United Kingdom Pharmacovigilance Market Size & Forecast:
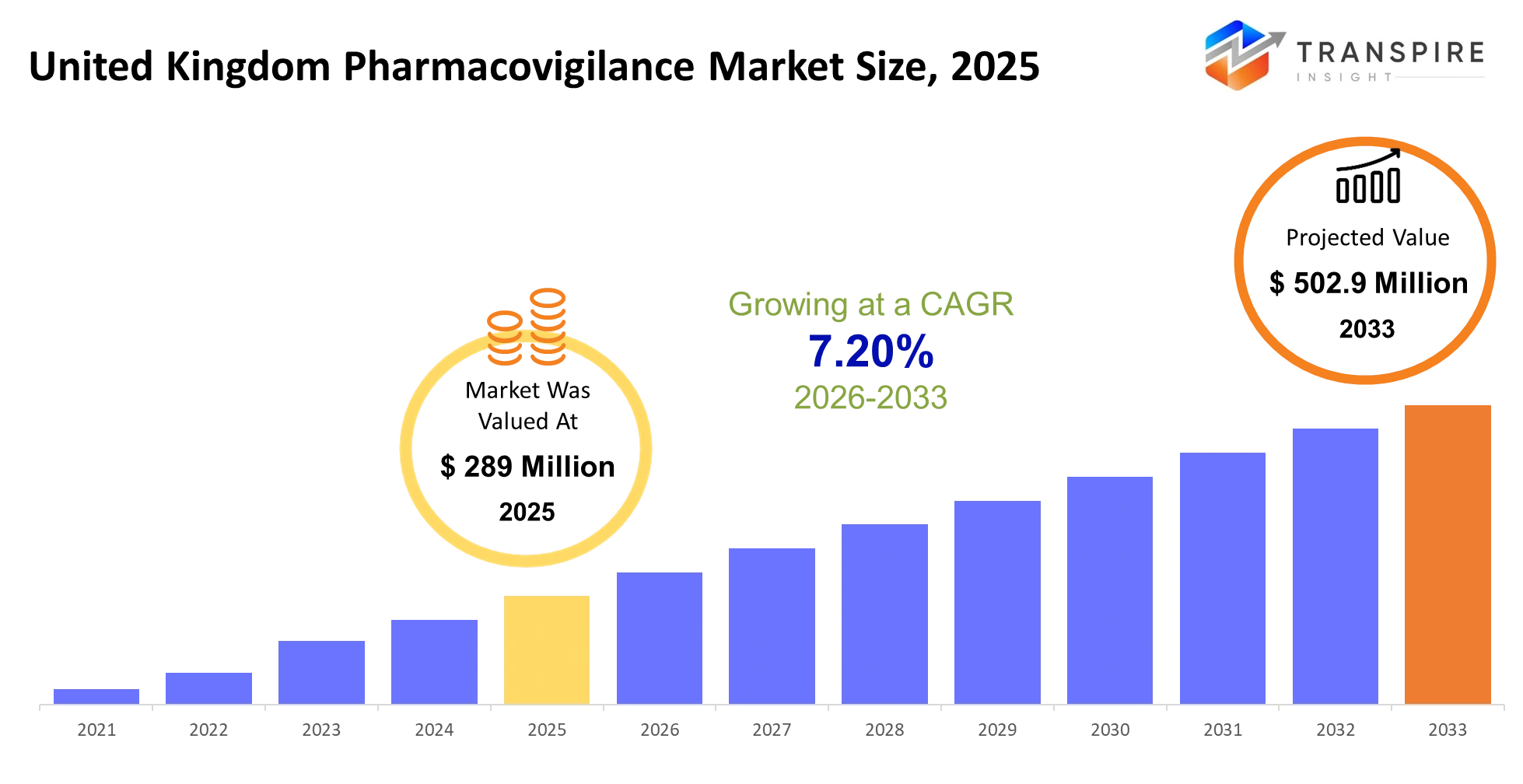
- United Kingdom Pharmacovigilance Market Size 2025: USD 289 Million
- United Kingdom Pharmacovigilance Market Size 2033: USD 502.9 Million
- United Kingdom Pharmacovigilance Market CAGR: 7.20%
- United Kingdom Pharmacovigilance Market Segments: By Service Provider (In-House, Contract Outsourcing), By Service Type (Adverse Event Reporting, Risk Management, Signal Detection, Medical Writing), By End-User (Pharmaceutical Companies, Biotechnology Companies, CROs).
To learn more about this report, Download Free Sample Report
United Kingdom Pharmacovigilance Market Summary:
The United Kingdom Pharmacovigilance Market size is estimated at USD 289 Million in 2025 and is anticipated to reach USD 502.9 Million by 2033, growing at a CAGR of 7.20% from 2026 to 2033.The United Kingdom's pharmacovigilance market, as a part of the life sciences and healthcare industry, is based on a system of drug safety, post-marketing surveillance, and risk management of patients. The United Kingdom's pharmacovigilance market is expected to be influenced by more stringent reporting requirements from authorities like the MHRA, as well as increased awareness of patients regarding adverse reactions of drugs. Healthcare and pharmaceutical companies are expected to adopt more proactive approaches towards tracking adverse reactions, as opposed to reactive approaches. Patients are expected to ask for more transparency, and data integration from both clinical and non-clinical sources is expected to redefine the way safety information is generated.
Key Market Trends & Insights:
Digital transformation of safety monitoring: The United Kingdom Pharmacovigilance Market is clearly embracing digital technologies, which will enhance adverse event monitoring and reporting. The workload will be minimized through automated case processing systems, cloud-based databases, and enhanced analytics. Healthcare organizations will increasingly depend on digital platforms to ensure faster identification of potential risks.
Regulatory tightening and compliance pressure: The United Kingdom Pharmacovigilance Market will be influenced by the strengthening of expectations from national authorities. The updated standards of operation will include changes to reporting timelines, audit procedures, and documentation. The environment will be suitable for pharmaceutical companies to focus on compliance systems. The focus will be on the implementation of structured procedures and professionals to ensure accuracy and consistency.
Increasing role of real-world evidence: The United Kingdom Pharmacovigilance Market will increasingly focus on real-world evidence. The focus will be on the information obtained from patient registries, electronic health records, and wearables. The focus on patient information will enhance the assessment of the impact of drugs. The environment will be suitable for making informed decisions and improving the overall risk assessment.
Increase in demand for outsourcing services: Many companies would opt for outsourcing services for their pharmacovigilance services. Cost management, access to expert professionals, and business flexibility are expected to increase. Contract research organizations would improve their services, providing overall services for both small biotechnology companies and large pharmaceutical companies.
Patient-centric reporting systems: The expectations of patients would also play a role in influencing reporting systems. Reporting systems would be more accessible, and patients would be encouraged through mobile apps and other tools. This would lead to more awareness, and patients would actively participate in reporting adverse events.
United Kingdom Pharmacovigilance Market Segmentation
By Service Provider
- In-House: The United Kingdom Pharmacovigilance Market will maintain its high dependence on in-house teams of pharmaceutical companies for pharmacovigilance and safety reporting activities. Organizations will want to maintain control over their critical data to ensure adherence to national guidelines and quality parameters. In-house systems will start to use automation tools to manage increased cases.
- Contract Outsourcing: Contract service providers will gain increased acceptance as organizations seek alternative and cost-effective options. Contract research organizations will be used for case processing, signal detection, and regulatory activities. This model will help pharmaceutical companies to grow their operations easily while leveraging expertise during product launch and peak reporting periods.

To learn more about this report, Download Free Sample Report
By Service Type
- Adverse Event Reporting: The United Kingdom Pharmacovigilance Market will also focus on the accurate reporting of adverse drug reactions. The healthcare sector will use digital technology to report adverse reactions, increasing the speed of reporting and eliminating the chances of errors that might occur during the reporting process. The increase in awareness among patients will also result in the reporting of adverse reactions, thereby increasing the scope of data collection.
- Risk Management: A well-planned risk management strategy will be the key to maintaining drug safety during the entire drug life cycle. The companies will be focused on identifying the risks that might occur during the drug development process, thereby implementing the necessary steps to manage the risks.
- Signal Detection: Advanced analytics and artificial intelligence will be used to a greater degree to aid in the detection of safety signals from large sets of data. This will help in the automation of detecting potential risks by recognizing patterns and trends. This process will aid in efficient decision-making and improve the efficiency of pharmacovigilance activities.
- Medical Writing: Accurate documentation will still be an integral part of regulatory activities. Medical writing services will be required for safety reports and regulatory activities. This will ensure uniformity in language and format for regulatory compliance and effective communication with regulatory bodies.
By End-User
- Pharmaceutical Companies: The United Kingdom Pharmacovigilance Market for pharmaceutical companies will witness a continued focus on drug safety activities with their internal teams handling a large amount of safety data being generated for marketed products.
- Biotechnology Companies: Biotechnology companies are likely to become more reliant on flexible pharmacovigilance models due to their relatively smaller scale of operation. With fewer internal resources available for drug safety activities, there will be a focus on partnerships with external experts.
- CROs: With the growing demand for pharmacovigilance services, contract research organizations are also likely to become more prominent in the United Kingdom Pharmacovigilance Market. They are likely to provide specialized pharmacovigilance services to their clients for case processing, signal detection, and regulatory submissions. This is because many organizations are looking for efficient solutions that can handle fluctuating workloads without investing in permanent infrastructure.
Country Insights
In the northern part of the country, the activity will be driven by the strong academic links and large healthcare infrastructure. Cities such as Manchester, Leeds, and Newcastle will provide an environment for close working between hospitals, universities, and the pharmaceutical industry. This will further encourage the use of shared data systems and research-based safety practices. The use of digital tools for reporting will also increase, helping healthcare practitioners identify ADRs more effectively and submit them more accurately.
In the southern part of the country, the activity will be driven by the large number of pharmaceutical headquarters and interactions with the regulators. This will be particularly the case around the London area. Companies in this region will heavily invest in the latest technologies, including the use of AI for the detection of signals and automated case handling. This region will also witness a greater emphasis on the use of third-party services for the handling of pharmacovigilance activity, helping the organization effectively handle the increasing volume of data.
Recent Development News
In March 2026, Indegene Limited announced the acquisition of UK-based DT Associates Research and Consulting Services Limited and Trilogy Writing and Consulting Limited for approximately £2.70 million, strengthening its pharmacovigilance, regulatory, and medical services capabilities in the UK life sciences sector.
In April 2026, Lupin Limited announced the completion of its acquisition of VISUfarma, expanding its European specialty pharmaceuticals portfolio and enhancing its pharmacovigilance and regulatory footprint across key markets including the UK.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 289 Million |
|
Market size value in 2026 |
USD 309.2 Million |
|
Revenue forecast in 2033 |
USD 502.9 Million |
|
Growth rate |
CAGR of 7.20% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
United Kingdom |
|
Key company profiled |
IQVIA Holdings Inc., Accenture plc, Cognizant Technology Solutions Corp., Parexel International Corporation, ICON plc, Labcorp Drug Development, Wipro Limited, Capgemini SE, ArisGlobal LLC, TCS (Tata Consultancy Services), IBM Corporation, Bioclinica Inc., Ennov Solutions Inc., Oracle Corporation, Quanticate Ltd. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Service Provider (In-House, Contract Outsourcing), By Service Type (Adverse Event Reporting, Risk Management, Signal Detection, Medical Writing), By End-User (Pharmaceutical Companies, Biotechnology Companies, CROs). |
Key United Kingdom Pharmacovigilance Market Company Insights
Some of the key activities that will be performed by the company in the united kingdom pharmacovigilance market will include the presence of established pharmaceutical companies, which will continue to improve their internal systems for handling safety issues. However, they will also look for external partners to help them manage the increased data that is being generated, which will include investments in automated systems, real-time monitoring, and the inclusion of artificial intelligence for greater accuracy in identifying potential risks. The overall aim will also be to ensure that the operations of the company align with the updated guidelines issued by the national government.
Service providers and contract research organizations will also become a larger part of the united kingdom pharmacovigilance market, as they will provide end-to-end services for pharmacovigilance. This will include providing services for global pharmaceutical companies, as well as smaller biotechnology companies that need flexible and cost-effective services. This will include expansion through digital platforms, recruitment of skilled employees, and strategic partnerships, which will ensure that the overall safety process is enhanced through the inclusion of different types of organizations.
Company List
- IQVIA Holdings Inc.
- Accenture plc
- Cognizant Technology Solutions Corp.
- Parexel International Corporation
- ICON plc
- Labcorp Drug Development
- Wipro Limited
- Capgemini SE
- ArisGlobal LLC
- TCS(Tata Consultancy Services)
- IBM Corporation
- Bioclinica Inc.
- Ennov Solutions Inc.
- Oracle Corporation
- Quanticate Ltd
United Kingdom Pharmacovigilance Market Report Segmentation
By Service Provider
- In-House
- Contract Outsourcing
By Service Type
- Adverse Event Reporting
- Risk Management
- Signal Detection
- Medical Writing
By End-User
- Pharmaceutical Companies
- Biotechnology Companies
- CROs
Frequently Asked Questions
Find quick answers to common questions.
The approximate United Kingdom Pharmacovigilance Market size for the market will be USD 502.9 Million in 2033.
Key segments for the United Kingdom Pharmacovigilance Market By Service Provider (In-House, Contract Outsourcing), By Service Type (Adverse Event Reporting, Risk Management, Signal Detection, Medical Writing), By End-User (Pharmaceutical Companies, Biotechnology Companies, CROs).
Major United Kingdom Pharmacovigilance Market players are IQVIA Holdings Inc., Accenture plc, Cognizant Technology Solutions Corp., Parexel International Corporation, ICON plc, Labcorp Drug Development, Wipro Limited, Capgemini SE, ArisGlobal LLC, TCS (Tata Consultancy Services), IBM Corporation, Bioclinica Inc., Ennov Solutions Inc., Oracle Corporation, Quanticate Ltd.
The United Kingdom Pharmacovigilance Market size is USD 289 Million in 2025.
The United Kingdom Pharmacovigilance Market CAGR is 7.20%.
- IQVIA Holdings Inc.
- Accenture plc
- Cognizant Technology Solutions Corp.
- Parexel International Corporation
- ICON plc
- Labcorp Drug Development
- Wipro Limited
- Capgemini SE
- ArisGlobal LLC
- TCS(Tata Consultancy Services)
- IBM Corporation
- Bioclinica Inc.
- Ennov Solutions Inc.
- Oracle Corporation
- Quanticate Ltd
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
May 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









