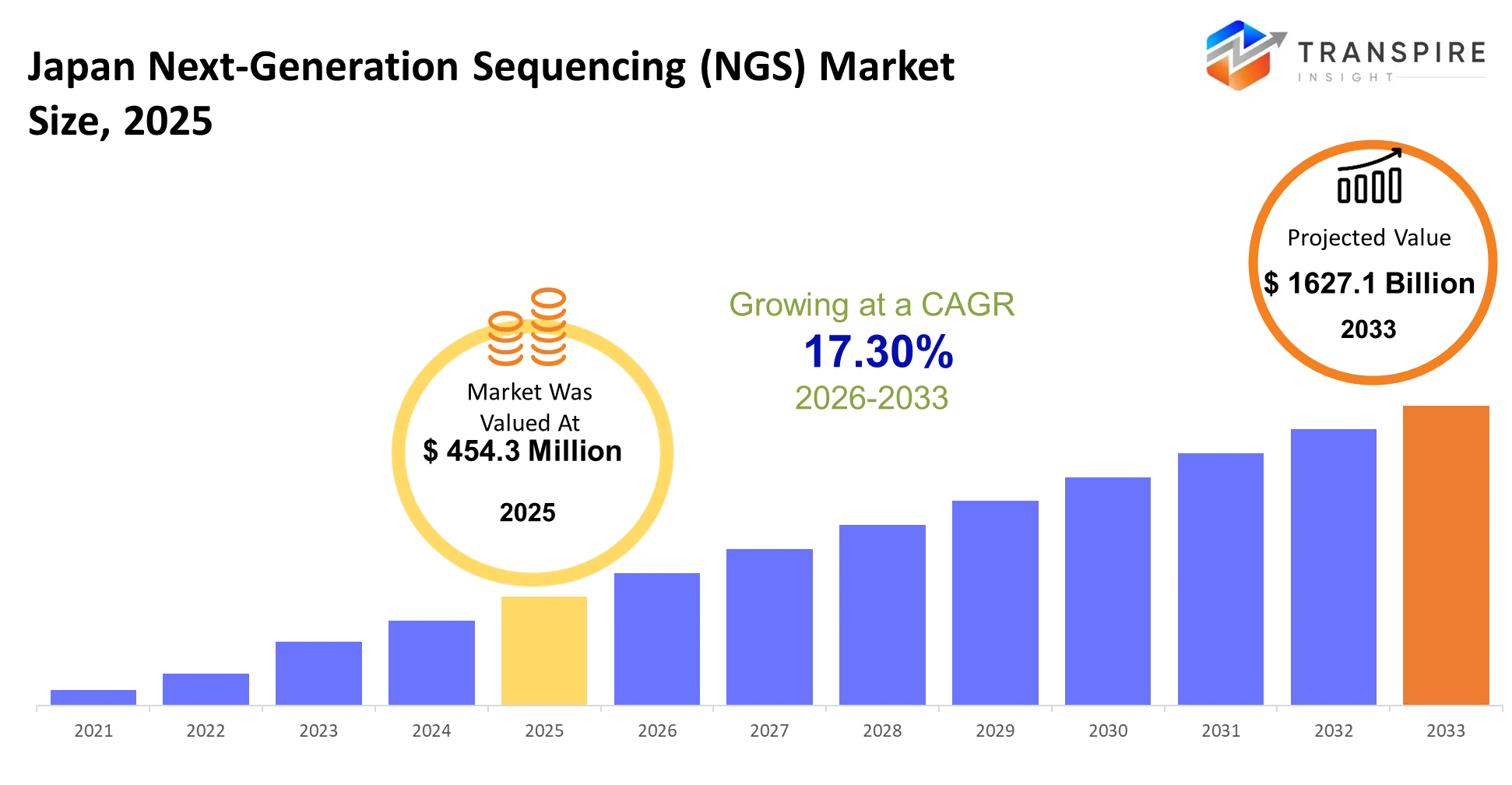
Japan Next-Generation Sequencing (NGS) Market Size & Forecast:
- Japan Next-Generation Sequencing (NGS) Market Size 2025: USD 454.3 Million
- Japan Next-Generation Sequencing (NGS) Market Size 2033: USD 1627.1 Million
- Japan Next-Generation Sequencing (NGS) Market CAGR: 17.30%
- Japan Next-Generation Sequencing (NGS) Market Segments: By Type (Sequencing Platforms, Reagents & Kits, Services, Software, Others); By Application (Genomics, Oncology, Clinical Diagnostics, Drug Discovery, Agriculture, Others); By End-User (Hospitals, Research Institutes, Pharma Companies, Biotech Firms, Academic Institutions, Others); By Technology (Whole Genome Sequencing, Targeted Sequencing, RNA Sequencing, Others)
To learn more about this report, Download Free Sample Report
Japan Next-Generation Sequencing (NGS) Market Summary
The Japan Next-Generation Sequencing (NGS) Market was valued at USD 454.3 Million in 2025. It is forecast to reach USD 1627.1 Million by 2033. That is a CAGR of 17.30% over the period.
The Japan next-generation sequencing (NGS) market enables hospitals, research institutes, and biopharmaceutical companies to decode genetic information at scale, which enables them to detect diseases more quickly and plan precise oncology treatments and diagnose rare diseases and develop drugs through biomarker research. The system helps to resolve a core healthcare problem because it decreases both expenses and time requirements for producing genomic data that medical practitioners and researchers need to make decisions.
The market has transformed during the past three to five years because hospitals now use sequencing technology for their daily operations instead of using it only for research purposes especially in cancer treatment and companion diagnostic tests. The structural change started to move faster after Japan began to offer financial support for genomic profiling tests and developed its national precision medicine programs. The COVID-19 pandemic created a fundamental change because it demonstrated how vital extensive sequencing systems are for monitoring pathogens and managing public health emergencies. Healthcare organizations are developing their own sequencing centers while pharmaceutical companies use genomic information to improve their clinical trial plans which has created commercial growth that stems from these developments.
The result of these developments leads to increased use of platforms and more frequent use of consumable products and ongoing service income throughout Japan's molecular diagnostics market.
Key Market Insights
- Kanto controlled the Japan Next-Generation Sequencing (NGS) Market with 43% market share in 2025, because Tokyo-based genomic institutes and advanced healthcare infrastructure enabled its operations.
- The Kansai region will become the fastest-expanding market until 2032, because regenerative medicine research clusters and clinical genomics adoption continue to increase.
- Hokkaido and Kyushu experience new demand growth that exceeds 10% each year, because regional hospitals invest in sequencing technologies.
- Diagnostic laboratories and translational research centers conducted repeat purchases of consumables which created a 49% market share in 2025.
- The market share of sequencing instruments reached approximately 30% which made it the second-largest segment.
- The fastest-growing segment of bioinformatics services will develop through 2032 because the market requires scalable solutions for genomic data interpretation.
- The oncology market reached its peak share of 40% in 2025 because molecular profiling now serves as the standard method for precision cancer therapy pathways.
- The genomics ecosystem in Japan experiences its most rapid growth through infectious disease surveillance and non-invasive prenatal testing applications.
- The Japan Next-Generation Sequencing (NGS) Market generated 52% of its revenue through hospitals and diagnostic laboratories in 2025.
- Biopharmaceutical companies represent the fastest-growing end-user category, which results from biomarker-led clinical trial optimization.
What are the Key Drivers, Restraints, and Opportunities in the Japan Next-Generation Sequencing (NGS) Market?
The primary force accelerating the Japan next-generation sequencing market is the integration of genomic testing into reimbursed clinical care pathways. The shift started to develop after Japan extended its national insurance system to cover complete genomic testing for cancer patients while supporting precision medicine through government-funded genomic research programs. The policy changes created better financial conditions for hospitals and diagnostic centers, which resulted in improved access to genomic testing services that now reach more facilities than just elite research institutions. The platform providers and bioinformatics service vendors have experienced increased testing activities and higher material usage, which generated additional ongoing income for their business operations.
The most important structural problem in Japan exists because of insufficient trained experts who can work as genomic data analysts and clinical bioinformatics specialists. The adoption of sequencing hardware has reached its peak, but organizations still depend on expert scientists who work at select academic institutions to interpret complicated genomic information. The development of clinical-grade genomic interpretation capabilities requires several years of specialized training and compliance with regulatory standards, which makes this issue impossible to solve in a short period. The result has been slower progress toward decentralizing sequencing services, which has postponed the use of these services in regional hospitals, while the existing sequencing systems remain unused because they prevent the market from growing.
The upcoming growth stage currently shows its most evident potential through AI-powered genomic interpretation platforms which serve as its primary growth opportunity. The biomedical innovation clusters of Kansai show fast development of automated variant classification systems together with cloud-based clinical decision support systems. The machine learning-powered diagnostic processes for rare diseases enable hospitals to increase their capacity for sequencing-based tests without the need to establish extensive internal bioinformatics departments.
What Has the Impact of Artificial Intelligence Been on the Japan Next-Generation Sequencing (NGS) Market?
The artificial intelligence and advanced digital technologies operate to transform the next-generation sequencing ecosystem in Japan through their capacity to automate laboratory processes and speed up genomic data analysis. AI-enabled sequencing platforms now automate sample quality assessment, library preparation optimization, and run monitoring, which results in decreased need for manual work in high-throughput laboratories. The clinical genomics centers in Japan use automated workflow orchestration systems to optimize their sequencing pipelines, which results in faster processing times and better instrument usage throughout their diagnostic operations.
Machine learning models are also transforming predictive genomic analysis. The systems process extensive sequencing data to discover clinically significant variants which establish disease connections and determine important biomarkers for both oncology and rare disease testing. AI-driven interpretation platforms enable faster variant classification through their ability to cut classification time by 30 to 40 percent compared to manual review, which benefits clinicians through faster report delivery while decreasing future diagnostic expenses. The advanced bioinformatics algorithms now function to enhance sequencing accuracy because they can detect low-frequency mutations with greater efficiency, which boosts confidence in precision treatment decisions while decreasing the need for repeat tests.
The major problem exists because there are not enough annotated genomic datasets which meet high-quality standards and include diverse genetic data from Japanese people. The data restriction affects model performance because it particularly impacts the interpretation of uncommon genetic variants. The high integration expenses prevent regional hospitals without advanced computational systems from adopting AI-based genomic workflows which operate at major research facilities.
Key Market Trends
- Japanese hospitals began using their sequencing funds for standard cancer tests and medically covered genomic testing in 2022.
- The expansion of national reimbursement policies between 2021 and 2024 led to an increase in the use of comprehensive genomic panels at tertiary healthcare facilities and cancer treatment centers.
- After 2023, Illumina and Thermo Fisher Scientific established more local support partnerships to enhance their clinical laboratory operations in Japan.
- Research centers studying rare diseases adopted long-read sequencing technology after 2023 because they needed better capability to detect structural variants than short-read platforms could offer.
- The implementation of AI-based bioinformatics tools for genomic analysis at laboratories from 2022 to 2025 resulted in a 35 percent decrease in genomic analysis time which led to changes in their equipment purchasing needs towards systems that included software.
- Pharmaceutical companies stopped outsourcing genomic analysis work to establish internal sequencing facilities which they use for biomarker-based cancer research and companion diagnostic testing.
- Laboratories needed to expand their reagent procurement methods beyond conventional international sources because supply chain interruptions during 2021 to 2022.
- After 2024 regional hospitals began using cloud-based genomic analytics which decreased their need to depend on academic centers located in metropolitan areas for expert variant interpretation.
- Organizations now concentrate their competitive efforts on developing automated analysis systems and accurate data processing instead of testing sequencing throughput performance.
Japan Next-Generation Sequencing (NGS) Market Segmentation
By Type:
The Japan Next-Generation Sequencing market depends on sequencing platforms because advanced instruments serve as essential equipment for research and clinical sequencing operations. Laboratories will implement more systems when processing speed and precision and automated technology continue to advance. Healthcare facilities and research institutions will base their investment choices on their requirement for systems that can process extensive sample sizes.
The market maintains stable activity through reagents and kits because these products get used multiple times during sequencing processes. Organizations now require external sequencing services because they need to complete their specialized projects through outsourced sequencing support. The Japan Next-Generation Sequencing market will increase operational efficiency through software solutions which enable complex genomic dataset analysis and interpretation. The market also benefits from sample preparation tools and accessories which provide additional support to software solutions.
By Application:
Sequencing technology supports extensive genetic research and population studies which establishes genomics as one of its largest application fields. Scientists require genomic testing because it provides critical insights into genetic disorders and genetic variation and biological pathways. Japanese research institutions establish sequencing as a permanent research tool which generates ongoing growth in this market sector.
Sequencing technology has established oncology as an essential field because it provides deep insights into tumor biology while enabling doctors to create personalized treatment strategies. The clinical diagnostic field is growing through its implementation in rare disease detection and inherited disorder testing. Genomic data helps scientists discover new drug targets while farmers use sequencing technology to develop better crops and study plant diseases. The Japan Next-Generation Sequencing (NGS) Market continues to expand its practical applications through the development of new applications.

To learn more about this report, Download Free Sample Report
By End-User:
The increasing implementation of sequencing technology in diagnostic procedures and personalized medicine treatment programs has resulted in hospitals becoming more frequent users of this technology. The healthcare system has started to use cutting-edge genomic technologies in order to achieve better patient results through quick and precise disease diagnosis. Increased knowledge about precision medicine will drive its implementation in clinical settings according to experts.
Research facilities have become essential organizations because they continue to fund genetic research projects and nationwide scientific research programs. Pharmaceutical companies utilize sequencing technology for their biomarker research and drug development processes whereas biotech companies use these tools to create new treatments in various medical fields. Academic institutions contribute through education and exploratory studies, and other specialized laboratories further strengthen operational demand within the Japan Next-Generation Sequencing (NGS) Market.
By Technology:
Whole genome sequencing provides complete genetic information which scientists use to conduct deep research studies and investigate diseases and perform advanced clinical research. The technology becomes more viable for various purposes because processing capabilities have improved and its costs have dropped. The market for genomic mapping services continues to grow because customers demand complete genomic mapping solutions.
Targeted sequencing enables efficient clinical testing and disease-specific research through its focus on selected genomic regions. RNA sequencing serves as a common method for researchers who study gene expression and cellular activity in cancer and molecular biology studies. The Japan Next-Generation Sequencing (NGS) Market experiences different adoption patterns because each technology meets specific operational requirements which create distinct licensing models.
What are the Key Use Cases Driving the Japan Next-Generation Sequencing (NGS) Market?
The primary purpose of sequencing technologies in Japan continues to be cancer genomics research. Genomic profiling allows hospitals and oncology centers to detect actionable mutations which help them choose targeted therapies and deliver precise treatments; as a result this method has become their most frequently used clinical sequencing technique.
The range of applications now extends to both rare disease diagnostics and infectious disease surveillance. Clinical laboratories are increasingly using sequencing to shorten diagnostic timelines for pediatric genetic disorders. Public health agencies and research institutions implement genomic analysis for two purposes: tracking pathogen evolution and enhancing their outbreak response mechanisms.
Researchers study how to advance non-invasive prenatal testing and regenerative medicine applications through their research activities. Japanese biotechnology firms use advanced sequencing technology to develop stem cell characterization methods and validate cell therapy treatments. Pharmacogenomic screening demonstrates strong future value as healthcare providers develop personalized medication selection systems.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 454.3 Million |
|
Market size value in 2026 |
USD 532.6 Million |
|
Revenue forecast in 2033 |
USD 1627.1 Million |
|
Growth rate |
CAGR of 17.30% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Illumina, Thermo Fisher, Roche, Agilent, PacBio, Oxford Nanopore, BGI, Qiagen, PerkinElmer, Bio-Rad, Danaher, Novogene, Macrogen, Eurofins, GATC Biotech |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Sequencing Platforms, Reagents & Kits, Services, Software, Others); By Application (Genomics, Oncology, Clinical Diagnostics, Drug Discovery, Agriculture, Others); By End-User (Hospitals, Research Institutes, Pharma Companies, Biotech Firms, Academic Institutions, Others); By Technology (Whole Genome Sequencing, Targeted Sequencing, RNA Sequencing, Others) |
Which Regions are Driving the Japan Next-Generation Sequencing (NGS) Market Growth?
The Kanto region remains the dominant force in Japan's next-generation sequencing market because it concentrates the country's most advanced clinical genomics infrastructure. The presence of major academic medical centers and national genomic research institutes and global life sciences company headquarters in Tokyo establishes a comprehensive framework for developing and testing sequencing technologies. The combination of government-backed precision medicine programs and early reimbursement policies has enabled faster implementation of clinical processes throughout oncology and rare disease testing. Kanto maintains its dominant position because multiple policies and technical resources and business collaborations support its operations.
Kansai contributes through a different strength which derives from its ability to conduct translational biomedical research and establish partnerships with industrial sectors. The region maintains steady funding for regenerative medicine and university-led genomics and pharmaceutical research which operates at a smaller scale than Kanto. Osaka and Kyoto have established a strong demand for sequencing services through their ongoing clinical trials and their effective collaboration between academic laboratories and biotechnology companies. This makes Kansai a dependable revenue contributor driven by research continuity rather than healthcare system concentration.
Chubu is emerging as the fastest-growing region as regional hospitals expand their ability to perform precision diagnostics tests which were previously limited to metropolitan areas. The local adoption of sequencing technology has grown since 2024 because of recent healthcare digitalization efforts and the establishment of decentralized molecular testing systems. The growing pharmaceutical manufacturing operations in Aichi and nearby prefectures have created a higher need for genomic testing in their drug development processes. Massive opportunities come ahead of investors and technology providers with the regional expansion, their ability to build service networks while capturing first-mover benefits through 2033.
Who are the Key Players in the Japan Next-Generation Sequencing (NGS) Market and How Do They Compete?
The next-generation sequencing market in Japan shows moderate market consolidation because global sequencing technology providers control the majority of platform installations while regional bioinformatics companies and specialized service laboratories battle for market share in data analysis and clinical support services.Incumbents continue to defend their position through platform upgrades, localized service expansion, and stronger regulatory alignment with Japan's clinical diagnostics framework. The market faces disruption from new market entrants who offer affordable sequencing solutions together with cloud-based genomic data analysis services. The sequencing industry faces competition based on sequencing precision and the ability to automate workflows and deliver quick data analysis results and enable clinical-level genomic decision-making.
Illumina maintains its advantage through high-throughput short-read platforms that integrate seamlessly into oncology testing pipelines. Its localized partnerships with Japanese cancer centers strengthen adoption by reducing workflow transition barriers. Thermo Fisher Scientific competes through scalable end-to-end solutions that combine sequencing hardware with companion diagnostic capabilities which enable better integration with pharmaceutical development programs. The company continues expanding through collaborations with regional clinical laboratories.
Oxford Nanopore Technologies, the company, distinguishes itself through its portable real-time sequencing systems which enable decentralized genomic testing. The system's flexibility provides a benefit to healthcare facilities located throughout different regions. Pacific Biosciences develops long-read sequencing technology to provide precise structural variant analysis, which makes the company suitable for research on rare diseases. QIAGEN uses bioinformatics-based interpretation tools to enhance its clinical genomic reporting capabilities which benefit Japanese diagnostic laboratories.
Company List
- Illumina
- Thermo Fisher
- Roche
- Agilent
- PacBio
- Oxford Nanopore
- BGI
- Qiagen
- PerkinElmer
- Bio-Rad
- Danaher
- Novogene
- Macrogen
- Eurofins
- GATC Biotech
Recent Development News
In May 2025, Illumina established a business partnership with Okinawa Institute of Science and Technology. The memorandum of understanding establishes a partnership between Illumina and OIST which aims to advance genomics research in Japan through the use of Illumina's next-generation sequencing technologies and OIST's comprehensive research capabilities to develop Japan's NGS research ecosystem and clinical translation system. Sourcehttps://www.oist.jp/news-center/
In February 2026, IIllumina launched major technology upgrades for its NovaSeq X platform. The newly introduced roadmap system enables scientists to achieve greater genomic project output through faster machine operation and enhanced Q70-quality sequencing capabilities. The system increases performance capabilities for high-scale genomic research projects while promoting advanced NGS technology use in laboratories throughout Japan's research and precision medicine fields.Source https://www.prnewswire.com/
What Strategic Insights Define the Future of the Japan Next-Generation Sequencing (NGS) Market?
The upcoming five to seven years will establish decentralized clinical genomics as the main structural direction for Japan's next-generation sequencing market which enables sequencing operations to reach beyond main academic institutions to regional medical centers and dedicated testing facilities. The main element driving this transition occurs through the combination of reimbursement-supported precision medicine with AI-based genomic analysis systems and the rising requirement for pharmaceutical companies to develop products based on biomarker research. The company will create new revenue streams that will generate ongoing revenue through the sale of consumables and the delivery of cloud analytics and clinical reporting services.
The organization faces a hidden threat because it relies too much on a small group of international sequencing platform vendors. Supply chain disruptions together with pricing pressures and geopolitical trade restrictions will create operational obstacles for Japan's genomic testing ecosystem which will delay clinical implementation while raising system-wide expenses.
The development of population-based genomic databases presents a major opportunity especially through cooperative projects established in Kansai's biomedical research hubs. The datasets will enhance the ability to understand rare diseases while they will also boost the effectiveness of precision oncology. Partners in the market should establish connections with local healthcare organizations while they allocate resources towards developing community-based bioinformatics systems. The establishment of genomic interpretation systems for Japan will strengthen our market position because genomic sequencing will become standard practice in medical settings.
Japan Next-Generation Sequencing (NGS) Market Report Segmentation
By Type
- Sequencing Platforms
- Reagents & Kits
- Services
- Software
By Application
- Genomics
- Oncology
- Clinical Diagnostics
- Drug Discovery
- Agriculture
By End-User
- Hospitals
- Research Institutes
- Pharma Companies
- Biotech Firms
- Academic Institutions
By Technology
- Whole Genome Sequencing
- Targeted Sequencing
- RNA Sequencing
Frequently Asked Questions
Find quick answers to common questions.
The Japan Next-Generation Sequencing (NGS) Market size is USD 1627.1 Million in 2033.
Key segments for the Japan Next-Generation Sequencing (NGS) Market are By Type (Sequencing Platforms, Reagents & Kits, Services, Software, Others); By Application (Genomics, Oncology, Clinical Diagnostics, Drug Discovery, Agriculture, Others); By End-User (Hospitals, Research Institutes, Pharma Companies, Biotech Firms, Academic Institutions, Others); By Technology (Whole Genome Sequencing, Targeted Sequencing, RNA Sequencing, Others).
Major Japan Next-Generation Sequencing (NGS) Market players are Illumina, Thermo Fisher, Roche, Agilent, PacBio, Oxford Nanopore, BGI, Qiagen, PerkinElmer, Bio-Rad, Danaher, Novogene, Macrogen, Eurofins, GATC Biotech
The Japan Next-Generation Sequencing (NGS) Market size is USD 454.3 Million in 2025.
The Japan Next-Generation Sequencing (NGS) Market CAGR is 17.30% from 2026 to 2033.
- Illumina
- Thermo Fisher
- Roche
- Agilent
- PacBio
- Oxford Nanopore
- BGI
- Qiagen
- PerkinElmer
- Bio-Rad
- Danaher
- Novogene
- Macrogen
- Eurofins
- GATC Biotech
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)