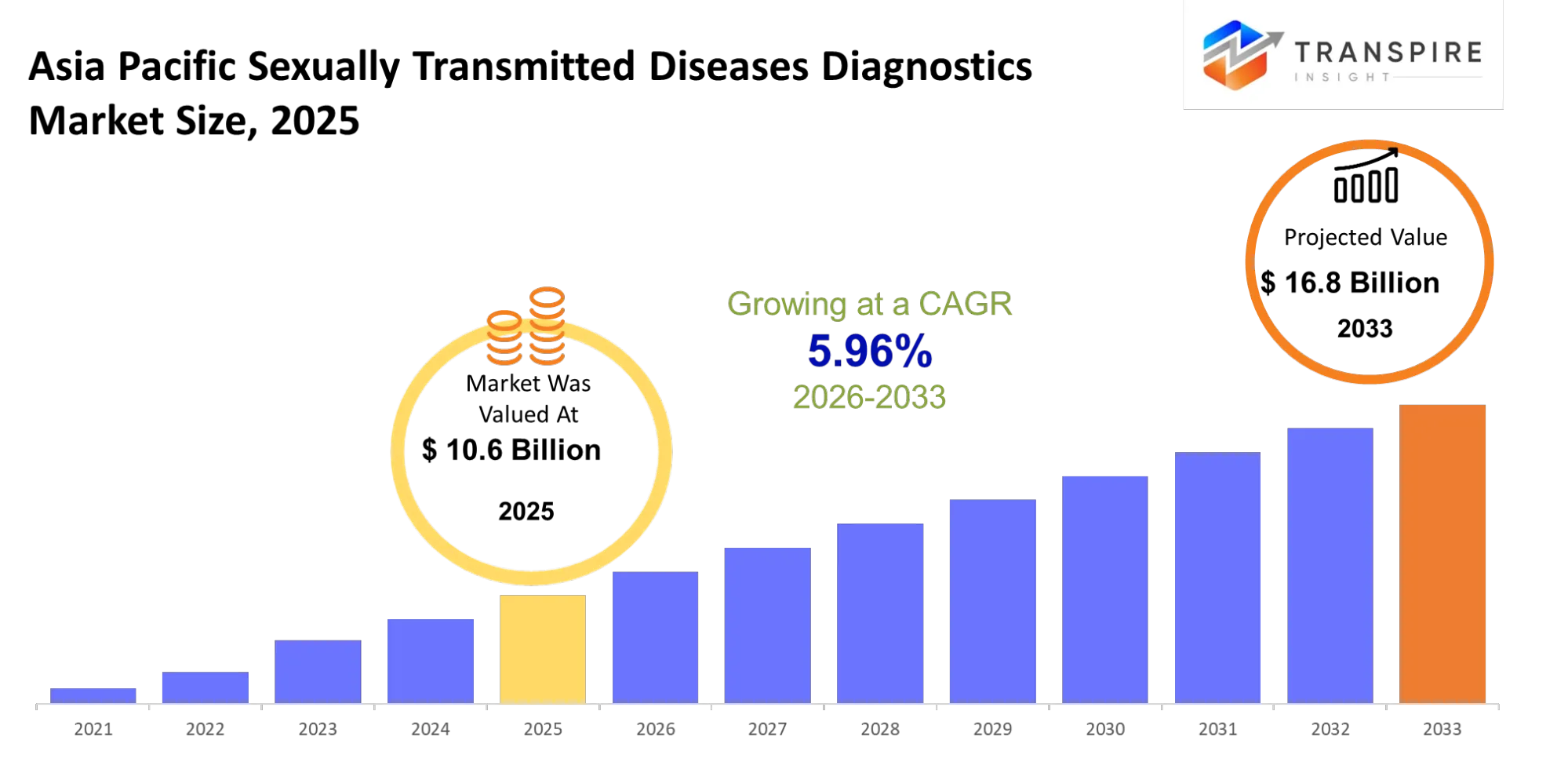
Asia Pacific Sexually Transmitted Diseases Diagnostics Market Size & Forecast:
- Asia Pacific Sexually Transmitted Diseases Diagnostics Market Size 2025: USD 10.6 Billion
- Asia Pacific Sexually Transmitted Diseases Diagnostics Market Size 2033: USD 16.8 Billion
- Asia Pacific Sexually Transmitted Diseases Diagnostics Market CAGR: 5.96%
- Asia Pacific Sexually Transmitted Diseases Diagnostics Market Segments: By Type (Laboratory Testing, Rapid Diagnostic Tests, Molecular Diagnostics, Others); By Application (HIV, HPV, Syphilis, Gonorrhea, Chlamydia, Others); By End-User (Hospitals, Clinics, Diagnostic Labs, Research Institutes, Homecare, Others); By Technology (PCR, Immunoassays, Nucleic Acid Testing, Others)
To learn more about this report, Download Free Sample Report
Asia Pacific Sexually Transmitted Diseases Diagnostics Market Summary
The Asia Pacific Sexually Transmitted Diseases Diagnostics Market was valued at USD 10.6 Billion in 2025. It is forecast to reach USD 16.8 Billion by 2033. That is a CAGR of 5.96% over the period.
The Asia Pacific sexually transmitted disease diagnostics market enables laboratories and hospitals and public health programs to detect infections through its testing methods which include laboratory testing platforms and rapid point-of-care kits and molecular diagnostic systems. The system provides its main function to support healthcare facilities which need to identify infections and control disease spread and make urgent treatment choices in crowded urban areas that need effective disease monitoring.
The market has experienced a fundamental transformation during the past five years because testing methods have shifted from traditional laboratory-based tests to decentralized testing which includes both molecular and rapid antigen test methods that enable quick results outside of centralized testing centers. The COVID-19 pandemic created an urgent need for enhanced testing facilities which led to the widespread adoption of molecular testing and the establishment of routine infectious disease screening procedures throughout healthcare systems.
The government-led sexual health initiatives in China and India and Australia have made sexual health testing more accessible to the public. The new diagnostic automation systems which medical providers use to analyze patient samples are generating continuous revenue through their testing materials because medical facilities choose to implement faster testing systems that can serve larger patient groups.
Key Market Insights
- China controls the Asia Pacific Sexually Transmitted Diseases Diagnostics Market because it will reach a 32% market share by 2025 through its large-scale screening programs.
- The public testing facilities which India has established will help the country achieve its status as the most rapidly developing regional market which will grow at more than 9% each year until 2030.
- Southeast Asian countries experience major market growth because their healthcare systems advance through better laboratory networks and easier access to diagnostic services.
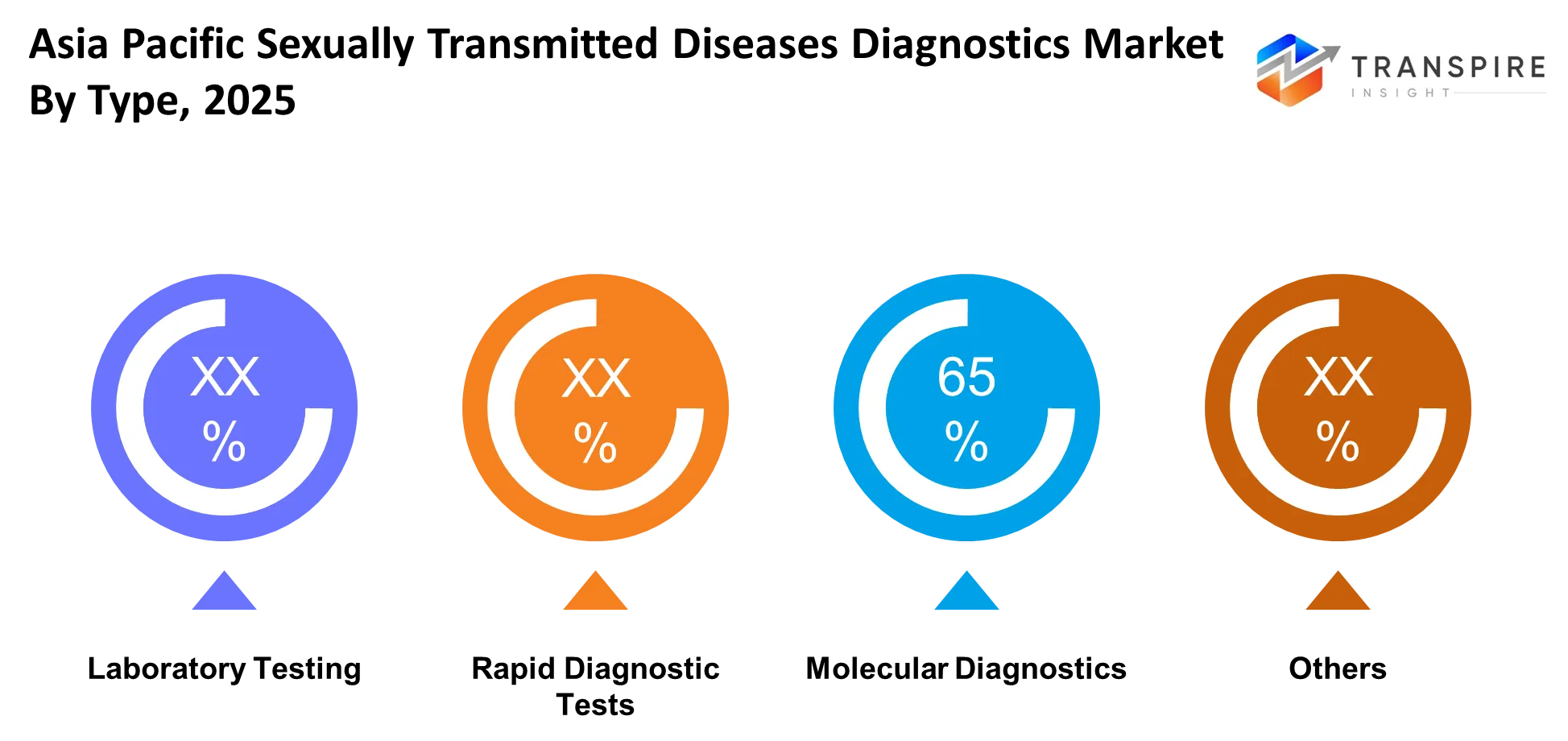
- The market for molecular diagnostics will reach a 42% share in 2025 because these tests offer better sensitivity and faster results than other diagnostic methods.
- The immunoassay-based diagnostics product range maintains its position as the second largest market segment because mid-tier clinical laboratories choose this testing option for its cost-effectiveness and common use.
- The decentralized testing market drives rapid point-of-care testing to become the fastest-growing market segment until 2030.
- The demand for sexual transmitted disease testing in the Asia Pacific region is primarily fulfilled through hospital-based testing which represents 46 percent of market needs.
- The application of sexual health screening for preventive purposes has become the most rapidly expanding sector which receives support from both educational programs aimed at young people and workplace health programs.
- The Asia Pacific Sexually Transmitted Diseases Diagnostics Market derives 54 percent of its revenue from hospitals and diagnostic laboratories.
- Specialty sexual health clinics are emerging as the fastest-growing end-user category through forecast years.
- Companies achieve market advantage through their use of AI-based assay development and multiplex molecular testing technologies.
What are the Key Drivers, Restraints, and Opportunities in the Asia Pacific Sexually Transmitted Diseases Diagnostics Market?
The number one factor driving growth for the Asia Pacific sexually transmitted disease testing market stems from the fast development of molecular testing facilities which emerged after the COVID-19 outbreak. The governments of China, India, South Korea, and Australia invested substantial funds to establish PCR laboratories and automated testing systems and to develop their diagnostic staff between 2020 and 2023. Healthcare systems started using established infrastructure for STI testing after its development which resulted in cost reductions for advanced testing procedures. The testing process now generates ongoing income through sales of reagents and consumables and through service agreements that support the automated testing systems.
The main obstacle that exists today for healthcare access in Asia Pacific countries arises from unequal medical services that affect remote areas and impoverished communities. Nucleic acid amplification tests become accessible for quick implementation at metropolitan medical facilities but peripheral clinics need basic laboratory testing equipment and refrigerator systems and skilled personnel for advanced STI testing. The existing restriction requires time to overcome because it requires sustained investment in public health facilities and development of healthcare personnel and regional medical facilities. Testing delays hinder earnings potential while they force many patients to rely on outdated diagnostic tests which provide less precise results.
Portable point-of-care molecular diagnostics stands as the most important growth opportunity. Compact PCR and isothermal amplification platforms are becoming cost-efficient enough for deployment in decentralized clinical settings. India and Southeast Asia present high-potential markets which public-private partnerships should use to enhance community health screening accessibility and drive market expansion.
What Has the Impact of Artificial Intelligence Been on the Asia Pacific Sexually Transmitted Diseases Diagnostics Market?
Artificial intelligence and advanced digital technologies are transforming sexually transmitted infection diagnostics throughout the Asia Pacific region by their capacity to enhance laboratory automation and increase clinical workflow efficiency and improve diagnostic precision. AI-enabled laboratory information systems are currently handling specimen triage and test prioritization and result validation for high-volume molecular testing centers which operate throughout China and Japan and Australia. The systems enable laboratories to process test samples at an increased rate because they decrease the need for manual work while maintaining the ability to process larger test amounts without requiring additional staff.
Machine learning models are now being used to create predictive analytics systems which operate within diagnostic networks. Healthcare providers use algorithm-driven screening tools to identify high-risk patient groups based on demographic, behavioral, and epidemiological datasets, which enables them to develop more focused testing methods. The use of AI-assisted signal interpretation in molecular diagnostics helps PCR amplification curve analysis which improves detection of anomalies that lead to false-negative and inconclusive test results. The implementation of this system has resulted in faster delivery times, more precise reporting, and better treatment start times which benefit residents of urban public health screening programs.
The combination of digital technologies with cloud-based surveillance systems has enhanced regional disease monitoring through its ability to provide current case information and predict possible outbreak events. The capabilities enhance public health response planning while enabling healthcare systems to distribute their available resources more efficiently across their extensive operational areas.
Key Market Trends
- Since 2021, more than 60 percent of newly installed molecular diagnostic systems in major urban hospitals have been used for testing multiple sexual transmitted infections.
- The Chinese and Indian and Australian governments changed their funding priorities after 2022 from single STI programs to comprehensive infectious disease testing programs.
- Diagnostic buyers have switched from batch laboratory testing to same-day molecular platforms which cut result processing time from 48 hours to less than six hours.
- The 2023 market expansion of Roche and Abbott Laboratories to their multiplex assay portfolios occurred to meet the need for faster clinical decision-making.
- The adoption of point-of-care molecular testing in Southeast Asia increased after 2022 because rural clinics preferred portable testing systems instead of centralized referral systems.
- The hospitals now prefer reagent rental contracts over purchasing complete analyzer systems because the procurement process has changed since 2024.
- Digital reporting platforms have replaced manual STI surveillance systems in several regional public health networks, which has resulted in a 30 percent improvement for case tracking efficiency.
- After 2023, Japanese and South Korean regulatory bodies established stricter validation requirements, which compelled smaller companies to form business alliances with international diagnostic companies.
- The business model now generates continuous income through bundled software and consumables and services instead of relying on single product sales.
Asia Pacific Sexually Transmitted Diseases Diagnostics Market Segmentation
By Type:
The Asia Pacific sexually transmitted diseases diagnostics market depends on laboratory testing, which remains the most popular testing method, because healthcare facilities require precise sample testing to achieve accurate disease diagnosis. The segment achieves its vital role in urban healthcare systems because it delivers dependable results together with extensive testing options. The ongoing growth of testing knowledge together with improvements in laboratory facilities leads to continuous testing activity across multiple regional markets.
The demand for rapid diagnostic tests is increasing because faster test results enable medical professionals to make treatment choices sooner. The tests serve as essential tools in outreach initiatives, clinic operations, and screening events that need to deliver instant results. Molecular diagnostics remain the dominant method because of their ability to deliver higher sensitivity together with precise pathogen identification, while the others segment includes culture-based methods and emerging diagnostic tools that support broader testing requirements.
To learn more about this report, Download Free Sample Report
By Application:
HIV diagnostics hold a major share of the market because screening programs, public health campaigns, and early detection efforts remain central across Asia Pacific healthcare systems. The healthcare system sees an upward trend in testing demand because healthcare providers now prioritize regular screening methods and improved treatment options. Public and private healthcare facilities use continuous monitoring to maintain effective disease control throughout their operations.
The testing of HPV syphilis gonorrhea and chlamydia shows substantial growth because of rising public knowledge and the development of preventive health programs. The implementation of cervical cancer prevention programs enables HPV testing to grow, while sexual health programs develop wider access to syphilis and gonorrhea testing. The other category includes less common sexually transmitted infections that still require diagnostic attention in specialized healthcare settings.
By End-User:
The hospital sector serves as a primary customer base because its medical staff uses advanced diagnostic tools to perform precise tests and deliver complete patient monitoring. Hospitals serve as the main facilities for sexually transmitted disease diagnosis because they handle large patient numbers and allow access to specialized medical expertise throughout multiple Asian Pacific nations. The market position of this segment receives additional support from strong institutional backing.
The growth of clinics and diagnostic laboratories occurs because they provide patients with better access points to receive their medical needs through improved speed of service. The research institutes support development by creating new testing methods which they test through their clinical validation process. The homecare sector experiences growth through rising demand for self-testing kits which provide users with private and convenient testing options. The other category includes community health centers and public health screening units serving regional populations.
By Technology:
People value PCR technology because it delivers accurate results and detects infections that are present during the early stages of an illness. Advanced healthcare facilities use PCR testing as their main method for confirmatory diagnosis. The increasing investment in molecular laboratories throughout Asia Pacific is driving better disease detection methods which healthcare facilities consider essential for their operations.
Immunoassays maintain their critical role in the market because these methods enable organizations to conduct extensive testing while delivering reliable results. Nucleic acid testing usage is expanding because it provides high detection capabilities and benefits from improved processing speeds. Testing platforms continue to advance which enables healthcare providers to achieve better diagnostic results while implementing regional disease surveillance initiatives.
What are the Key Use Cases Driving the Asia Pacific Sexually Transmitted Diseases Diagnostics Market?
The main application of sexually transmitted diseases diagnostic testing in hospitals throughout the Asia Pacific region centers around molecular tests which detect chlamydia and gonorrhea and syphilis infections. The highest testing volume at large tertiary hospitals occurs because these facilities need to achieve fast and precise test results which enable them to make treatment decisions and control infections and conduct antenatal screening.
The development of new application methods is taking place at specialized sexual health clinics and diagnostic laboratory chains which now offer preventive screening and standard reproductive health evaluations. Universities and their affiliated health centers together with corporate healthcare programs, are implementing rapid testing solutions to enhance their early intervention programs.
New testing applications involve at-home testing through self-collection methods which use digital reporting systems, and mobile diagnostic units that serve remote rural communities. Public health organizations in India and Southeast Asia are investigating decentralized testing methods to enhance accessibility while decreasing their need for centralized laboratory systems.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 10.6 Billion |
|
Market size value in 2026 |
USD 11.2 Billion |
|
Revenue forecast in 2033 |
USD 16.8 Billion |
|
Growth rate |
CAGR of 5.96% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Asia Pacific (China, India, Japan, South Korea, Australia, Rest of Asia Pacific) |
|
Key company profiled |
Abbott, Roche, Thermo Fisher, Siemens Healthineers, BD, Bio-Rad, Hologic, Qiagen, Danaher, PerkinElmer, Agilent, Sysmex, Cepheid, bioMérieux, Fujirebio |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Laboratory Testing, Rapid Diagnostic Tests, Molecular Diagnostics, Others); By Application (HIV, HPV, Syphilis, Gonorrhea, Chlamydia, Others); By End-User (Hospitals, Clinics, Diagnostic Labs, Research Institutes, Homecare, Others); By Technology (PCR, Immunoassays, Nucleic Acid Testing, Others) |
Which Regions are Driving the Asia Pacific Sexually Transmitted Diseases Diagnostics Market Growth?
East Asia holds the top position in the Asia Pacific market for sexually transmitted disease testing, which China, Japan, and South Korea lead as the main testing countries. The region shows this dominance because its healthcare system operates at a mature stage and provides strong reimbursement policies and the government implements disease control systems which mandate regular STI testing. The governments of these nations have increased their investment in molecular diagnostics since 2020, which led to the implementation of advanced testing technology in hospitals and public laboratories throughout the country. The domestic manufacturers of testing equipment together with automated laboratory systems and clear regulatory procedures create a strong foundation for the region to maintain its dominance.
Australia and New Zealand lead Oceania while the region provides market stability through its public health policies and its healthcare spending patterns, which health systems predictably follow. The region uses screening through regular tests and the combination of digital health systems with high compliance to clinical standards, which create its operational framework, while East Asia handles its operations through its capacity-based approach. National sexual health programs support regular testing across hospitals, community clinics, and preventive care networks, creating steady diagnostic demand. Oceania functions as a stable revenue source for global diagnostics companies, which aim to expand into regions with lower risk.
The region of South and Southeast Asia constitutes the most rapidly expanding market because healthcare accessibility improvements together with decentralized diagnostic services drive its development. The recent public investment in laboratory modernization projects throughout India, Indonesia, Vietnam and Thailand has led to increased adoption of molecular testing methods. The regional governments have established more extensive reproductive health screening programs which now include sexually transmitted infection detection as a fundamental service. The period from 2026 to 2033 provides market entrants and investors with substantial growth opportunities because underserved populations will gain access to improved diagnostic technologies and services.
Who are the Key Players in the Asia Pacific Sexually Transmitted Diseases Diagnostics Market and How Do They Compete?
The Asia Pacific sexually transmitted diseases diagnostics market shows moderate competition because international diagnostics companies control advanced molecular testing systems while local businesses use regional pricing and distribution capabilities to compete. The main competitive factor has changed from instrument placement to three evaluation criteria which include assay accuracy and workflow automation and long-term reagent service agreements. The existing market players protect their market position by developing multiplex testing systems and adding digital reporting solutions while new companies focus on providing portable affordable products for remote testing needs.
Roche uses its cobas high-throughput molecular platforms to enable laboratories to handle large testing volumes through their highly efficient automated systems. The company generates its main benefits through complete integration of its hardware, software, and consumables which creates ongoing revenue streams. The company has expanded its operations throughout the Asia Pacific region by establishing partnerships with hospitals and public laboratories to conduct infectious disease research.
Abbott Laboratories uses portable molecular systems to create a unique testing solution which allows mid-sized clinics to conduct tests at their locations. The flexibility of Abbott products enables the company to establish a strong presence in developing markets throughout India and Southeast Asia. Hologic develops its examination equipment to deliver accurate results through its shared testing panels, which target high-end medical testing facilities. Qiagen builds its regional presence through distribution partnerships and its sample-to-result systems, which help labs operate more effectively while they handle their reduced diagnostic testing timelines.
Company List
- Abbott
- Roche
- Thermo Fisher
- Siemens Healthineers
- BD
- Bio-Rad
- Hologic
- Qiagen
- Danaher
- PerkinElmer
- Agilent
- Sysmex
- Cepheid
- bioMérieux
- Fujirebio
Recent Development News
In December 2025, Becton, Dickinson and Company entered a partnership with Certest Biotec to expand the BD MAX™ sexually transmitted infection diagnostics menu through IVDR-certified VIASURE assays. The certification broadened automated STI molecular testing capabilities for laboratories across Europe and Asia Pacific-adjacent regulated markets, strengthening BD’s regional infectious disease diagnostics footprint. Source https://www.prnewswire.com/
In July 2025, GeneProof launched a new line of STI PCR assays on its myCROBE™ platform as part of milestones announced by NuvinkaDx. The launch expanded automated multiplex detection of key sexually transmitted pathogens and improved access to scalable molecular STI testing solutions for international laboratories, including Asia Pacific distributors.
Source https://www.prnewswire.com/
What Strategic Insights Define the Future of the Asia Pacific Sexually Transmitted Diseases Diagnostics Market?
The Asia Pacific sexually transmitted diseases diagnostic market will transition to decentralized testing systems which use digital technology to deliver precise test results at medical facilities during the next five to seven years. The healthcare system digitization together with new guidelines for tracking infectious diseases and the need to shorten diagnostic times in crowded urban and semi-urban areas drive this development. Testing will increasingly shift closer to community care settings rather than remaining concentrated in large reference laboratories.
The market concentration problem creates hidden dangers because it limits access to molecular platform technology which only a few suppliers control. The operational capacity of healthcare networks will suffer if reagent prices increase or supply chain problems disrupt cartridge distribution because patient screening needs continue to grow. The need for this product will restrict price access to markets which depend on affordable solutions.
AI-assisted self-collection diagnostic pathways present new business opportunities for testing facilities in India and Southeast Asia which are experiencing rapid growth in telehealth services. The approval of remote diagnostic testing validation by regulatory bodies will establish a new approach for testing patients. The market should start developing digital platforms that support interoperability together with establishing regional manufacturing agreements because these two elements will create market advantages during the upcoming expansion period.
Asia Pacific Sexually Transmitted Diseases Diagnostics Market Report Segmentation
By Type
- Laboratory Testing
- Rapid Diagnostic Tests
- Molecular Diagnostics
By Application
- HIV
- HPV
- Syphilis
- Gonorrhea
- Chlamydia
By End-User
- Hospitals
- Clinics
- Diagnostic Labs
- Research Institutes
- Homecare
By Technology
- PCR
- Immunoassays
- Nucleic Acid Testing
Frequently Asked Questions
Find quick answers to common questions.
The Asia Pacific Sexually Transmitted Diseases Diagnostics Market size is USD 16.8 Billion in 2033.
Key segments for the Asia Pacific Sexually Transmitted Diseases Diagnostics Market are By Type (Laboratory Testing, Rapid Diagnostic Tests, Molecular Diagnostics, Others); By Application (HIV, HPV, Syphilis, Gonorrhea, Chlamydia, Others); By End-User (Hospitals, Clinics, Diagnostic Labs, Research Institutes, Homecare, Others); By Technology (PCR, Immunoassays, Nucleic Acid Testing, Others).
Major Asia Pacific Sexually Transmitted Diseases Diagnostics Market players are Abbott, Roche, Thermo Fisher, Siemens Healthineers, BD, Bio-Rad, Hologic, Qiagen, Danaher, PerkinElmer, Agilent, Sysmex, Cepheid, bioMérieux, Fujirebio.
The Asia Pacific Sexually Transmitted Diseases Diagnostics Market size is USD 10.6 Billion in 2025.
The Asia Pacific Sexually Transmitted Diseases Diagnostics Market CAGR is 5.96% from 2026 to 2033.
- Abbott
- Roche
- Thermo Fisher
- Siemens Healthineers
- BD
- Bio-Rad
- Hologic
- Qiagen
- Danaher
- PerkinElmer
- Agilent
- Sysmex
- Cepheid
- bioMérieux
- Fujirebio
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)