Asia Pacific STD Testing Devices Market Size & Forecast:
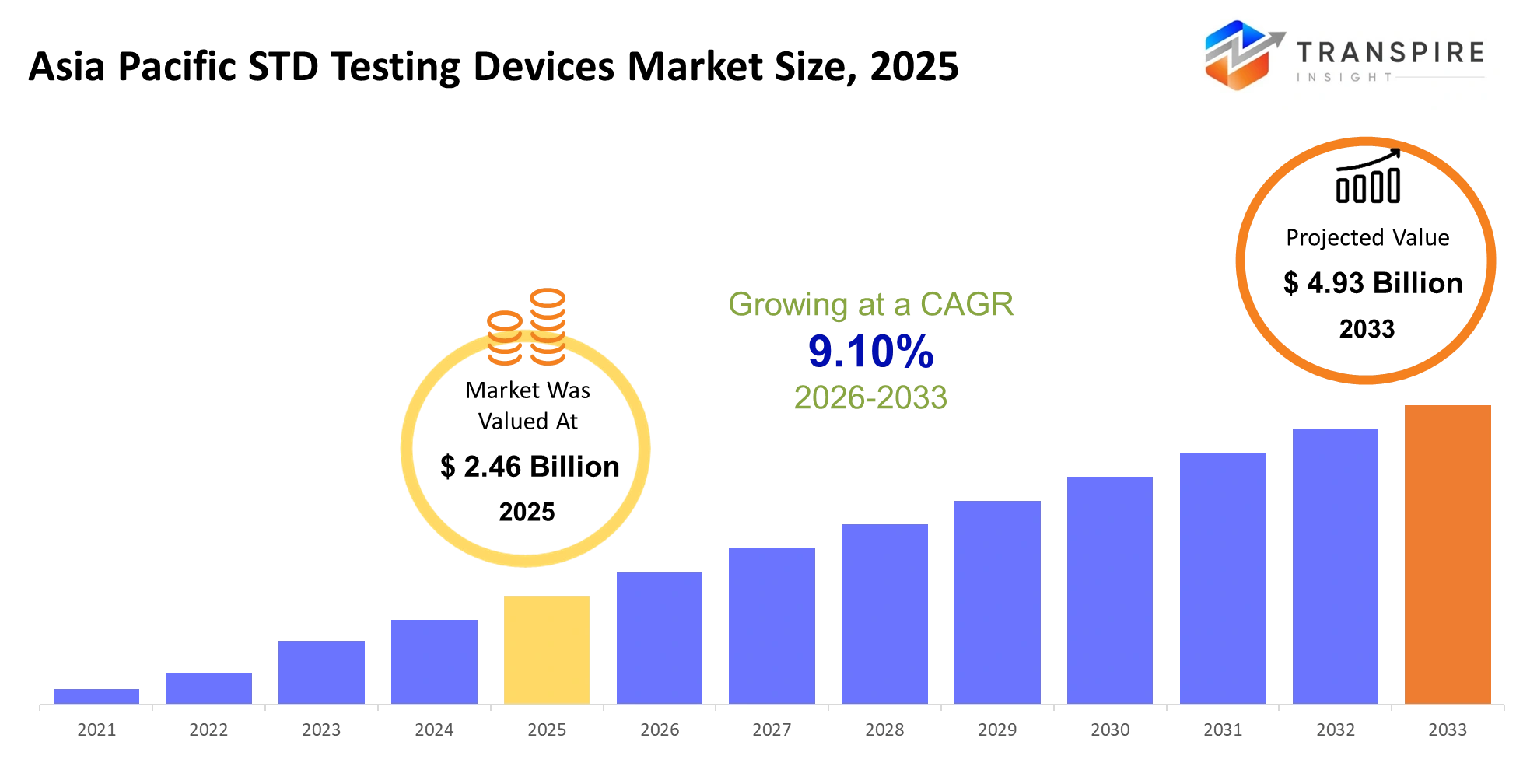
- Asia Pacific STD Testing Devices Market Size 2025: USD 2.46 Billion
- Asia Pacific STD Testing Devices Market Size 2033: USD 4.93 Billion
- Asia Pacific STD Testing Devices Market CAGR: 9.10%
- Asia Pacific STD Testing Devices Market Segments: By Type (Rapid Test Kits, Molecular Diagnostics Devices, Laboratory Equipment, Others); By Application (HIV Testing, HPV Testing, Syphilis Testing, Gonorrhea Testing, Chlamydia Testing, Others); By End-User (Hospitals, Clinics, Diagnostic Labs, Research Institutes, Homecare, Others); By Technology (PCR, Immunoassay, Nucleic Acid Testing, Others).
To learn more about this report, Download Free Sample Report
Asia Pacific STD Testing Devices Market Summary
The Asia Pacific STD Testing Devices Market was valued at USD 2.46 Billion in 2025. It is forecast to reach USD 4.93 Billion by 2033. That is a CAGR of 9.10% over the period.
The Asia Pacific STD Testing Devices Market supports clinical and at-home detection of sexually transmitted infections which enables healthcare organizations and testing facilities and health authorities to detect infections at early stages and prevent disease outbreaks. The devices function to decrease time needed for diagnosis while they enable healthcare facilities to deliver precise treatment and establish monitoring systems in locations with high population density and ongoing human movement.
The market during the past three to five years has transitioned from testing procedures that required centralized laboratory facilities to testing methods which enable immediate results through point-of-care testing and self-testing. Testing facilities now operate through decentralized systems which provide testing services at both clinical sites and pharmacy locations and residential environments. The COVID-19 pandemic operated as a significant catalyst because it stopped traditional screening methods while it increased acceptance of remote diagnostics and home-based testing procedures. The demand currently exists because customers expect quicker results and multiple access options which leads manufacturers to develop portable devices that customers can easily operate and which allow digital results to connect with various healthcare systems.
Key Market Insights
- China leads the Asia Pacific STD Testing Devices Market because its extensive screening programs provide the country with an estimated 30 to 35 percent market share.
- India stands as the most rapidly expanding area because its public health programs develop while STI testing rates increase until 2032.
- The advanced diagnostic systems and precise testing needs of Japan and South Korea create continuous market demand for testing services.
- The Asia Pacific STD Testing Devices Market maintains its leading position because molecular diagnostic devices provide accurate results for detecting early-stage infections.
- The second-largest market share for immunoassay-based testing kits exists because they provide cost-effective solutions that clinics throughout the world use.
- The segment of rapid point-of-care testing devices shows the highest growth because customers want speedier diagnostic methods which can operate outside traditional medical facilities.
- HIV and chlamydia testing applications dominate with nearly 40% combined share due to high screening frequency programs.
- The testing of gonorrhea and syphilis emerged as the most rapidly expanding field because urban centers experienced increased rates of infection identification.
- The Asia Pacific STD Testing Devices Market shows its highest testing volume which hospitals and diagnostic laboratories control with their combined testing operations.
- The self-testing and homecare users group shows the highest growth because diagnostic users choose tests which protect their privacy while providing them with convenient testing solutions.
What are the Key Drivers, Restraints, and Opportunities in the Asia Pacific STD Testing Devices Market?
The Asia Pacific STD Testing Devices Market experiences its highest growth rate because decentralized testing and rapid diagnostic testing have become standard medical procedures since molecular amplification technologies and compact immunoassay systems became available. The COVID-19 pandemic created a testing emergency because hospitals reached their maximum capacity, which forced healthcare systems to develop testing methods that could perform rapid testing across multiple locations. The testing volume increased because hospitals started using the technology while users could now access testing services through clinics and pharmacies and home testing. Indian and Chinese governments have established screening programs which create regular purchasing patterns that help diagnostic manufacturers achieve steady revenue flow.
The high-cost barrier prevents advanced STD testing devices from being accessible to lower-income residents and rural communities throughout the Asia Pacific region. The expensive molecular diagnostic platforms together with insufficient laboratory capacity create an essential barrier which requires more time than temporary policy solutions can provide. The testing gap prevents early disease recognition among disadvantaged groups while it inhibits market growth for high-accuracy testing technologies which need specialized skills and secure testing environments.
The combination of multiplex molecular testing platforms with digital health ecosystems creates a new opportunity for development. Abbott Laboratories develops portable STI panels which enable multiple infection testing through a single diagnostic test. Southeast Asia experiences an increase in telehealth diagnostics funding which allows users to collect samples at home while accessing cloud reporting services, making it possible to expand services into regions that previously had low service availability.
What Has the Impact of Artificial Intelligence Been on the Asia Pacific STD Testing Devices Market?
Artificial intelligence and advanced digital systems are transforming marine emission control technology and scrubber performance systems because they create continuous optimization systems from existing methods that react to events. The system now uses AI technology through its monitoring platforms which collect sensor data from exhaust gas cleaning systems to control wash water flow and pressure levels and chemical dosing in real time without human assistance.
Machine learning models use historical engine load data and corrosion rate information and particulate accumulation records to predict when maintenance work will be needed and which system components will fail. Operators have reported better fuel efficiency through optimized operating windows because the shift from scheduled servicing to condition-based maintenance brings greater vessel availability and decreases unplanned scrubber downtime.
AI-based fleet compliance dashboards help ship operators predict emissions according to IMO standards while they try to achieve regulatory compliance and reduce fuel use. The technology faces challenges because high-quality maritime datasets and stable sea connectivity restrict both real-time model updates and remote system calibration. The high costs required to integrate new technologies with existing vessel systems create obstacles for mid-tier fleet operators who want to implement their solutions throughout their operations across the Asia Pacific region.
Key Market Trends
- The usage of molecular diagnostics increased immediately after 2021 when hospitals and laboratories throughout Asia Pacific began using these tests instead of traditional culture methods.
- China expanded national STI screening coverage beyond 60% urban clinics by 2025 which led to standardized device procurement across public health systems.
- The usage of rapid point-of-care testing increased in India after 2022 because pharmacies and community health centers began distributing diagnostic tests.
- Manufacturers developed multiplex testing platforms to decrease diagnostic turnaround time by moving from single-disease kits to multi-infection detection.
- The usage of self-testing devices increased through the pandemic period because urban residents changed their sexual health testing practices to protect their personal information.
- Abbott Laboratories developed new portable STI testing solutions which enable better decentralized diagnostic testing in Southeast Asia in 2023.
- Digital reporting systems replaced paper lab reports at multiple urban hospitals which enhanced their traceability capabilities and supported real-time disease monitoring efforts.
- The competitive environment for AI-assisted diagnostics development shifted toward companies that focused on using automation to improve result interpretation and decrease errors in their operations.
- The Asia Pacific region experienced increased supply chain localization after 2022 because companies began developing local manufacturing capabilities to replace imported diagnostic kits.
Asia Pacific STD Testing Devices Market Segmentation
By Type :
The testing kits enable healthcare workers to identify infections through instant results which they can obtain within minutes. The molecular diagnostics equipment identifies genetic material to provide better diagnostic results. The laboratory equipment enables scientists to conduct precise experiments within controlled environments. Healthcare systems use various diagnostic tools which include supportive diagnostic devices for their different screening requirements.
The technology selection process uses three criteria which include selecting between faster systems and precise systems and cheaper systems. The immediate screening process uses rapid kits while molecular tools provide testing results for confirmation purposes. Laboratories are necessary for processing large quantities of samples. The devices enable testing in remote areas and regular healthcare facilities throughout various healthcare networks across different locations.

To learn more about this report, Download Free Sample Report
By Application :
The need for HIV testing remains important because of the substantial need to track infections. HPV testing supports cervical cancer prevention programs. The testing process for syphilis allows healthcare providers to identify cases and manage them effectively. The gonorrhea test evaluates patients who have suspected bacterial infections. Chlamydia testing addresses common sexually transmitted infection screening. The other applications handle both new and uncommon infectious disease diagnostic needs.
The demand for testing has increased because people became more aware of health risks and screening initiatives and preventive medical programs began. The tests assist hospital laboratories and clinics with their initial patient evaluations. The research institutes use these methods for their investigative work. The new screening approach enables health systems to find more infected cases which leads to better treatment results in public and private healthcare facilities.
By End-User :
The hospital system operates essential testing laboratories through its advanced diagnostic testing facilities. The clinic system provides outpatient services which use screening methods for early disease identification. The diagnostic laboratories process extensive sample quantities through their advanced testing equipment. Research institutes dedicate their work to both scientific investigations and technological development. Homecare solutions allow users to perform self-testing while their health conditions are monitored from a distance. The health programs and mobile units make up the remaining group of end-users who use the system.
Testing devices become popular with end-users who consider three main factors which include device availability and testing expenses and necessary precision level. The medical testing field is mainly controlled by hospitals and laboratories because their testing facilities provide them with better operational capabilities. The clinic system enhances its service area through its expanding network of medical facilities. The demand for homecare services increases because people want to perform self-testing. Research activities contribute to the development of new diagnostic techniques which improve disease detection methods that work in both urban and rural areas.
By Technology :
PCR technology enables the process of genetic material amplification which results in accurate pathogen identification through its infection detection methods. The immunoassay testing procedure uses antigen-antibody interactions to deliver immediate results for infection detection. Nucleic acid testing offers advanced pathogen detection capabilities through its ability to identify pathogens at their most basic molecular form. These diagnostic technologies provide dependable medical assessments which healthcare facilities use for both clinical practice and laboratory testing.
The decision to adopt a technology depends on three factors which include its testing accuracy and operational speed and complete testing capabilities. The PCR testing method continues to serve as the accepted benchmark for precise testing results. The immunoassay testing method provides essential support for quick testing procedures. Nucleic acid testing enables healthcare professionals to achieve both high detection rates and quick results. The healthcare sector benefits from using these methods together because they improve disease detection capabilities and diagnostic efficiency in hospitals and laboratories.
What are the Key Use Cases Driving the Asia Pacific STD Testing Devices Market?
The main application area for STD Testing Devices in the Asia Pacific region is hospital and diagnostic laboratory-based screening of HIV and chlamydia and syphilis infections. The facilities with the highest testing requirements operate under high patient volume conditions while using advanced molecular and immunoassay testing methods to meet their clinical and public health reporting needs.
The current testing scope has expanded to include community health clinics and pharmacy-led testing services which are becoming more popular across India and Southeast Asia because of decentralized healthcare systems that are emerging in those regions. Home-based self-testing kits are becoming more popular among urban residents and private healthcare users because people want to obtain fast and private medical evaluations that do not require hospital visits.
The new use case enables patients to perform STD testing through telehealth platforms which provide them with testing kits and results via mobile health applications. The application of health screening programs is expanding to workplace and travel environments which involve cross-border labor centers and tourism-heavy markets because organizations and government officials are enforcing stricter health regulations.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 2.46 Billion |
|
Market size value in 2026 |
USD 2.68 Billion |
|
Revenue forecast in 2033 |
USD 4.93 Billion |
|
Growth rate |
CAGR of 9.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Asia Pacific (India, China, Japan, Australia,South Korea, Rest of Asia Pacific) |
|
Key company profiled |
Abbott, Roche, Thermo Fisher, Siemens Healthineers, BD, Bio-Rad, Hologic, Qiagen, Danaher, PerkinElmer, Agilent, Sysmex, Cepheid, bioMérieux, Fujirebio. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Rapid Test Kits, Molecular Diagnostics Devices, Laboratory Equipment, Others); By Application (HIV Testing, HPV Testing, Syphilis Testing, Gonorrhea Testing, Chlamydia Testing, Others); By End-User (Hospitals, Clinics, Diagnostic Labs, Research Institutes, Homecare, Others); By Technology (PCR, Immunoassay, Nucleic Acid Testing, Others). |
Which Regions are Driving the Asia Pacific STD Testing Devices Market Growth?
China leads the Asia Pacific STD Testing Devices Market because its public health screening programs and diagnostic manufacturing ecosystem support extensive testing operations. Government-backed STI surveillance initiatives have expanded testing coverage across urban and semi-urban hospitals which established ongoing procurement processes for medical equipment. The country also benefits from a dense network of accredited laboratories that support high-throughput molecular diagnostics.Established diagnostic firms in the country maintain strong domestic production capacity which decreases the need for imports while ensuring product availability. The integrated system enables testing facilities to implement both centralized and decentralized operations at an accelerated pace.
Japan serves as a stable technological partner who displays mature technological capacities but his development path differs from Chinese expansion which relies on market volume growth. The market here receives support through the combination of stringent clinical quality requirements and ongoing funding for advanced diagnostic equipment which delivers precise results. The healthcare system which follows structured pathways requires regular infectious disease testing because of the increasing number of elderly people. Japan prefers testing methods which provide dependable results through precise measurements and legal compliance instead of quick market introduction. The business model generates stable revenue streams which result from hospitals buying products for extended periods and continuously updating their high-tech diagnostic systems.
The decentralized healthcare access system and growing STI awareness campaigns make India the fastest developing region in the world. The Indian government has expanded testing capabilities through its recent investments in primary healthcare infrastructure and pharmacy-based diagnostic systems. The development process has gained speed because both digital health implementation and affordable rapid testing kits have become more common in semi-urban regions. India presents investors and market newcomers with significant growth opportunities that will last until 2026 through 2033 because the demand for scalable affordable mobile diagnostic solutions will increase.
Who are the Key Players in the Asia Pacific STD Testing Devices Market and How Do They Compete?
The Asia Pacific STD Testing Devices Market shows moderate consolidation, which enables global diagnostic companies to compete against local manufacturers in both hospital and decentralized testing markets. The existing companies in the market protect their market share through technological innovations, while molecular accuracy and multiplex detection capabilities serve as essential criteria for customers. Competition now centers on rapid turnaround time, integration with digital reporting systems, and ability to support large-scale public health screening programs. Emerging economies continue to experience pricing pressure that affects procurement choices yet tertiary hospitals have strong demand for premium systems.
Abbott Laboratories focuses on portable rapid diagnostic platforms which enable decentralized STI screening throughout India and Southeast Asia because of their public health partnerships. Roche competes through its molecular diagnostic technology which delivers high accuracy results and its automated laboratory systems which serve hospitals that need high testing capacity and precise results. Siemens Healthineers establishes its competitive edge through its lab automation systems which provide complete laboratory solutions that unify diagnostic testing with data management and hospital information technology systems to expand its market presence in modern healthcare facilities.
Danaher Corporation uses cartridge-based GeneXpert systems which enable quick multiplex PCR testing because they require limited laboratory equipment. bioMérieux focuses on infectious disease diagnostics with hybrid culture and molecular platforms backed by strong regulatory approvals. Hologic specializes in women's health STI assays which include chlamydia and gonorrhea testing, through partnerships with clinics throughout Asia Pacific.
Company List
- Abbott
- Roche
- Thermo Fisher
- Siemens Healthineers
- BD
- Bio-Rad
- Hologic
- Qiagen
- Danaher
- PerkinElmer
- Agilent
- Sysmex
- Cepheid
- bioMérieux
- Fujirebio
Recent Development News
In January 2026, OraSure Technologies announced submission of its rapid molecular self-test for Chlamydia trachomatis and Neisseria gonorrhoeae along with an at-home urine collection device for STI testing to the U.S. FDA. The development strengthens the company’s decentralized testing portfolio, which is increasingly relevant to Asia Pacific markets where demand for home-based STD diagnostics is rising.
Source: https://www.nasdaq.com/
In April 2026, South Korea’s Ministry of Food and Drug Safety (MFDS) expanded the approved scope of self-diagnostic devices to include sexually transmitted infections such as syphilis, gonorrhea, and chlamydia. This regulatory shift is expected to accelerate commercialization of STD testing kits by domestic manufacturers and improve access to at-home diagnostic devices across the South Korean market.
Source: https://en.sedaily.com/
What Strategic Insights Define the Future of the Asia Pacific STD Testing Devices Market?
The Asia Pacific STD Testing Devices Market is moving toward decentralized testing which uses digital connections to provide diagnostic services through pharmacies and clinics and home testing systems. The implementation of this system will be enabled through the combination of multiplex molecular testing with digital health platforms which allow faster diagnostic processes and broader screening capabilities to reach both highly populated urban areas and remote rural locations. The upcoming 5 to 7 years will see integrated testing ecosystems become the primary source of value creation instead of separate testing devices.
The growing need for proprietary molecular systems creates a hidden threat which will enable a few global diagnostic companies to dominate the market while public health organizations face restricted purchasing options. The market concentration will force buyers into fixed pricing agreements while they experience supply chain disruptions during periods of increased demand.
The integration of at-home STD testing with telehealth services presents a new growth prospect which shows particular promise in Southeast Asia because of its quick adoption of digital health technologies. This model combines home sample collection with cloud reporting and doctor consultation to establish an effective preventive healthcare system. Market participants should establish partnerships with telehealth providers while developing affordable multiplex testing platforms which can be used in remote areas to meet the rising demand for their services over time.
Asia Pacific STD Testing Devices Market Report Segmentation
By Type
- Rapid Test Kits
- Molecular Diagnostics Devices
- Laboratory Equipment
By Application
- HIV Testing
- HPV Testing
- Syphilis Testing
- Gonorrhea Testing
- Chlamydia Testing
By End-User
- Hospitals
- Clinics
- Diagnostic Labs
- Research Institutes
- Homecare
By Technology
- PCR
- Immunoassay
- Nucleic Acid Testing
Frequently Asked Questions
Find quick answers to common questions.
The Asia Pacific STD Testing Devices Market size is USD 4.93 Billion in 2033.
Key segments for the Asia Pacific STD Testing Devices Market are By Type (Rapid Test Kits, Molecular Diagnostics Devices, Laboratory Equipment, Others); By Application (HIV Testing, HPV Testing, Syphilis Testing, Gonorrhea Testing, Chlamydia Testing, Others); By End-User (Hospitals, Clinics, Diagnostic Labs, Research Institutes, Homecare, Others); By Technology (PCR, Immunoassay, Nucleic Acid Testing, Others).
Major Asia Pacific STD Testing Devices Market players are Abbott, Roche, Thermo Fisher, Siemens Healthineers, BD, Bio-Rad, Hologic, Qiagen, Danaher, PerkinElmer, Agilent, Sysmex, Cepheid, bioMérieux, Fujirebio.
The Asia Pacific STD Testing Devices Market size is USD 2.46 Billion in 2025.
The Asia Pacific STD Testing Devices Market CAGR is 9.10% from 2026 to 2033.
- Abbott
- Roche
- Thermo Fisher
- Siemens Healthineers
- BD
- Bio-Rad
- Hologic
- Qiagen
- Danaher
- PerkinElmer
- Agilent
- Sysmex
- Cepheid
- bioMérieux
- Fujirebio
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









