Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Size & Forecast:
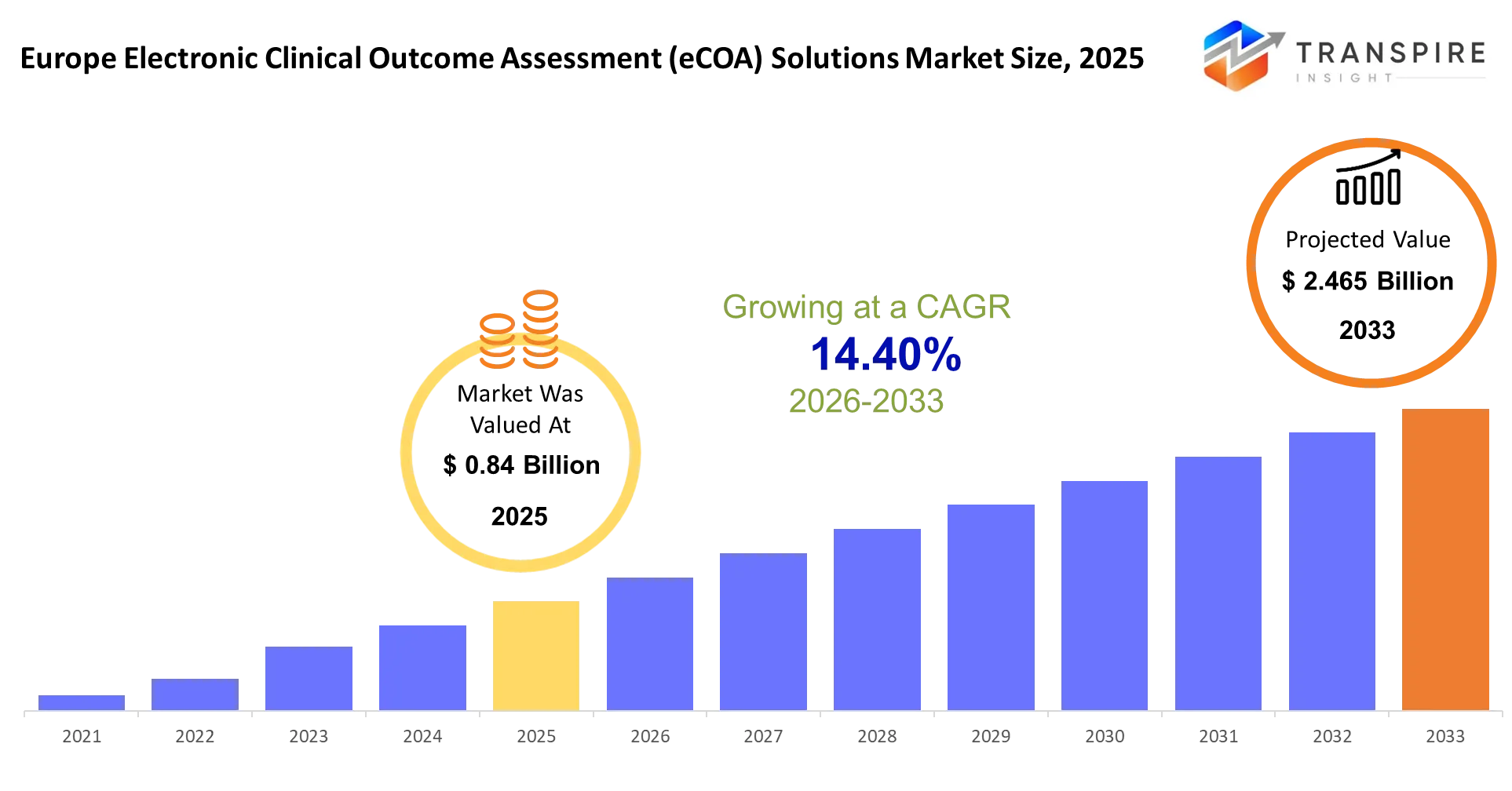
- Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Size 2025: USD 0.84 Billion
- Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Size 2033: USD 2.465 Billion
- Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market CAGR: 14.40%
- Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Segments: By Type (Software, Services, Platforms, Analytics Tools, Others); By Application (Clinical Trials, Patient Monitoring, Data Collection, Drug Development, Real-world Evidence, Others); By End-User (Pharma Companies, CROs, Hospitals, Research Institutes, Biotech Firms, Others); By Deployment (Cloud, On-premise, Hybrid, Mobile-based, Others)
To learn more about this report, Download Free Sample Report
Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Summary:
The Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market size is estimated at USD 0.84 Billion in 2025 and is anticipated to reach USD 2.465 Billion by 2033, growing at a CAGR of 14.40% from 2026 to 2033.
The European electronic clinical outcome assessment eCOA solutions market in digital health shows the shift of European clinical research to patient-centered data collection methods which provide immediate research findings. Sponsors will start using mobile-based reporting tools because trial participants demand transparent reporting methods which improve their experience and decrease their need for site visits. The regulatory authorities will tighten their standards for data integrity requirements and patient privacy standards which will lead to standardized digital system requirements. The development of wearable technology and multilingual interface systems will improve the efficiency of international clinical research operations. Trial participants will demand user-friendly systems which deliver instant results, which will influence how solution providers build their systems and services for different healthcare systems across Europe.
What Has the Impact of Artificial Intelligence Been on the Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market?
The artificial intelligence technology currently establishes new standards for data collection and data interpretation and data usage throughout the European electronic clinical outcome assessment (ECOA) solutions market, which now reaches higher levels of efficiency and accuracy. The European electronic clinical outcome assessment (ECOA) solutions market uses artificial intelligence to accelerate market research through its capability to analyze extensive patient-reported outcomes data at a rate that surpasses traditional methods. Machine learning models will continue to identify hidden patterns in patient behavior, which will enable researchers to create more accurate demand forecasts and improve their ability to track emerging market patterns. The current situation enables stakeholders to develop proactive decision-making capabilities because they can obtain near real-time insights which match ongoing clinical trial requirements.
Artificial intelligence operates within European electronic clinical outcome assessment (ECOA) solutions market systems to establish automated data collection and validation processes as its primary operational function. Automated systems will reduce manual errors while they help solution providers perform compliance checks and boost their production processes. The use of predictive analytics will support resource management and supply chain operations, which will decrease operational expenses while preserving data integrity. AI-powered platforms will develop personalized patient interfaces and engagement methods because personalization now leads the platform development in European markets. The Europe electronic clinical outcome assessment (ECOA) solutions market will become a more flexible and innovative part of digital health through its increasing adoption of intelligent systems.
Key Market Trends & Insights:
- The europe electronic clinical outcome assessment ecoa solutions market shows Western Europe as its leading region because it holds more than 45 percent market share which Germany France and the UK drive during 2025.
- Eastern Europe shows the fastest growth among all regions because its market will expand at a compound annual growth rate above 15 percent between 2025 and 2030.
- The web-based eCOA platforms maintain a market share of approximately 40 percent because their scalability together with centralized data access features provide significant value to users.
- The mobile-based solutions create the second-largest market segment because more people use smartphones while patients find these solutions more convenient.
- The wearable-integrated eCOA tools represent the most rapidly expanding market segment because their demand will continue to grow until 2030 through increased need for remote monitoring.
- Clinical trials dominate with approximately 60% market share because researchers use the method during Phase II and III studies.
- Decentralized trials show the fastest growth because hybrid study models let people participate in research from remote locations.
- The pharmaceutical and biopharmaceutical industries maintain their market leadership with over 55% of the market because they create ongoing requirements for advanced data capture technologies.
- The end-user market for contract research organizations CROs shows its fastest growth because companies increasingly rely on outsourcing their work.
- The firms use technology advancements which include cloud integration and real-time analytics to achieve competitive advantages.
Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Segmentation
By Type:
The software solutions will create user-friendly systems which enable safe acquisition of information, while the services provide assistance for creating systems, teaching users, and conducting system upkeep. The platforms will combine various operations into one centralized system, while the analytics tools will analyze extensive data sets to extract essential business information. The company will provide specialized solutions which fulfill specific requirements of each trial through its additional offerings.
By Application:
Clinical trials will remain the primary application, driven by structured protocols and regulatory oversight. Patient monitoring will expand with remote capabilities, while data collection tools will ensure accuracy and consistency. The field of drug development will progress with the use of real-world evidence because digital endpoints have achieved acceptance in various areas.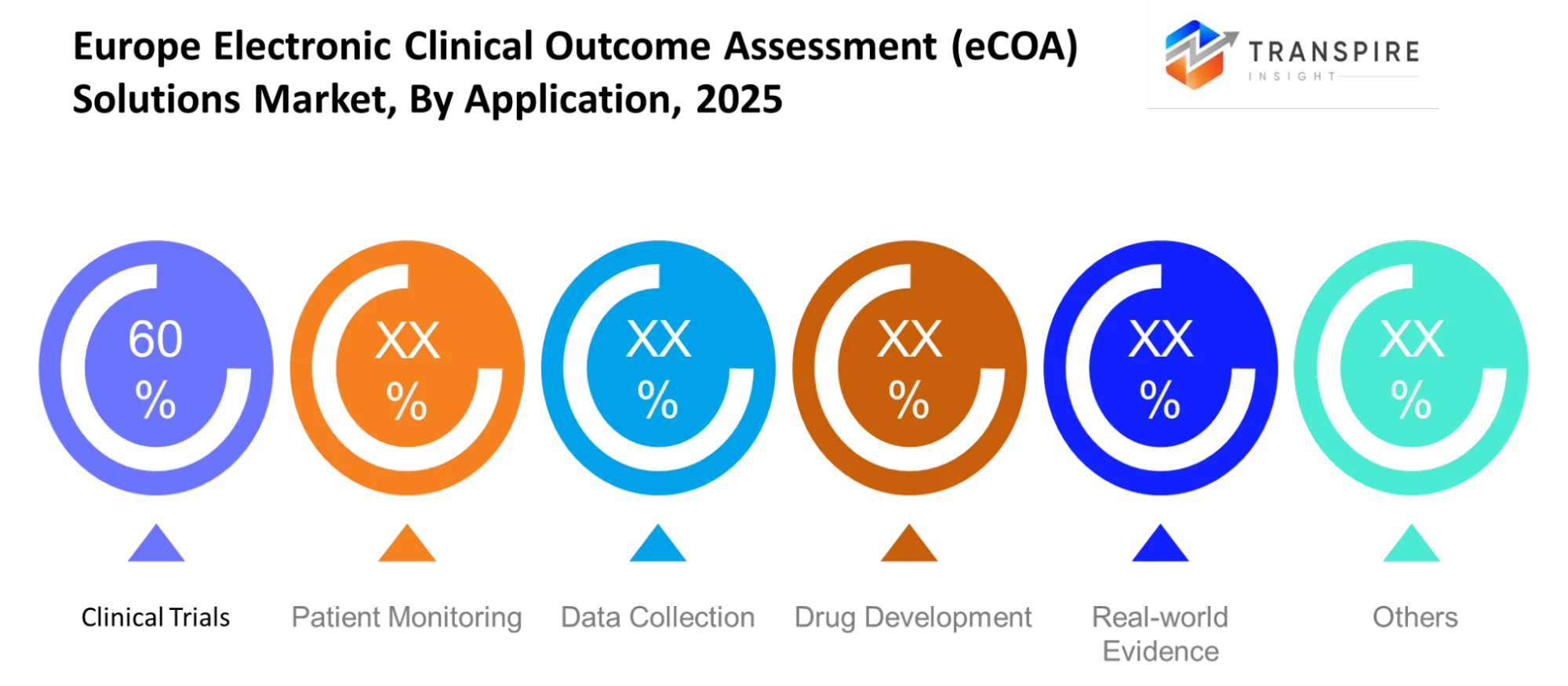
To learn more about this report, Download Free Sample Report
By End-User:
Pharmaceutical companies will drive technology adoption because they invest heavily in large-scale clinical trials, while contract research organizations will increase their usage through their research partnerships. Research institutes and hospitals will implement academic and clinical study systems, while biotech companies will pursue adaptable system options. Other users will include smaller organizations with specialized needs.
By Deployment:
Organizations that need to control their data will choose on-premise systems, while cloud deployment will become the leading choice because it enables system expansion and remote access. The hybrid system will provide organizations with both operational flexibility and protection from security threats, while mobile-based systems will help patients interact with healthcare providers. The organization will select other methods for deployment to meet their specific business operations and necessary standards for compliance with regulations.
What are the Main Challenges for the Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Growth?
The Europe electronic clinical outcome assessment (ECOA) solutions market experiences slow progress because of ongoing technical and operational challenges which require systems to process extensive confidential patient information with complete precision. The existing clinical trial systems require system integration which causes both project delays and higher expenses. The operational capacity of remote trials is hindered by performance issues which include problems with internet connection. The European electronic clinical outcome assessment (ECOA) solutions market needs dependable flexible systems which can operate in various healthcare environments.
The manufacturing and commercialization barriers which exist within the industry serve as important factors which restrict business growth. The data protection and clinical validation standards enforcement system requires European organizations to follow strict regulations which creates continuous compliance obligations that prolong development time. Software development costs together with validation and deployment expenses create financial challenges that decrease provider profit margins. The Europe electronic clinical outcome assessment (ECOA) solutions market presents market difficulties which prevent small businesses from competing, which results in slower innovation and longer delays before new technologies become available.
The existing adoption obstacles demonstrate their presence especially in areas which lack digital infrastructure and which have budget limitations. The healthcare sector and research organizations encounter problems when they try to teach personnel and implement fresh technologies into their current operational procedures. The insufficient availability of qualified workers functions as a second barrier which prevents organizations from implementing new technologies. The market for electronic clinical outcome assessment (ecoa) solutions in Europe will experience ongoing growth limitations which will especially impact emerging European markets.
The market faces two main threats which include competitive threats from other digital health solutions and ongoing changes in regulatory requirements. The industry must deal with two major challenges which include pricing wars and swift technological advancements that require businesses to enhance their products continuously while their business operations become more difficult to manage.
Regional Insights
The Europe Electronic Clinical Outcome Assessment eCOA Solutions Market will show different results across its regions because of three factors which are healthcare spending and rules which are clear and the capability to use digital technology. Western Europe will maintain a strong position due to established clinical research infrastructure while other regions will gradually improve adoption through policy support and technology access which will create a balanced yet uneven regional landscape.
Western Europe will maintain its leadership position because of its ongoing financial support and its advanced research networks and its robust regulatory framework which enables the use of digital technologies in clinical research. The countries of Germany and France and the United Kingdom will maintain their focus on technologies which enhance patient care. This will result in increased demand for electronic data capture solutions which will be needed to conduct research across multiple large research sites.
Eastern Europe will experience rapid growth because it has both affordable business expenses and rising numbers of people who enter international clinical research studies. The government support initiatives combined with improved digital infrastructure will drive higher adoption rates. Local research organizations will modernize their platforms which will enable them to adopt European standards while creating new business opportunities for solution providers.
Recent Development News
In April 2026, Veeva Systems announced enhancements to its clinical trial platform, expanding integration capabilities to improve site collaboration and data flow across eCOA-related systems, aiming to reduce operational delays in digital trials. https://intuitionlabs.ai
In March 2026, Veeva Systems reported strong fiscal 2026 earnings performance, highlighting continued investment in clinical data solutions, including eCOA technologies, to support growing demand for decentralized and patient-centric trials. https://finance.yahoo.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 0.84 Billion |
|
Market size value in 2026 |
USD 0.961 Billion |
|
Revenue forecast in 2033 |
USD 2.465 Billion |
|
Growth rate |
CAGR of 14.40% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Oracle, Medidata, IQVIA, Parexel, ERT, ICON, Signant Health, Veeva Systems, CRF Health, IBM, Dassault Systèmes, ArisGlobal, Clario, Castor, OpenClinica |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Software, Services, Platforms, Analytics Tools, Others); By Application (Clinical Trials, Patient Monitoring, Data Collection, Drug Development, Real-world Evidence, Others); By End-User (Pharma Companies, CROs, Hospitals, Research Institutes, Biotech Firms, Others); By Deployment (Cloud, On-premise, Hybrid, Mobile-based, Others) |
How Can New Companies Establish a Strong Foothold in the Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market?
New companies aiming to enter the europe electronic clinical outcome assessment (ecoa) solutions market will need a focused strategy built on niche targeting and clear differentiation. Emerging players will achieve better outcomes when they dedicate their efforts to specialized areas which include decentralized trials and rare disease studies that still have unaddressed needs. This approach will enable companies to enter markets more quickly while they adopt current industry patterns and focus on specific market development possibilities.
Innovation will play a central role in gaining traction within the europe electronic clinical outcome assessment (ecoa) solutions market. Startups that develop AI-driven data validation tools with multilingual patient interfaces will address key pain points such as data accuracy and patient engagement. Signant Health demonstrates how advanced analytics can enhance trial results while Kayentis develops more effective user-focused solutions for clinical research. Companies that are entering the market should develop their competitive edge through two main areas which are predictive analytics and seamless device integration.
European electronic clinical outcome assessment ec oa solutions market position will benefit from strategic partnerships which will enhance market presence. New companies will gain access to existing networks through their partnerships with contract research organizations and pharmaceutical companies which will help them boost market acceptance rates. Companies will achieve better operational performance through technology partnerships which enable them to integrate cloud and wearable technology solutions.
The solution requires both strong compliance frameworks and developed local solutions. Companies that adapt to European regulatory standards while offering flexible, patient-centric platforms will build trust and long-term credibility in this competitive landscape.
Key Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Company Insights
The european electronic clinical outcome assessment (ecoa) solutions market will display a competitive framework which develops through technological strengths and regulatory compliance and the excellence of its provided services. Market participants will focus on improving platform usability and expanding service networks which will help them establish stronger positions in clinical research environments that operate under multiple compliance standards and different operational needs.
The leading companies will maintain their competitive position through their advanced platforms and their strong client relationships and their ongoing development of analytics capabilities. Established firms will maintain their market control through data accuracy improvements which enhance patient engagement tools and their system integration capabilities across multiple countries involved in clinical trial operations.
The emerging companies will enter the market through their development of affordable yet flexible solutions which meet the needs of small trials and specialized therapeutic markets. The market will experience new entry through mobile-based tool innovations and speedier deployment systems. New companies will develop their competitive position through strategic partnerships and their efforts to expand into new geographical markets.
Company List
- Oracle
- Medidata
- IQVIA
- Parexel
- ERT
- ICON
- Signant Health
- Veeva Systems
- CRF Health
- IBM
- Dassault Systèmes
- ArisGlobal
- Clario
- Castor
- OpenClinica
What are the Key Use-Cases Driving the Growth of the Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market?
The market for Europe electronic clinical outcome assessment (ECOA) solutions starts to develop its market base because clinical trials need these solutions to deliver accurate patient data which needs to be confirmed by regulatory bodies for successful study results. The pharmaceutical industry now uses digital outcome assessment tools to replace traditional paper procedures which leads to improved data accuracy and reduced duration of clinical studies. The current trend results in an upsurge of market growth which enables businesses to expand their operations to international research projects.
The European Electronic Clinical Outcome Assessment (ECOA) Solutions Market shows another main application through its support of remote patient observation which functionalizes in both decentralized and hybrid research studies. Healthcare providers are using mobile-based platforms and wearable integrations to track patient symptoms outside clinical settings. This approach not only enhances patient engagement but also ensures continuous data collection which supports better clinical decisions and long-term treatment evaluation.
The European Electronic Clinical Outcome Assessment (ECOA) Solutions Market expands because it enables organizations to generate real-world evidence and conduct post-marketing research studies. Organizations use digital platforms to collect continuous patient feedback after drug approval which helps them develop better treatment methods while satisfying regulatory requirements. The current use case provides opportunities which enable researchers to use data analytics for studying larger groups of patients.
Research institutes and biotechnology companies use these solutions for their initial drug research projects which focus on specialized treatment fields. Digital health systems now utilize scalable platforms which include multilingual support to establish international research studies while accommodating future system development needs.
Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market Report Segmentation
By Type
- Software
- Services
- Platforms
- Analytics Tools
- Others
By Application
- Clinical Trials
- Patient Monitoring
- Data Collection
- Drug Development
- Real-world Evidence, Others
By End-User
- Pharma Companies
- CROs
- Hospitals
- Research Institutes
- Biotech Firms
- Others
By Deployment
- Cloud
- On-premise
- Hybrid
- Mobile-based
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market size for the Market will be USD 2.465 Billion in 2033.
Key Segments for the Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market are By Type (Software, Services, Platforms, Analytics Tools, Others); By Application (Clinical Trials, Patient Monitoring, Data Collection, Drug Development, Real-world Evidence, Others); By End-User (Pharma Companies, CROs, Hospitals, Research Institutes, Biotech Firms, Others); By Deployment (Cloud, On-premise, Hybrid, Mobile-based, Others).
Major Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market players are Oracle, Medidata, IQVIA, Parexel, ERT, ICON, Signant Health, Veeva Systems, CRF Health, IBM, Dassault Systèmes, ArisGlobal, Clario, Castor, OpenClinica.
The Current Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market size is USD 0.84 Billion in 2025.
The Europe Electronic Clinical Outcome Assessment (eCOA) Solutions Market CAGR is 14.40%.
- Oracle
- Medidata
- IQVIA
- Parexel
- ERT
- ICON
- Signant Health
- Veeva Systems
- CRF Health
- IBM
- Dassault Systèmes
- ArisGlobal
- Clario
- Castor
- OpenClinica
Recently Published Reports
-
Apr 2026
3D Optical Profiler Market
3D Optical Profiler Market Size, Share & Analysis Report By Type (Desktop 3D Optical Profiler, and Portable 3D Optical Profiler), By Technology (Confocal Technology, and White Light Interference), By End-Use Industry (Manufacturing, Research Institutions, Automotive, Aerospace and Defense, Medical Devices, and Other), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Depth Sensor Market
Depth Sensor Market Size, Share & Analysis Report By Type (Infrared Depth Sensors, Time-of-Flight (ToF) Sensors, Stereo Vision Sensors, Structured Light Sensors, Ultrasonic Depth Sensors), By Application (Automotive, Robotics, Gaming, Consumer Electronics, Industrial Automation, Healthcare, Security & Surveillance, Others), By End Users (Automotive Manufacturers, Consumer Electronics Companies, Healthcare Providers, Industrial Companies, Security Agencies, Gaming Companies, Robotics Companies, Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Digital Manufacturing Market
Digital Manufacturing Market Size, Share & Analysis Report By Component (Hardware, Software, and Services), By Technology (Robotics, 3D Printing, Internet of Things (IoT), and Others), By Application (Automotive and Transportation, Aerospace and Defense, Consumer Electronics, Industrial Machinery, and Others), By Process Type (Computer-Based Designing, Computer-Based Simulation, Computer 3D Visualization, Analytics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Digital Visa Services Market
Digital Visa Services Market Size, Share & Analysis Report By Type (Individual Travelers, Group Travelers), By Application (Tourism, Business Travel, Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031


