Asia Pacific Prefilled Syringe Market Size & Forecast:
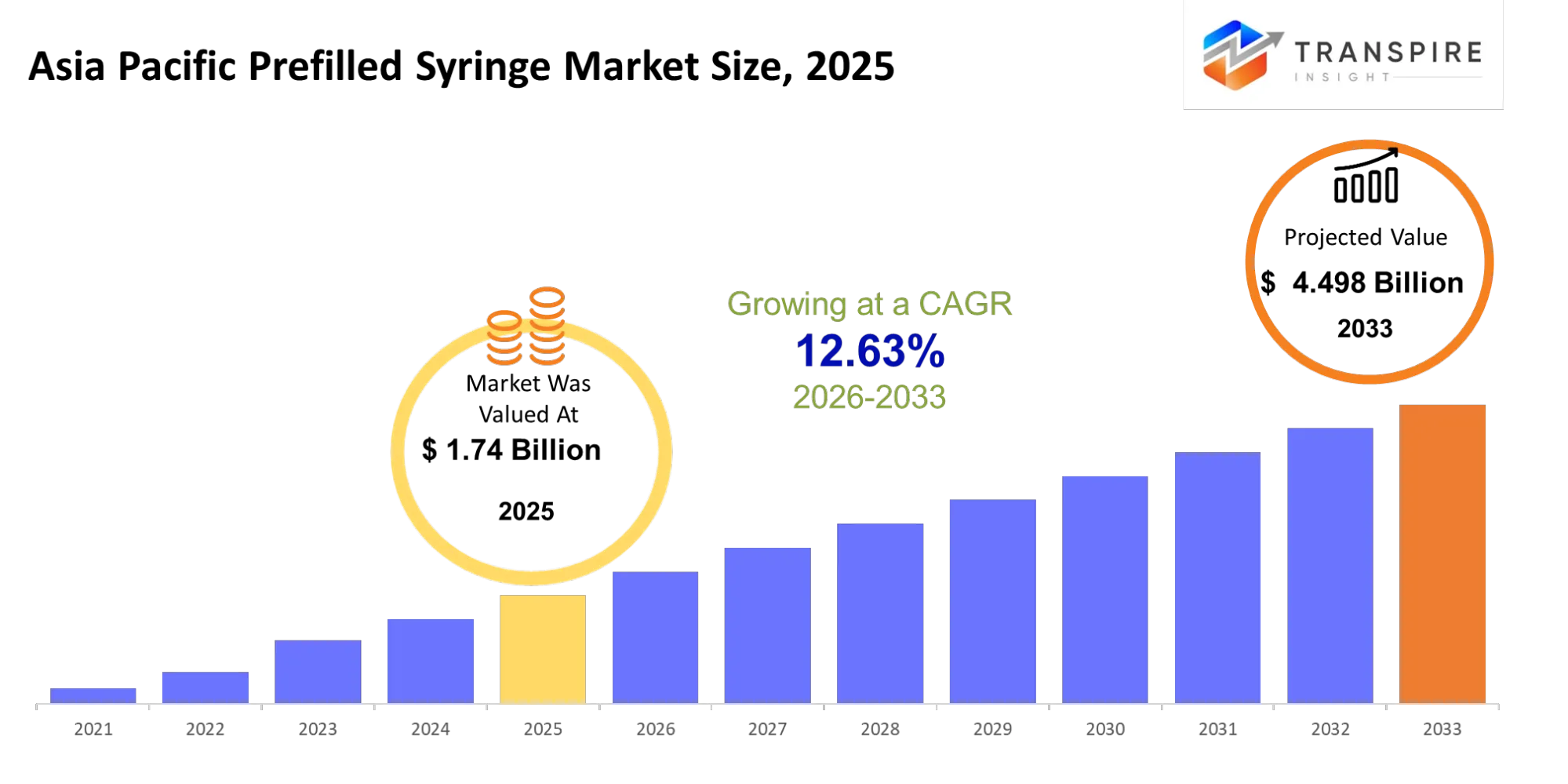
- Asia Pacific Prefilled Syringe Market Size 2025: USD 1.74 Billion
- Asia Pacific Prefilled Syringe Market Size 2033: USD 4.498 Billion
- Asia Pacific Prefilled Syringe Market CAGR: 12.63%
- Asia Pacific Prefilled Syringe Market Segments: By Type (Glass Syringes, Plastic Syringes, Dual-chamber Syringes, Disposable Syringes, Safety Syringes, Others); By Application (Vaccines, Biologics, Insulin, Anticoagulants, Hormones, Others); By End-User (Hospitals, Clinics, Pharma Companies, Homecare, Research Institutes, Others); By Design (Single-dose, Multi-dose, Custom, Standard, Ready-to-use, Others)
To learn more about this report, Download Free Sample Report
Asia Pacific Prefilled Syringe Market Summary
The Asia Pacific Prefilled Syringe Market was valued at USD 1.74 Billion in 2025. It is forecast to reach USD 4.498 Billion by 2033. That is a CAGR of 12.63% over the period.
The Asia Pacific prefilled syringe market addresses a practical challenge in modern drug delivery: getting injectable therapies to patients quickly, accurately, and with minimal preparation error. Hospitals and specialty clinics and home-care providers depend on these systems to enhance dosing efficiency while minimizing contamination risks and achieving safer medication delivery for biologics and vaccines and emergency therapeutics. They decrease treatment duration through their application in actual healthcare environments while maintaining high safety standards and regulatory compliance requirements.
The market has moved away from its previous glass-based systems during the last three to five years because modern polymer syringes and safety-engineered devices provide better compatibility and stability requirements for biologic formulations. The transition to ready-to-administer formats began after COVID-19 vaccinations because healthcare systems needed to address traditional fill-finish capacity limits across their networks. Pharmaceutical manufacturers have developed new injectable delivery systems because healthcare decentralization and stricter regulations on medication error reduction have created new requirements. The prefilled syringe market is growing because more high-value biologics enter regional supply chains, which leads to increased packaging innovation spending and greater revenue prospects for device manufacturers.
Key Market Insights
- Japan maintained its top status in the Asia Pacific Prefilled Syringe Market until 2025, when it reached almost 32% market share, due to its strong biologics manufacturing capabilities.
- The Chinese market will experience its most rapid growth until 2030, because the country is developing its injectable drugs and making major pharmaceutical investments.
- India has become a major driver of growth, because its vaccine production facilities are expanding and its contract manufacturing operations are increasing.
- The industry distribution in 2025 showed that glass prefilled syringes obtained 58% market share, because they had proven compatibility with high-value injectable drugs.
- Plastic polymer-based syringes achieved the second-largest market share, because their design features allowed for better break resistance and created multiple usage options.
- The dual-chamber prefilled syringe market will experience its highest growth rate until 2030, because there is increasing demand for lyophilized biologics delivery systems.
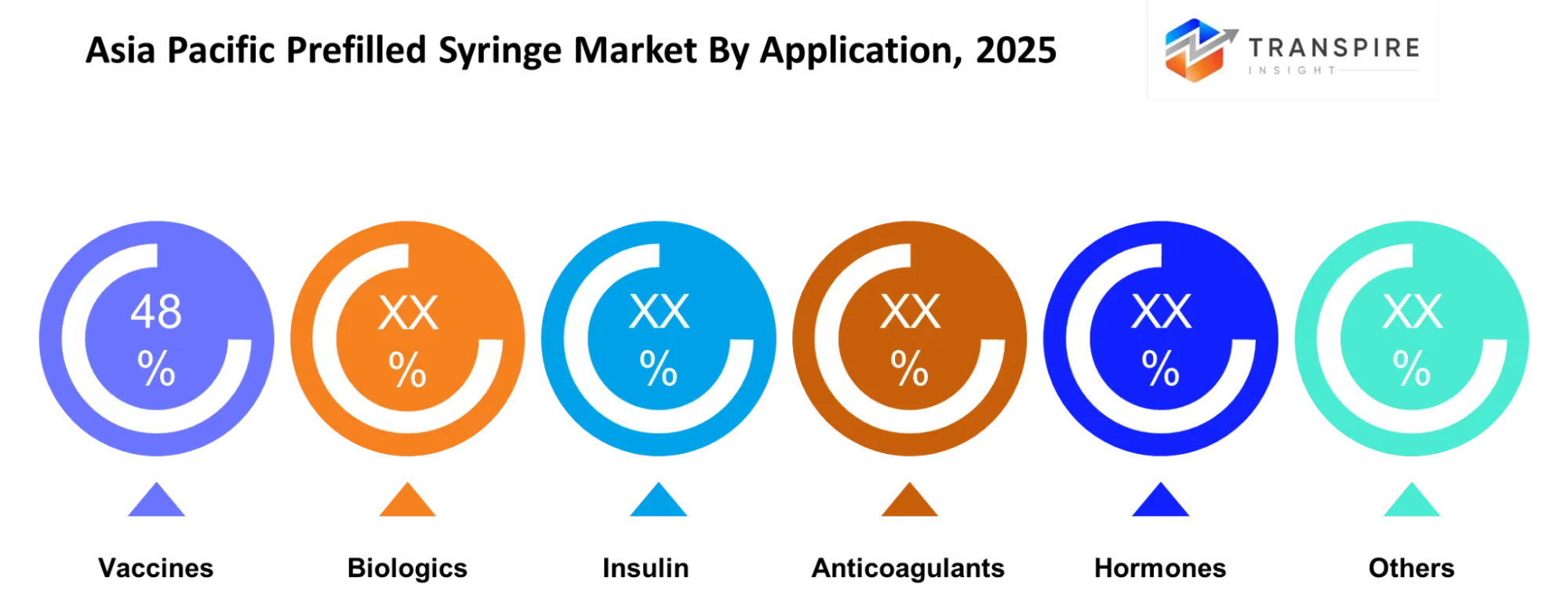
- The national immunization program expansion resulted in vaccination and immunization applications capturing approximately 36% market share in 2025.
- The fastest-growing application segment for biologics and biosimilars results from the rapid approval process of specialty pharmaceutical products.
- The hospital and specialty clinic sectors together held approximately 48% market share in 2025 because they treated high volumes of injectable patients.
- The largest increase in falls-related incidents has primarily targeted areas such as homes, residential communities, and major roadways.
What are the Key Drivers, Restraints, and Opportunities in the Asia Pacific Prefilled Syringe Market?
The growth of the Asia Pacific prefilled syringe market reaches its highest point because essential biologics and biosimilars together with specialty injectable therapies drive their use through all regional pharmaceutical development pipelines.After Japan and China and South Korea simplified their biologic approval processes through post-pandemic healthcare self-sufficiency initiatives they created manufacturing pathways which expedited local product development. The preferred delivery method for prefilled syringes exists because they cut down dosing preparation time while they lower contamination chances and they enhance precise instruction execution for complicated drug formulations. The special delivery systems enable higher product adoption because they support pharmaceutical companies to charge higher prices through their unique delivery methods.
The region's most critical structural challenge exists because it lacks sufficient resources to manufacture advanced polymer syringes and sterile injectable packaging through high-quality fill-finish production facilities. The establishment of production lines that meet compliance standards demands both significant financial resources and specialized technical knowledge together with a prolonged period for regulatory approval processes. The organization needs to develop infrastructure systems while it expands its workforce capabilities to solve these particular limitations. The situation compels regional manufacturers to keep using foreign components which leads to higher expenses and longer production times and decreased revenue opportunities because it postpones their product releases.
A significant chance exists through the growth of self-administered chronic illness treatments which can benefit both India and Southeast Asia. The development of connected drug delivery systems through smart injectable devices that include adherence tracking capabilities creates additional value. The regional healthcare systems which implement decentralized treatment approaches will benefit from the advanced prefilled platforms which enable them to achieve better market reach over time.
What Has the Impact of Artificial Intelligence Been on the Asia Pacific Prefilled Syringe Market?
Manufacturers of prefilled syringes throughout the Asia Pacific region now utilize artificial intelligence and advanced digital systems to enhance their production quality and sterility assurance and supply chain precision. AI-powered visual inspection systems perform automatic defect detection during high-speed fill-finish operations by detecting microcracks and particulate contamination and plunger misalignment with higher precision than standard manual inspection methods. Leading pharmaceutical packaging facilities have achieved reduced batch rejection rates while their production uptime has increased as a result of this system. Smart manufacturing platforms utilize machine learning technology to enhance filling line calibration processes while operators use real-time monitoring to maintain strict control over their entire sterilization cycle and environmental assessment operations.
Predictive analytics is becoming particularly valuable in equipment maintenance. Machine learning models analyze vibration patterns, pressure fluctuations, and thermal data from aseptic filling systems to forecast component wear before breakdowns occur. The organization achieved continuous operations while minimizing unplanned downtime through this solution which also decreased maintenance expenses and ensured more predictable production levels. Digital twins are also being tested to simulate syringe filling performance and identify process inefficiencies before full-scale deployment.
Manufacturers face a critical obstacle because AI-ready manufacturing infrastructure requires expensive integration expenses. Manufacturers in Asia Pacific continue to use outdated fill-finish systems which lack sufficient sensors and data systems for effective model development, which hinders their ability to transition to digital operations.
Key Market Trends
- The pharmaceutical companies in the region have stopped importing syringe systems since 2022 because they want to cut supply interruptions and they need to speed up their process of bringing biologics to market.
- The period between 2021 and 2025 saw rapid growth in polymer syringe usage because SCHOTT Pharma and other manufacturers introduced break-resistant designs which worked for delicate biologic products.
- After 2021, Japan and South Korea established stronger regulations for injectable packaging which required manufacturers to improve their automated inspection systems and contamination detection capabilities.
- Healthcare providers started using ready-to-administer syringe formats more frequently during the COVID-19 vaccine rollout because they wanted to decrease staff errors throughout vaccine delivery.
- Gerresheimer and other companies began increasing their production facility investments across Asian countries from 2023 forward to improve their supply chain disruption management capabilities.
- Pharmaceutical buyers have shifted procurement strategies since 2022, increasingly favoring long-term supply contracts over spot purchasing to secure sterile component availability.
- The development of dual-chamber syringes has progressed rapidly since 2024 because biologics manufacturers needed better stability solutions for their lyophilized and reconstituted injectable products.
- Home-care treatment models expanded throughout India and Southeast Asia after 2023 which resulted in device manufacturers creating ergonomic self-injection devices that meet customer needs.
- The major industry companiesBecton Dickinson and Terumo Corporation have redirected their research and development investments towards safety-engineered syringe systems since 2022.
Asia Pacific Prefilled Syringe Market Segmentation
By Type :
The Asia Pacific Prefilled Syringe Market depends on type-based segmentation because product choice requires assessment of drug compatibility and safety needs and storage conditions. The glass syringes continue to lead the market because their essential components provide stability and chemical resistance which manufacturers need to create sensitive injectable drugs. Pharmaceutical manufacturers often choose glass formats for biologics and specialty drugs where they need to keep product purity throughout the entire shelf life.
Plastic syringes have started to receive more attention because their lightweight design and improved break resistance make them easier to transport and handle. The medical field requires dual-chamber syringes more because certain drugs need their active ingredients to remain separate until doctors administer the medication. Disposable and safety syringes are also seeing wider acceptance due to infection prevention standards, while other specialized designs support niche therapeutic requirements across regional healthcare systems.
By Application :
The increasing use of prefilled syringes in various therapeutic fields demonstrates the growing popularity of Application-based segmentation throughout the Asia Pacific region. The vaccine market holds a substantial revenue share because both developing and developed countries expand their vaccination programs. The prefilled delivery system enables healthcare providers to achieve more effective large-scale vaccine administration through its capacity to enhance dosing precision while decreasing preparation requirements.
The increasing number of chronic diseases drives faster adoption of biologics and insulin treatments because patients now prefer self-administered injectable solutions. The delivery system enables anticoagulants and hormone therapies to achieve better treatment results through its ability to deliver precise dosage amounts. The prefilled systems provide modern treatment delivery systems a practical solution for emergency medications and specialty therapies because they offer both convenience and lower chances of contamination.
To learn more about this report, Download Free Sample Report
By End-User :
The Asia Pacific Prefilled Syringe Market shows different demand patterns through its end-user segmentation which shows how demand differs between various healthcare delivery settings. Hospitals remain the primary users of these systems because their high patient volume needs fast and efficient medication delivery systems. Emergency departments and surgical units and inpatient care facilities depend on prefilled syringes which decrease preparation mistakes and enhance operational processes.
The rise of clinics and homecare environments marks a major trend because treatment delivery now takes place closer to patients. People show growing confidence in self-injection therapies for chronic health condition management which leads to rapid growth in the homecare sector. Pharmaceutical companies use prefilled systems to create distinct product identities which help patients use their products more easily. Research institutes use these devices as tools for their clinical studies and controlled dosing applications.Other end users include specialty centers which focus on providing precise therapeutic treatment through targeted drug delivery.
By Design :
The design segmentation needs to adapt for different treatment needs which require flexible injectable drug delivery systems. Hospitals and homecare facilities prefer single-dose syringes because their user-friendly design decreases contamination risks while providing precise dosage measurement. The specific designs deliver high value to medical treatments which demand precise drug delivery to achieve successful treatment results.
Multi-dose designs serve applications requiring repeated use over short treatment periods, offering practical advantages in certain clinical settings. Pharmaceutical companies are increasingly adopting custom designs to develop delivery systems which match their unique formulation requirements. The standard designs enable extensive commercial applications because their manufacturing processes maintain cost efficiency while healthcare facilities prefer ready-to-use syringes because they simplify preparations and speed up patient care processes.
What are the Key Use Cases Driving the Asia Pacific Prefilled Syringe Market?
The primary application of prefilled syringes in Asia Pacific regions continues to focus on hospitals which use these syringes for vaccine and biologic therapy administration. The requirement for precise dosing during high-volume clinical operations together with the need for rapid vaccine preparation and minimal contamination risks creates the most significant market demand for this method of vaccination and specialized injectable treatment distribution.
Specialty clinics and home-care facilities now implement new applications which support diabetes management and rheumatoid arthritis treatment and oncology support therapies. Healthcare providers in Japan, China and India currently adopt these use cases because they enable outpatient treatment which decreases hospital demand and allows patients to administer their treatment safely.
The introduction of advanced combination products such as connected injection devices with adherence monitoring capabilities and dual-chamber systems for lyophilized biologics represents a new use case development. These formats are still early-stage in regional adoption but show strong potential as pharmaceutical developers invest in complex injectable formulations and regulators place greater emphasis on patient-centric drug delivery technologies.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1.74 Billion |
|
Market size value in 2026 |
USD 1.956 Billion |
|
Revenue forecast in 2033 |
USD 4.498 Billion |
|
Growth rate |
CAGR of 12.63% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
India, China, Japan, and Australia |
|
Key company profiled |
BD, Gerresheimer, Schott, West Pharmaceutical, Nipro, Terumo, Stevanato Group, Catalent, AptarGroup, Baxter, Fresenius Kabi, Pfizer, Novartis, Roche, Sanofi |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Glass Syringes, Plastic Syringes, Dual-chamber Syringes, Disposable Syringes, Safety Syringes, Others); By Application (Vaccines, Biologics, Insulin, Anticoagulants, Hormones, Others); By End-User (Hospitals, Clinics, Pharma Companies, Homecare, Research Institutes, Others); By Design (Single-dose, Multi-dose, Custom, Standard, Ready-to-use, Others) |
Which Regions are Driving the Asia Pacific Prefilled Syringe Market Growth?
The Asia Pacific prefilled syringe market which East Asia controls shows its maximum market share through Japan, China and South Korea because these nations possess established pharmaceutical production networks and their advanced systems for injectable drug manufacturing. The Japanese PMDA and Chinese NMPA regulatory bodies impose strict requirements which compel manufacturers to achieve higher sterility standards while they adopt advanced drug delivery systems. The region operates through multiple connections that link biologics developers with packaging technology suppliers and automated fill-finish facilities which enable companies to produce their products at commercial scale. The combined system of this ecosystem enables quick product introductions which maintain financial backing for the development of advanced syringe manufacturing methods.
Southeast Asia holds the second-largest position, but its strength comes from operational consistency rather than manufacturing scale. The pharmaceutical packaging sector in Singapore, Thailand and Malaysia operates under stable conditions because these countries maintain predictable regulatory systems and attract foreign investments which support the development of sterile production facilities. This region relies on contract manufacturing services and constant supply chain operations for multinational pharmaceutical companies while East Asia depends on innovation to drive its market leadership. The gradual development of healthcare infrastructure and economic stability make this area a reliable source for regional market income.
South Asia currently experiences its highest growth rates because India expands its vaccine production and injectable drug export operations. The recent implementation of production-linked incentive programs together with investments in sterile fill-finish facilities has created faster progress development at local capacity building. Domestic pharmaceutical companies are increasingly shifting toward higher-value injectable delivery formats to compete globally. The period from 2026 to 2033 will provide investors and new market entrants with strong chances to develop advanced packaging technologies and polymer syringe manufacturing and digital manufacturing solutions.
Who are the Key Players in the Asia Pacific Prefilled Syringe Market and How Do They Compete?
The Asia Pacific prefilled syringe market shows moderate consolidation because a few global packaging specialists handle high-end production while regional manufacturers produce standard injectable products. The market is not being disrupted by low-cost entrants because sterile fill-finish production needs both strict regulatory compliance and advanced inspection systems and high capital expenditures. The main method of competition now revolves around technology differentiation which applies especially to polymer syringe design and contamination control and product compatibility with sensitive biologic formulations. Pharmaceutical companies now consider geographic manufacturing proximity as an essential requirement for achieving supply chain resilience and obtaining faster delivery times.
SCHOTT Pharma uses material science innovation to compete through its high-performance polymer syringe platforms designed for biologics which need superior break resistance and chemical stability. Its expansion across Asia focuses on strengthening local production support for regional biologics manufacturers. Gerresheimer differentiates itself through integrated containment solutions that combine syringe systems with advanced inspection and automation capabilities, giving pharmaceutical clients tighter process control from development through commercialization.
Becton Dickinson dedicates its research efforts to developing safety-engineered devices which include needle protection systems that comply with updated clinical administration requirements. This advantage enables hospitals to procure products through channels which base their purchasing decisions on medication safety standards. Terumo Corporation uses its extensive domestic healthcare connections and accurate manufacturing capabilities to operate in Japan's regulated injectable treatment market while Nipro Corporation expands its business through regional production facilities which enhance supply reliability for its Asian pharmaceutical clients.
Company List
- BD
- Gerresheimer
- Schott
- West Pharmaceutical
- Nipro
- Terumo
- Stevanato Group
- Catalent
- AptarGroup
- Baxter
- Fresenius Kabi
- Pfizer
- Novartis
- Roche
- Sanofi
Recent Development News
In August 2025, SCHOTT announced the launch of syringe and cartridge glass tubing production in India. The expansion at its Jambusar, Gujarat facility made SCHOTT the first company to locally manufacture high-precision syringe glass tubing in India, strengthening Asia Pacific’s regional supply chain for prefilled syringes and supporting growing demand for GLP-1 and biologic injectable therapies. Source https://www.schott.com/
In October 2025, SCHOTT Pharma launched its 5.5 ml large-volume prefillable staked-needle glass syringe and expanded its strategic compatibility partnership with Ypsomed. The product’s integration with Ypsomed’s YpsoMate® 5.5 autoinjector accelerates large-volume biologic self-administration and supports broader adoption of advanced prefilled syringe systems across Asia Pacific homecare markets. Source https://www.schott-pharma.com/
What Strategic Insights Define the Future of the Asia Pacific Prefilled Syringe Market?
The Asia Pacific prefilled syringe market is undergoing structural changes during the next five to seven years toward high-value specialized delivery platforms which medical systems use to deliver biologics and self-administered therapies and digitally enabled treatment systems. The region now leads its market away from generic injectables which have high production volumes toward specialty drugs which need advanced dosing systems that match specific materials and safety components. The market will reward manufacturers who create advanced formulation solutions which meet emerging pharmaceutical needs.
The company faces a hidden danger because it relies on medical-grade polymer resins and elastomer components and specialized coatings from only a few suppliers. The pharmaceutical industry depends on these materials for medical equipment production. The production schedule will be affected by any delays in these essential materials which will result in expensive production halts for pharmaceutical companies. The Indian and Southeast Asian markets present a major opportunity for connected prefilled injection systems which provide adherence tracking. The region now experiences expansion in outpatient chronic care services.
Biologics manufacturers should establish regional co-development partnerships which businesses should treat as their primary focus. The initial drug development pipeline integration will establish strong switching barriers together with long-term business contracts for commercial supply which will anticipate future market competition.
Asia Pacific Prefilled Syringe Market Report Segmentation
By Type
- Glass Syringes
- Plastic Syringes
- Dual-chamber Syringes
- Disposable Syringes
- Safety Syringes
By Application
- Vaccines
- Biologics
- Insulin
- Anticoagulants
- Hormones
By End-User
- Hospitals
- Clinics
- Pharma Companies
- Homecare
- Research Institutes
By Design
- Single-dose
- Multi-dose
- Custom
- Standard
- Ready-to-use
Frequently Asked Questions
Find quick answers to common questions.
The Asia Pacific Prefilled Syringe Market size is USD 4.498 Billion in 2033.
Key segments for the Asia Pacific Prefilled Syringe Market are By Type (Glass Syringes, Plastic Syringes, Dual-chamber Syringes, Disposable Syringes, Safety Syringes, Others); By Application (Vaccines, Biologics, Insulin, Anticoagulants, Hormones, Others); By End-User (Hospitals, Clinics, Pharma Companies, Homecare, Research Institutes, Others); By Design (Single-dose, Multi-dose, Custom, Standard, Ready-to-use, Others).
Major Asia Pacific Prefilled Syringe Market players are BD, Gerresheimer, Schott, West Pharmaceutical, Nipro, Terumo, Stevanato Group, Catalent, AptarGroup, Baxter, Fresenius Kabi, Pfizer, Novartis, Roche, Sanofi.
The Asia Pacific Prefilled Syringe Market size is USD 1.74 Billion in 2025.
The Asia Pacific Prefilled Syringe Market CAGR is 12.63% from 2026 to 2033.
- BD
- Gerresheimer
- Schott
- West Pharmaceutical
- Nipro
- Terumo
- Stevanato Group
- Catalent
- AptarGroup
- Baxter
- Fresenius Kabi
- Pfizer
- Novartis
- Roche
- Sanofi
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)