational base away from its existing testing centers toward more distributed testing locations. The period from 2026 to 2033 will establish this region as the main area where CROs and investors will expand their operations through cost-effective service delivery methods.
Who are the Key Players in the Europe CRO Mass Spectroscopy Services Market and How Do They Compete?
The Europe CRO Mass Spectroscopy Services Market shows moderate consolidation because two major global laboratory networks and specialized CROs together control most of the market share while smaller regional companies fight for specific market segments. Clients need to establish regulatory compliance standards which require them to maintain data integrity while achieving fast results with their complex LC-MS and GC-MS testing procedures. Established companies maintain their market position through their recognized laboratory networks and their ISO and GLP certifications and their investments in advanced high-resolution equipment. Mid-sized CROs disrupt markets through their ability to deliver projects more rapidly while providing customized service solutions that meet the specific needs of biotech companies.
The technology-based strategy of Eurofins Scientific International enables the company to strengthen its market position through its development of high-resolution LC-MS technology which it plans to implement in its European laboratories. The company has created a competitive advantage through its implementation of standardized global workflows which enable multinational pharmaceutical companies to easily compare their data across different locations. The company has expanded its operations through the strategic acquisition of analytical laboratories located in Eastern Europe which has enabled better operational efficiency and increased geographical presence. SGS competes through its regulatory knowledge which it uses to offer clients access to its extensive compliance certification network for both clinical trial verification and toxicology assessment services. Labcorp provides complete clinical and bioanalytical solutions through its international operations which enable it to assist drug development efforts for clients worldwide.
Thermo Fisher Scientific provides its unique services through instrumentation-based solutions which combine its proprietary mass spectrometry technology with CRO testing services to achieve better detection capabilities and operational efficiency. The company establishes a strong technological advantage through its complete integration of hardware components with software products and service offerings. Charles River Laboratories establishes its market position by offering specialized preclinical and translational research services which focus on biologics and safety evaluation testing. The company establishes its expansion plan through partnerships with biotech companies and its development of advanced bioanalytical facilities throughout Europe which will allow them to access critical drug development processes.
Company List
- Thermo Fisher
- Agilent
- Danaher
- Bruker
- Waters
- Shimadzu
- PerkinElmer
- Eurofins
- Charles River
- SGS
- Intertek
- Covance
- Labcorp
- WuXi AppTec
- Syneos Health
Recent Development News
In April 2026, Eurobio Scientific Acquires CareDx’s Lab Products Division to Expand Transplant Diagnostics Portfolio: French diagnostics company Eurobio Scientific announced a €145 million acquisition of CareDx’s transplant laboratory products business. The deal strengthens Eurobio’s genomics and molecular diagnostics capabilities, including next-generation sequencing and HLA typing solutions widely used in CRO and mass spectrometry-supported clinical workflows across Europe.
Source: https://live.euronext.com
In April 2026, Eurofins Sells MET Labs Unit to UL Solutions to Refocus on Core Pharmaceutical Testing Services: Luxembourg-based Eurofins Scientific agreed to divest its electrical testing business, MET Labs, for €575 million. The company stated the proceeds would support investments in pharmaceutical testing, analytical chemistry, AI, and laboratory automation — key growth areas connected to Europe’s CRO and mass spectrometry services ecosystem.
Source: https://ca.marketscreener.com
What Strategic Insights Define the Future of the Europe CRO Mass Spectroscopy Services Market?
The Europe CRO Mass Spectroscopy Services Market is moving toward analytical outsourcing solutions which handle complex multiple testing methods because biopharmaceuticals and precision medicine development and EU regulatory testing requirements are becoming more restrictive. Pharmaceutical companies will increasingly adopt integrated LC-MS and high-resolution workflows during the next 5 to 7 years because they prefer to contract their complete analytical needs instead of separate testing services. Cost pressures together with faster regulatory submission requirements have created a new development path which the industry now follows.
The operational risk which organizations face emerges from their growing reliance on a restricted group of advanced equipment distributors and common software platforms which lead to vendor dependence and restrict market competitiveness for contract research organizations. At present Eastern Europe offers a significant business opportunity because EU-supported biotechnology development initiatives are establishing specialized testing facilities which global contract research organizations have not yet fully accessed.
Market participants should focus on developing hybrid capabilities which merge AI-powered data analysis with geographically distributed laboratory systems. The company will achieve three goals through this approach: reduced operational delays,better capacity to meet regulatory requirements, and access to future high-demand biologics outsourcing opportunities.
Europe CRO Mass Spectroscopy Services Market Report Segmentation
By Type
- Small Molecule Analysis
- Large Molecule Analysis
- Others
By Application
- Drug Discovery
- Clinical Trials
- Proteomics
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Others
By Technology
- LC-MS
- GC-MS
- MALDI-TOF
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe CRO Mass Spectroscopy Services Market size for the market will be USD 1213.43 Million in 2033.
The key segments of the Europe CRO Mass Spectroscopy Services Market are By Type (Small Molecule Analysis, Large Molecule Analysis, Others), By Application (Drug Discovery, Clinical Trials, Proteomics, Others), By End-User (Pharma Companies, Biotech Firms, Research Institutes, Others), By Technology (LC-MS, GC-MS, MALDI-TOF, Others).
Major players in the Europe CRO Mass Spectroscopy Services Market are Thermo Fisher, Agilent, Danaher, Bruker, Waters, Shimadzu, PerkinElmer, Eurofins, Charles River, SGS, Intertek, Covance, Labcorp, WuXi AppTec, Syneos Health.
The current market size of the Europe CRO Mass Spectroscopy Services Market is USD 387.25 Million in 2025.
The Europe CRO Mass Spectroscopy Services Market CAGR is 15.35%.
- Thermo Fisher
- Agilent
- Danaher
- Bruker
- Waters
- Shimadzu
- PerkinElmer
- Eurofins
- Charles River
- SGS
- Intertek
- Covance
- Labcorp
- WuXi AppTec
- Syneos Health
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)