Europe Advanced Therapy Medicinal Products Market Size & Forecast:
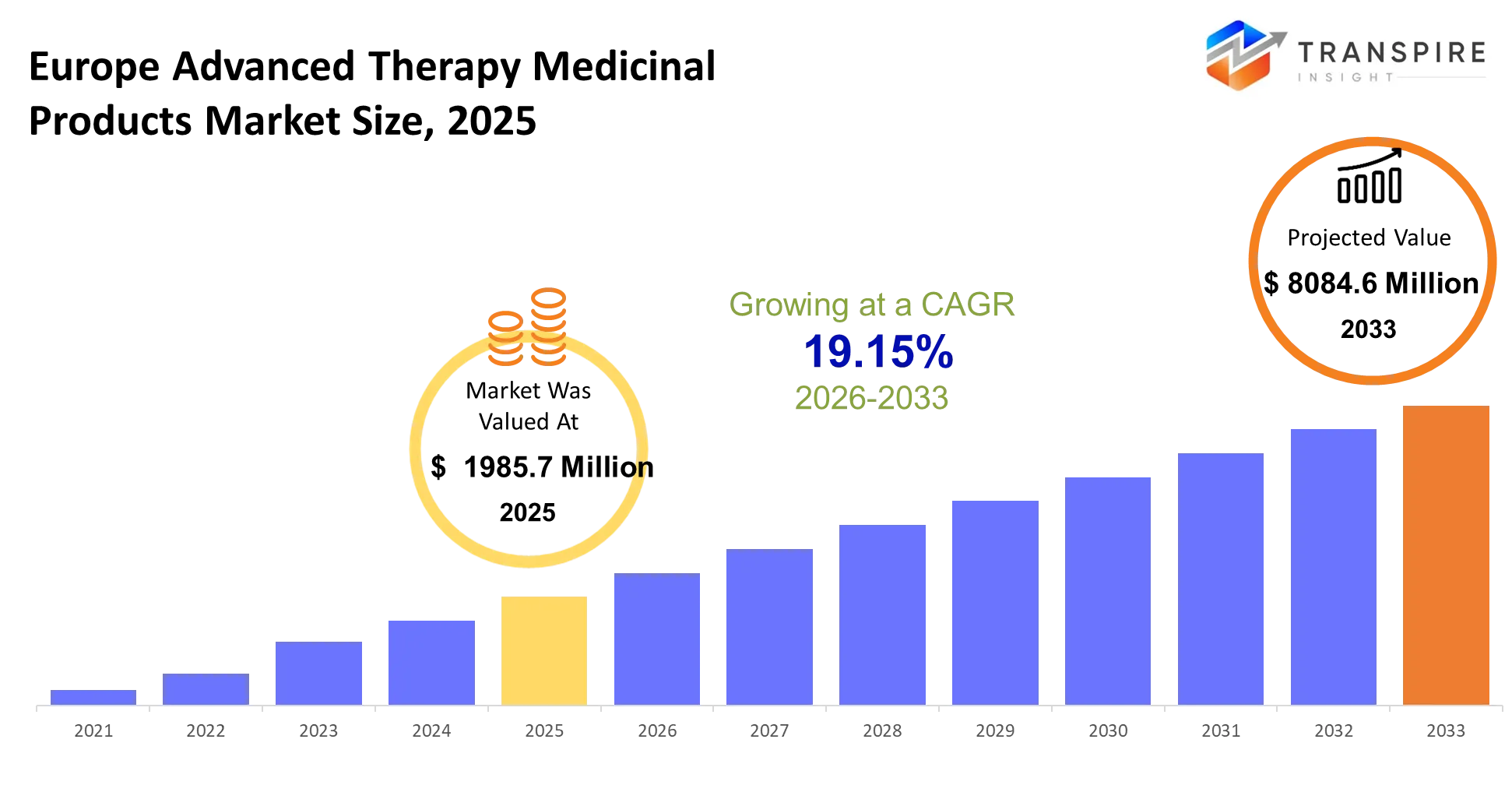
- Europe Advanced Therapy Medicinal Products Market Size 2025: USD 1985.7 Million
- Europe Advanced Therapy Medicinal Products Market Size 2033: USD 8084.6 Million
- Europe Advanced Therapy Medicinal Products Market CAGR: 19.15%
- Europe Advanced Therapy Medicinal Products Market Segments: By Type (Gene Therapy, Cell Therapy, Tissue Engineering, Others); By Application (Oncology, Rare Diseases, Genetic Disorders, Regenerative Medicine, Others); By End-User (Hospitals, Pharma Companies, Biotech Firms, Research Institutes, Others); By Delivery (Viral Vectors, Non-viral Vectors, Ex vivo, In vivo, Others)
To learn more about this report, Download Free Sample Report
Europe Advanced Therapy Medicinal Products Market Summary:
The Europe Advanced Therapy Medicinal Products Market size is estimated at USD 1985.7 Million in 2025 and is anticipated to reach USD 8084.6 Million by 2033, growing at a CAGR of 19.15% from 2026 to 2033. The European advanced therapy medicinal products market functions as a distinct sector within the biopharmaceutical industry which specializes in treatments that use gene and cell and tissue-based therapies. The demand for sustainable personal treatment methods from patients will determine market development because this need arises particularly in case of rare and chronic diseases which lack effective solutions from existing drugs. European regulatory agencies will keep developing approval processes which maintain safety standards while providing quicker market access. Market expansion will occur because of advancements in viral vector technology and gene editing and manufacturing processes which have improved production capabilities. The way treatments reach patients and the way companies market their new solutions will be affected by both pricing challenges and reimbursement discussions.
What Has the Impact of Artificial Intelligence Been on the Europe Advanced Therapy Medicinal Products Market?
The European market for advanced therapy medicinal products uses artificial intelligence to improve decision-making and operational processes which handle the complex demands of its biopharmaceutical sector. The European advanced therapy medicinal products market uses AI technology to enhance market research by processing clinical and genomic data plus real-world datasets which were previously difficult to interpret at large scale. Companies use machine learning and predictive analytics to identify emerging market trends while creating forecasts for therapy demand and developing patient-specific products. European advanced therapy medicinal products market stakeholders can now implement insights faster because this shift leads to better outcomes in both strategic planning and investment activities.
Artificial intelligence systems use intelligent automation to advance operational activities in the European advanced therapy medicinal products market by automating both manufacturing facilities and supply chain operations. AI systems control the production of cell and gene therapies by establishing operational standards which lead to consistent production quality and decreased operational variability. Predictive models enable supply chain networks to predict potential disruptions which lead to better inventory management and reduced total operating expenses. The European advanced therapy medicinal products market uses artificial intelligence to create personalized solutions which help companies develop new products at a faster rate while gaining a competitive advantage against rival businesses through successful technology implementation.
Key Market Trends & Insights:
- The advanced therapy medicinal products market in Europe allows Western Europe to become the leading market because it has advanced healthcare systems which enable them to obtain 65% of the total market share in 2025.
- The biotech industry expansion will enable Eastern Europe to become the fastest-growing region which will achieve more than 18% compound annual growth rate from 2025 to 2032.
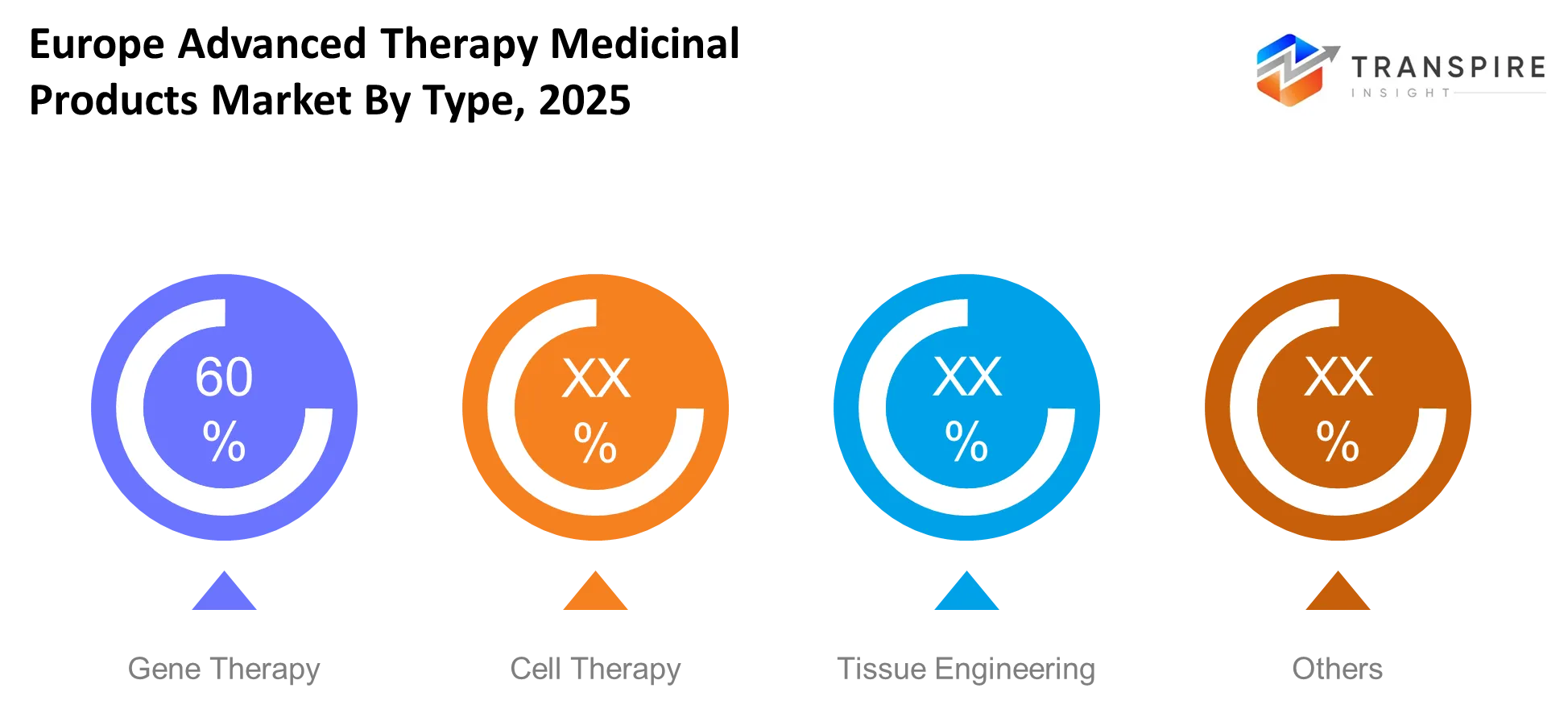
- The gene therapy segment leads the market with its more than 40% market share which results from multiple product approvals and active research development in rare disease therapies.
- The cell therapy market segment ranks as the second largest because the industry experiences more oncology applications and higher success rates for clinical trials.
- The tissue-engineered products market segment experiences the fastest growth rate which will continue until 2030 because of increasing demand for regenerative medicine products.
- The oncology field uses 55% of the total market share because people want personalized cancer therapies which target their specific needs.
- The application of rare genetic disorders has become the fastest growing field because orphan drug incentives support agencies which need new clinical solutions.
- The medical system operates through hospitals and specialty clinics which hold more than 60% market share because they possess superior treatment facilities and qualified staff members.
- The research institutes and biotechnology laboratories which use our products.
- The research and development funding increase together with clinical trial growth lead research institutes and biotechnology laboratories to become new end-user markets.
- The combination of AI-based drug discovery technology and intelligent manufacturing systems results in improved operational efficiency and enhanced market competitiveness.
Europe Advanced Therapy Medicinal Products Market Segmentation
By Type:
The research section divides into four types which include Gene Therapy Cell Therapy Tissue Engineering and other unspecified categories. Gene therapy will focus on correcting defective genes which provide long-term treatment possibilities. Cell therapy will use living cells to repair or replace damaged tissues with a focus on treating cancer. Tissue engineering will enable organs and tissues to undergo regenerative processes. Other approaches will include emerging hybrid therapies which combine multiple advanced techniques.
To learn more about this report, Download Free Sample Report
By Application:
Oncology will remain a leading application due to increasing cancer cases and demand for precision treatment. Rare diseases will gain focus with supportive policies. Genetic disorders will see targeted solutions through gene editing. Regenerative medicine will expand steadily, while other applications will include niche therapeutic areas under development.
By End-User:
Hospitals will act as primary treatment centers with required infrastructure. Pharma companies will drive commercialization and large-scale production. Biotech firms will lead innovation and early-stage development. Research institutes will support clinical trials and discoveries, while other end-users will include specialized therapy centers and academic collaborations.
By Delivery:
Viral vectors will dominate delivery due to high efficiency in gene transfer. Non-viral vectors will offer safer alternatives with ongoing improvements. Ex vivo methods will involve modifying cells outside the body before reintroduction. In vivo delivery will target cells directly inside the body, while other methods will continue to evolve with research advancements.
What are the Main Challenges for the Europe Advanced Therapy Medicinal Products Market Growth?
The implementation of advanced therapy medicinal products in Europe faces serious technical and operational difficulties which will medically slow down their widespread acceptance. The product stability issue persists as a major challenge that affects both cell and gene therapies which depend on specific handling and storage protocols. The treatment process requires individualized patient treatment which will prevent the system from achieving its full operational capacity. The Europe advanced therapy medicinal products market will face continuous growth impediments because of supply chain limitations which include challenges in maintaining cold chain operations and shortages of essential raw materials.
The European advanced therapy medicinal products market will experience restricted growth because of obstacles that exist in both the production and marketing processes. The production process will need advanced manufacturing facilities which must comply with stringent Good Manufacturing Practice standards to achieve operational requirements. The high expenses related to product development and manufacturing will create difficulties for companies that need to set pricing because it will make their treatments less affordable. The regulatory approval process will require extensive assessment periods which will lead to product release delays while increasing financial risks for businesses in the European advanced therapy medicinal products market.
The combination of existing infrastructure limitations and insufficient availability of professionals who possess advanced therapy skills will maintain adoption difficulties throughout the process. The healthcare systems in emerging European regions will encounter difficulties when they attempt to implement these treatments within their current medical frameworks. The market will experience restrictions which prevent patient access because funding gaps and reimbursement uncertainties serve as primary market obstacles. The advanced therapy medicinal products market in Europe will experience uneven market adoption because of regional differences in healthcare environments.
The development of alternative treatment methods together with changing regulatory requirements will create new business risks for organizations. The stakeholders will increase pricing pressure because they require affordable products while the rapid advancement of technology will force businesses to spend more money, which creates obstacles for the development of Europe advanced therapy medicinal products market.
Regional Insights
The European advanced therapy medicinal products market will exhibit different growth patterns because European countries have distinct healthcare systems and their regulatory frameworks and research capabilities differ from one another. The development of various regions will depend on their capacity to invest in innovative technologies and clinical research whereas their ability to deliver new treatments to patients will be determined by their current access to advanced medical facilities.
Western Europe will lead the market because its established healthcare systems and its capacity to spend more money and its regulatory framework will enable quicker product development and market entry. The revenue growth will be driven by Germany and France. Northern Europe will experience steady growth because research funding will support its development. Eastern Europe will experience slow growth until improvements in infrastructure development and investment activities begin to take effect.
Recent Development News
In April 2026, Novartis received a positive CHMP opinion for its gene therapy Itvisma aimed at treating spinal muscular atrophy, marking a significant step toward EU approval and broader patient access. https://www.rttnews.com
In April 2026, Gilead Sciences announced acquisition of German biotech Tubulis. The deal, valued at up to $3.15 billion, will strengthen antibody-drug conjugate capabilities and expand oncology-focused advanced therapy pipelines. https://www.reuters.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1985.7 Million |
|
Market size value in 2026 |
USD 2371.5 Million |
|
Revenue forecast in 2033 |
USD 8084.6 Million |
|
Growth rate |
CAGR of 19.15% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Novartis, Gilead, Roche, Pfizer, Bristol Myers Squibb, AstraZeneca, Sanofi, Johnson & Johnson, Bluebird Bio, Spark Therapeutics, CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, Sarepta, Takeda |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Gene Therapy, Cell Therapy, Tissue Engineering, Others); By Application (Oncology, Rare Diseases, Genetic Disorders, Regenerative Medicine, Others); By End-User (Hospitals, Pharma Companies, Biotech Firms, Research Institutes, Others); By Delivery (Viral Vectors, Non-viral Vectors, Ex vivo, In vivo, Others) |
How Can New Companies Establish a Strong Foothold in the Europe Advanced Therapy Medicinal Products Market?
New entrants aiming to build a strong position in the europe advanced therapy medicinal products market will need to focus on targeted innovation rather than broad competition. The europe advanced therapy medicinal products market rewards companies that address unmet clinical needs, especially in rare diseases and personalized therapies. The emerging players will achieve early-stage market expansion by focusing on specific indications and using advanced technologies which include gene editing and cell engineering.
The europe advanced therapy medicinal products market requires strategic partnerships to help businesses break through their entry obstacles. New companies will obtain necessary infrastructure and regulatory knowledge and funding through their partnerships with established pharmaceutical companies and research institutions and contract manufacturing organizations. The use of artificial intelligence to drive drug discovery processes together with automated manufacturing processes will create unique advantages which make products more efficient while shortening timeframes needed for development.
The europe advanced therapy medicinal products market will become more competitive through the development of solutions which solve production cost problems and scalability restrictions and logistical challenges. Companies that develop cost-effective manufacturing models or scalable delivery systems will gain faster acceptance. Startups such as CRISPR Therapeutics show how their gene-editing platforms lead to accelerated innovation while emerging biotech companies use modular manufacturing systems to make their products more accessible.
The European advanced therapy medicinal products market will achieve lasting success through its requirement to balance two key elements which are innovative development and compliance with regulatory standards and product commercialization preparation. New companies that invest in robust clinical validation adaptive pricing models and regional expansion strategies will enhance their credibility while establishing sustainable market share in a highly competitive environment.
Key Europe Advanced Therapy Medicinal Products Market Company Insights
The European Advanced Therapy Medicinal Products Market will experience high market competition because of two factors, which are product innovations and regulatory body compliance and successful clinical outcomes. The market players will dedicate their efforts to developing new treatment options while they work to achieve better patient results. Organizations will establish their competitive standing through three factors, which are their research capabilities and manufacturing resources and their capacity to adapt to changing medical requirements.
The leading companies will enhance their market positions through ongoing product development and the introduction of new advanced therapies. The partnership between biotech companies and major pharmaceutical firms will enable both companies to bring their products to market more quickly while expanding their distribution network. The investment in technology through automation and data-driven research will result in better operational efficiency. The two factors of regional business development and pricing strategies will create additional advantages for competing companies.
Company List
- Novartis
- Gilead
- Roche
- Pfizer
- Bristol Myers Squibb
- AstraZeneca
- Sanofi
- Johnson & Johnson
- Bluebird Bio
- Spark Therapeutics
- CRISPR Therapeutics
- Editas Medicine
- Intellia Therapeutics
- Sarepta
- Takeda
What are the Key Use-Cases Driving the Growth of the Europe Advanced Therapy Medicinal Products Market?
The europe advanced therapy medicinal products market is gaining momentum through high-impact use-cases in oncology, where gene and cell therapies are transforming cancer treatment. The clinical adoption of CAR-T therapies and gene-modified immune cells is increasing because these treatments lead to better blood cancer patient survival rates. The use-case for personalized treatment models keeps expanding because hospitals adopt these models which directly contribute to market development.
The europe advanced therapy medicinal products market uses another important application which supports medical treatment of rare and genetic disorders. Gene therapies provide patients with spinal muscular atrophy and inherited retinal diseases two treatment choices that include permanent solutions and single-time therapies. These applications solve unmet medical requirements which lead to regulatory approval and funding support that enables faster market entry throughout European healthcare systems.
The europe advanced therapy medicinal products market benefits from regenerative medicine which specializes in tissue repair and organ regeneration. The medical field is seeing increasing use of applications which support wound healing and orthopedic treatment and cardiovascular system restoration. Specialized clinics and research centers are adopting these therapies which enables them to provide new treatment options that can be expanded to larger populations.
Advanced therapies are transforming biomanufacturing and research fields by their ability to create precise medical treatments and develop products through data analysis. The europe advanced therapy medicinal products market will experience growth through two emerging trends which include AI-integrated therapy design and modular production systems that will create new applications while enhancing operational efficiency and sustaining long-term business growth.
Europe Advanced Therapy Medicinal Products Market Report Segmentation
By Type
- Gene Therapy
- Cell Therapy
- Tissue Engineering
- Others
By Application
- Oncology
- Rare Diseases
- Genetic Disorders
- Regenerative Medicine
- Others
By End-User
- Hospitals
- Pharma Companies
- Biotech Firms
- Research Institutes
- Others
By Delivery
- Viral Vectors
- Non-viral Vectors
- Ex vivo
- In vivo
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Advanced Therapy Medicinal Products Market size for the market will be USD 8084.6 Million in 2033.
Key segments for the Europe Advanced Therapy Medicinal Products Market are By Type (Gene Therapy, Cell Therapy, Tissue Engineering, Others); By Application (Oncology, Rare Diseases, Genetic Disorders, Regenerative Medicine, Others); By End-User (Hospitals, Pharma Companies, Biotech Firms, Research Institutes, Others); By Delivery (Viral Vectors, Non-viral Vectors, Ex vivo, In vivo, Others).
Major Europe Advanced Therapy Medicinal Products Market players are Novartis, Gilead, Roche, Pfizer, Bristol Myers Squibb, AstraZeneca, Sanofi, Johnson & Johnson, Bluebird Bio, Spark Therapeutics, CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, Sarepta, Takeda.
The Europe Advanced Therapy Medicinal Products Market size is USD 1985.7 Million in 2025.
The Europe Advanced Therapy Medicinal Products Market CAGR is 19.15%.
- Novartis
- Gilead
- Roche
- Pfizer
- Bristol Myers Squibb
- AstraZeneca
- Sanofi
- Johnson & Johnson
- Bluebird Bio
- Spark Therapeutics
- CRISPR Therapeutics
- Editas Medicine
- Intellia Therapeutics
- Sarepta
- Takeda
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









